Revolutionizing Zolpidem Intermediate Production via Green One-Step Cyclization Technology
Introduction to Advanced Pharmaceutical Intermediate Synthesis
The pharmaceutical industry continuously seeks robust methodologies for producing critical active pharmaceutical ingredient (API) intermediates with enhanced efficiency and environmental compliance. Patent CN107383005B introduces a groundbreaking preparation method for 6-methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridine-3-acetic acid, a pivotal precursor in the synthesis of Zolpidem, a widely prescribed sedative-hypnotic agent. This compound plays an indispensable role in treating various sleep disorders, including episodic and transient insomnia, by selectively binding to omega-1 receptor subtypes in the central nervous system. The structural integrity and high purity of this intermediate are paramount for ensuring the safety and efficacy of the final drug product. As depicted in the molecular structure below, the imidazopyridine core requires precise construction to maintain pharmacological activity.
![Chemical structure of 6-methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridine-3-acetic acid](/insights/img/zolpidem-intermediate-synthesis-supplier-pharma-20260308203845-01.png)
Historically, the manufacturing landscape for this key intermediate has been fraught with challenges related to complex multi-step sequences and the utilization of hazardous chemicals. The innovation presented in this patent addresses these systemic issues by proposing a streamlined synthetic pathway that not only achieves exceptional purity levels exceeding 99% but also aligns with modern green chemistry principles. For R&D directors and procurement specialists, understanding the nuances of this technology is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS). The transition from legacy processes to this novel approach represents a significant leap forward in process chemistry, offering a viable solution for both laboratory-scale optimization and large-scale commercial production without compromising on quality or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 6-methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridine-3-acetic acid have relied on convoluted reaction sequences that introduce substantial operational risks and environmental burdens. One prominent historical route involves a Mannich reaction followed by cyanation and hydrolysis, necessitating the use of highly toxic reagents such as formaldehyde, dimethylamine, and sodium cyanide. These substances pose severe safety hazards to personnel and require extensive waste treatment protocols to mitigate environmental impact. Furthermore, alternative pathways utilizing nitrostyrene derivatives often demand expensive catalysts like DMAP and corrosive acylating agents such as acetyl chloride, which complicate equipment maintenance and increase capital expenditure. Another documented approach employs oxalyl chloride and hydrazine hydrate, reagents known for their pungent odors, high toxicity, and carcinogenic potential, rendering them increasingly undesirable in regulated pharmaceutical manufacturing environments.
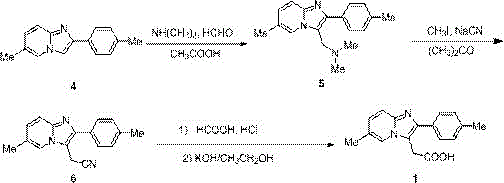
The cumulative effect of these traditional strategies is a production process characterized by long cycle times, low overall yields, and difficult purification steps that struggle to remove trace impurities effectively. The reliance on volatile organic solvents and aggressive chemical conditions exacerbates the generation of hazardous three wastes (wastewater, waste gas, and solid waste), creating significant compliance hurdles for manufacturers aiming to meet stringent environmental regulations. Consequently, the industry has faced a persistent need for a safer, more economical, and technically superior alternative that can deliver high-purity intermediates without the associated liabilities of legacy chemistry. The inefficiencies inherent in these older methods directly translate to higher production costs and supply chain vulnerabilities, prompting a strategic shift towards more sustainable synthetic technologies.
