Advanced Synthesis of 1,2,3,4-Tetrahydrocinnoline Scaffolds for Oncology Drug Discovery
Introduction to Next-Generation Cinnoline Scaffold Synthesis
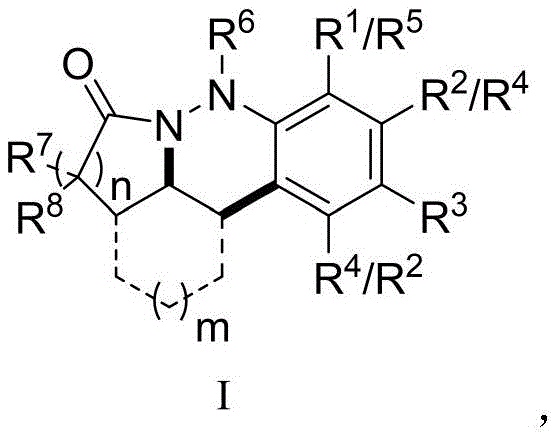
The continuous evolution of nitrogen-containing heterocyclic compounds remains a cornerstone of modern medicinal chemistry, particularly in the relentless pursuit of novel anticancer therapeutics. As highlighted in recent FDA drug approval analyses, nitrogen heterocycles constitute a staggering 59% of approved small molecule drugs, a figure that has risen to 88% in the past five years, underscoring their critical role in pharmacological efficacy. Within this vast chemical space, the cinnoline skeleton and its reduced derivatives, specifically the 1,2,3,4-tetrahydrocinnoline framework, have emerged as privileged structures due to their potent anti-inflammatory, antibacterial, and antitumor activities. Patent CN113387955A introduces a groundbreaking synthetic methodology that addresses long-standing challenges in accessing these valuable scaffolds. By leveraging easily oxidized nitrogen-containing unsaturated hydrazides as key substrates, this invention enables the efficient construction of diverse 1,2,3,4-tetrahydrocinnoline derivatives through an elegant in-situ oxidation strategy. This approach not only expands the chemical diversity available for drug discovery but also offers a robust platform for generating candidate molecules with enhanced biological profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinnoline derivatives has been dominated by the von Richter reaction, a classical transformation that involves the cyclization of 2-ethynylbenzenediazonium precursors under strongly acidic conditions. While this method laid the foundation for cinnoline chemistry, it suffers from significant practical drawbacks that hinder its application in modern process chemistry. The requirement for highly acidic environments often leads to poor functional group tolerance, limiting the scope of substituents that can be introduced onto the heterocyclic core. Furthermore, the reliance on diazonium intermediates poses serious safety concerns due to their inherent instability and potential explosiveness, especially when scaling up to industrial quantities. Alternative methods developed to access multi-substituted cinnolines have attempted to mitigate these issues, yet few have successfully combined mild reaction conditions with the use of stable, commercially accessible starting materials. Consequently, the synthesis of complex tetrahydrocinnoline skeletons, which are vital for exploring new biological spaces, has remained a synthetically challenging bottleneck for both academic researchers and industrial process chemists.
The Novel Approach
In stark contrast to these traditional limitations, the synthetic protocol disclosed in patent CN113387955A represents a paradigm shift towards milder and more versatile chemistry. This novel approach utilizes unsaturated hydrazides as the primary building blocks, which are subjected to an in-situ oxidation process to generate an electron-deficient diene containing a dinitrogen unit. This reactive intermediate subsequently undergoes an intramolecular [4+2] polar cycloaddition with an olefin amine moiety, effectively closing the ring to form the desired 1,2,3,4-tetrahydrocinnoline skeleton. The brilliance of this method lies in its operational simplicity and exceptional substrate scope; it tolerates a wide array of substituents including halogens, alkyl groups, alkoxy chains, and even complex fused aromatic systems like naphthalene and carbazole. By avoiding harsh acids and unstable diazonium salts, this route provides a safer, more reliable pathway for generating high-purity intermediates essential for the development of next-generation antitumor agents.