The Novel Approach
In stark contrast to the cumbersome procedures of the past, the method disclosed in patent CN107383005B utilizes a direct, one-step condensation reaction that dramatically simplifies the manufacturing workflow. This innovative process involves the reaction of 3-halo-4-(4-methylphenyl)-4-oxobutyric acid with 2-amino-5-methylpyridine in the presence of a mild condensing agent. By eliminating the need for multiple isolation steps and avoiding the use of notorious toxins like cyanides and formaldehyde, this approach fundamentally reshapes the economic and safety profile of the synthesis. The reaction proceeds under remarkably mild conditions, typically within a temperature range of -10°C to 100°C, with optimal results observed between 60°C and 70°C, thereby reducing energy consumption and thermal stress on the equipment. The use of benign solvents, including water or aqueous alcohols, further underscores the environmental friendliness of this technique, facilitating easier solvent recovery and waste management.
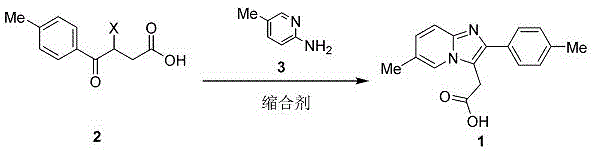
The operational simplicity of this novel route allows for straightforward scale-up from benchtop to industrial reactors without the need for specialized containment systems required for handling highly toxic gases or liquids. The direct formation of the imidazopyridine ring system ensures that the product is generated with high selectivity, minimizing the formation of side products that typically complicate downstream purification. This efficiency not only accelerates the time-to-market for the final API but also enhances the reliability of the supply chain by reducing the number of potential failure points in the synthesis. For procurement managers, the shift to readily available and non-hazardous starting materials translates into more stable pricing and reduced logistical complexities. Ultimately, this method represents a paradigm shift towards sustainable pharmaceutical manufacturing, balancing high performance with ecological responsibility.
Mechanistic Insights into One-Step Condensation Cyclization
The chemical elegance of this synthesis lies in its well-defined mechanistic pathway, which leverages the intrinsic reactivity of the starting materials to drive the cyclization efficiently. The process initiates with the nucleophilic attack of the amino nitrogen atom from 2-amino-5-methylpyridine on the electrophilic alpha-carbon of the carbonyl group in the 3-halo-4-(4-methylphenyl)-4-oxobutyric acid. This interaction triggers an SN2 nucleophilic substitution reaction, resulting in the displacement of the halogen atom and the concurrent elimination of a hydrogen halide molecule to form a key intermediate. The lone pair of electrons on the nitrogen atom subsequently engages in P-pi conjugation with the adjacent aromatic system, facilitating a rearrangement that stabilizes the molecular structure. This electronic redistribution is critical for setting up the final ring closure, ensuring that the thermodynamic driving force favors the formation of the desired imidazopyridine core over potential linear byproducts.
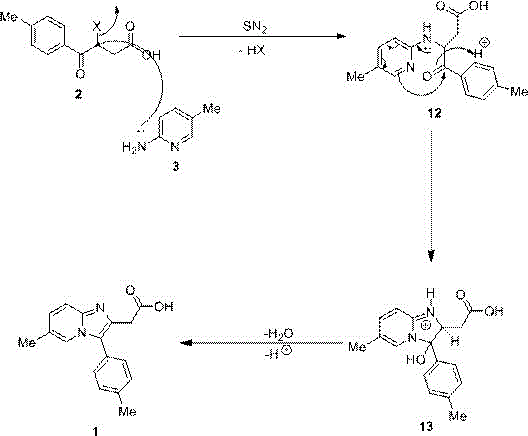
Following the initial substitution and rearrangement, the intermediate undergoes a dehydration step accompanied by proton transfer to yield the final target molecule. Understanding this mechanism is vital for R&D teams tasked with process optimization, as it highlights the importance of maintaining appropriate pH levels and stoichiometric ratios to maximize conversion rates. The choice of condensing agent plays a pivotal role in neutralizing the generated acid and driving the equilibrium towards product formation without inducing degradation of the sensitive functional groups. By controlling the reaction kinetics through temperature modulation and solvent selection, manufacturers can effectively suppress the formation of oligomeric impurities or hydrolyzed side products. This deep mechanistic understanding allows for precise tuning of the process parameters, ensuring consistent batch-to-batch reproducibility and adherence to strict quality specifications required for pharmaceutical intermediates.