Mechanistic Insights into TEMPO-Catalyzed Oxidative Cyclization
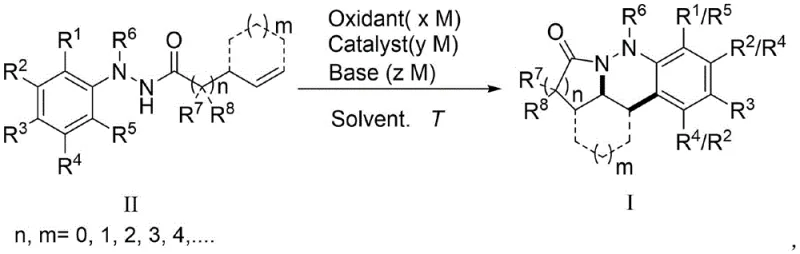
The core of this innovative synthesis relies on a sophisticated oxidative cyclization mechanism mediated by nitroxyl radical catalysts, specifically TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and its derivatives such as 4-AcNH-TEMPO or 4-OH-TEMPO. The reaction initiates with the oxidation of the unsaturated hydrazide substrate by a stoichiometric oxidant, such as TEMPO+BF4- or PhI(OAc)2, in the presence of a catalytic amount of the nitroxyl radical. This oxidation step generates a highly reactive electron-deficient diene species containing a dinitrogen functionality, which serves as the key electrophilic partner in the subsequent cyclization event. The presence of a base, typically an inorganic carbonate like K2CO3 or Cs2CO3, facilitates the deprotonation steps necessary to drive the equilibrium forward and neutralize acidic byproducts. The reaction proceeds through a concerted or stepwise [4+2] polar cycloaddition pathway, where the newly formed diene reacts with the tethered olefinic amine to construct the tetrahydrocinnoline core with high stereocontrol. This mechanistic pathway is distinct from radical-mediated processes, offering superior regioselectivity and minimizing the formation of unwanted polymeric side products.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the specificity of the TEMPO-mediated oxidation. Unlike transition metal-catalyzed couplings which often leave behind trace metal contaminants requiring costly removal steps, this organocatalytic system generates benign organic byproducts that are easily separated during workup. The compatibility of the reaction with various solvents, ranging from polar aprotic acetonitrile to chlorinated solvents like dichloroethane, allows for fine-tuning of the reaction kinetics to suppress side reactions such as over-oxidation or polymerization. The ability to perform this transformation under an inert atmosphere of nitrogen or argon further protects sensitive functional groups from degradation. For R&D directors focused on purity specifications, this mechanism ensures that the resulting 1,2,3,4-tetrahydrocinnoline scaffolds meet stringent quality standards without the need for extensive downstream purification, thereby streamlining the overall development timeline for new drug candidates.
How to Synthesize 1,2,3,4-Tetrahydrocinnoline Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the precise weighing of the unsaturated hydrazide precursor, which acts as the foundational scaffold for the final heterocycle. This substrate is combined with the TEMPO catalyst system and a suitable oxidant in a dry reaction vessel, ensuring that moisture does not interfere with the oxidation potential of the reagents. The choice of solvent is critical, with acetonitrile often providing the optimal balance of solubility and reaction rate for this transformation. Once the reagents are mixed, the system is sealed and purged with protective gas to create an oxygen-free environment, preventing uncontrolled radical pathways that could lead to decomposition. The reaction is then heated or cooled to the specified temperature range, typically between ambient and moderately elevated temperatures, and monitored via TLC or HPLC until the starting material is fully consumed. Following the reaction, standard workup procedures involving concentration and column chromatography yield the target compound in high purity.
- Prepare the reaction mixture by adding unsaturated hydrazide precursor, TEMPO-based catalyst, inorganic base, and oxidant into a dry reaction vessel containing an appropriate organic solvent such as acetonitrile.
- Seal the reaction tube and replace the atmosphere with protective gas like nitrogen or argon, then stir the mixture at a controlled temperature ranging from -80°C to 200°C until the starting material is fully consumed.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the high-purity 1,2,3,4-tetrahydrocinnoline target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TEMPO-catalyzed synthesis offers substantial strategic advantages over legacy manufacturing routes. The elimination of hazardous diazonium intermediates significantly reduces the safety risks associated with production, lowering insurance costs and simplifying regulatory compliance regarding explosive materials. Moreover, the use of stable, commercially available unsaturated hydrazides as starting materials ensures a reliable supply chain, mitigating the risk of raw material shortages that often plague specialized heterocyclic synthesis. The mild reaction conditions also translate to reduced energy consumption, as the process does not require extreme heating or cryogenic cooling, contributing to a lower carbon footprint and aligning with global sustainability goals. These factors collectively enhance the economic viability of producing 1,2,3,4-tetrahydrocinnoline derivatives at scale.
- Cost Reduction in Manufacturing: The organocatalytic nature of this process eliminates the need for expensive transition metal catalysts such as palladium or rhodium, which are not only costly to purchase but also require rigorous and expensive removal steps to meet pharmaceutical residual metal limits. By utilizing inexpensive TEMPO derivatives and simple inorganic bases, the direct material cost of the synthesis is drastically reduced. Additionally, the high yields reported in the patent examples, often exceeding 90% for various substrates, minimize waste generation and improve the overall atom economy of the process. This efficiency directly translates to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against varying substrate structures means that a single standardized protocol can be applied to produce a wide library of analogues without needing to re-optimize conditions for each new derivative. This flexibility is crucial for supply chain continuity, as it allows manufacturers to quickly pivot production between different cinnoline variants based on market demand without significant downtime or retooling costs. The compatibility with common industrial solvents and reagents further ensures that sourcing logistics remain straightforward and resilient to geopolitical disruptions affecting specialty chemical supplies.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen challenges, particularly with exothermic oxidation reactions. However, the controlled nature of the TEMPO-mediated oxidation described in this patent allows for safe scale-up, as the heat generation can be effectively managed within standard reactor configurations. The absence of heavy metal waste streams simplifies wastewater treatment and disposal, reducing the environmental burden and associated compliance costs. This green chemistry profile makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer sustainable production solutions to their multinational clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 1,2,3,4-tetrahydrocinnoline scaffolds. These answers are derived directly from the technical disclosures and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing drug discovery pipelines or manufacturing portfolios.
Q: What are the advantages of this TEMPO-catalyzed method over classical von Richter synthesis?
A: Unlike the classical von Richter synthesis which requires harsh acidic conditions and unstable diazonium precursors, this novel method utilizes mild oxidative conditions with stable unsaturated hydrazides, offering broader substrate compatibility and safer operational parameters for large-scale manufacturing.
Q: Can this synthetic route accommodate complex fused ring systems?
A: Yes, the methodology demonstrates excellent tolerance for diverse substituents and fused ring systems, including naphthalene and carbazole frameworks, allowing for the creation of structurally complex candidates essential for modern oncology drug screening.
Q: Is the process suitable for commercial scale-up?
A: The process utilizes readily available reagents and avoids explosive intermediates, making it highly amenable to scale-up from laboratory grams to multi-ton commercial production while maintaining high purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,4-Tetrahydrocinnoline Supplier
As the demand for complex nitrogen heterocycles continues to surge in the oncology sector, partnering with an experienced CDMO is critical for translating innovative chemistry into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2,3,4-tetrahydrocinnoline intermediate meets the highest international standards. We understand the nuances of handling sensitive oxidative chemistries and have the technical expertise to optimize this TEMPO-catalyzed process for maximum efficiency and safety at any scale.
We invite you to collaborate with us to unlock the full potential of this novel synthetic route for your drug development projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can reduce your overall manufacturing expenses. Please contact us today to request specific COA data for our available cinnoline derivatives and to discuss detailed route feasibility assessments for your custom synthesis needs. Let us be your strategic partner in delivering high-quality pharmaceutical intermediates to the global market.