How to Synthesize 6-Methyl-2-(4-Methylphenyl)imidazo[1,2-a]pyridine-3-acetic Acid Efficiently
Implementing this advanced synthesis strategy requires careful attention to reaction conditions and reagent quality to fully realize its potential benefits. The standardized protocol involves dissolving the halo-keto acid and aminopyridine in a selected solvent system, followed by the controlled addition of a base such as sodium carbonate or sodium hydroxide. The mixture is then heated to the optimal temperature range and maintained for a specific duration to ensure complete conversion. Detailed operational guidelines regarding stirring rates, addition sequences, and workup procedures are essential for achieving the reported high yields and purity. For a comprehensive breakdown of the specific experimental steps and parameter ranges validated in the patent examples, please refer to the structured guide below.
- Prepare the reaction mixture by combining 3-halo-4-(4-methylphenyl)-4-oxobutyric acid and 2-amino-5-methylpyridine in a suitable solvent such as water or alcohol.
- Add a condensing agent like sodium carbonate or sodium hydroxide and heat the mixture to a temperature between 60°C and 70°C.
- Maintain the reaction for 6 to 7 hours, then adjust pH to precipitate the high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel synthesis method offers profound commercial advantages that extend far beyond the laboratory, directly impacting the bottom line and operational resilience of pharmaceutical manufacturers. By transitioning away from multi-step processes reliant on scarce or regulated toxic chemicals, companies can achieve substantial cost savings in raw material procurement and waste disposal. The elimination of expensive catalysts and hazardous reagents like sodium cyanide and oxalyl chloride removes significant overhead costs associated with special handling, storage, and regulatory compliance. Furthermore, the simplified one-pot nature of the reaction reduces labor requirements and equipment occupancy time, leading to improved throughput and asset utilization. These factors collectively contribute to a more competitive cost structure, enabling suppliers to offer high-quality intermediates at more attractive price points while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The streamlined single-step process drastically cuts down on utility consumption and solvent usage compared to traditional multi-stage syntheses. By avoiding the need for complex purification columns and extensive solvent exchanges, the overall material cost is significantly lowered. The use of inexpensive and abundant starting materials further insulates the production cost from market volatility associated with specialty chemicals. Additionally, the reduction in hazardous waste generation minimizes the financial burden of environmental remediation and disposal fees, creating a leaner and more efficient manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing non-toxic and stable raw materials ensures a more robust and predictable supply chain, free from the disruptions often caused by strict controls on precursor chemicals. The ability to utilize common solvents like water and ethanol reduces dependency on specialized solvent supply lines, enhancing logistical flexibility. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. Moreover, the safety profile of the process reduces the risk of unplanned shutdowns due to safety incidents, thereby guaranteeing consistent product availability.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of corrosive or explosive reagents make this process inherently safer and easier to scale from pilot plants to full commercial production. The compatibility with standard stainless steel reactors eliminates the need for exotic metallurgy, lowering capital investment barriers. From an environmental perspective, the significant reduction in toxic emissions and wastewater load aligns perfectly with global sustainability goals and increasingly stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the feasibility and benefits of this technology, we have compiled answers to common inquiries regarding the synthesis and application of this intermediate. These responses are grounded in the technical data provided in the patent documentation and reflect practical considerations for industrial implementation. Understanding these details is key to making informed decisions about process adoption and supplier qualification. The following section addresses specific concerns related to purity, safety, and scalability that are often raised during technical audits.
Q: How does this new method improve upon traditional Zolpidem intermediate synthesis?
A: Unlike conventional multi-step routes requiring toxic reagents like cyanide or formaldehyde, this patent describes a direct one-step cyclization that significantly reduces hazardous waste and simplifies purification.
Q: What are the purity specifications achievable with this process?
A: The disclosed method consistently yields product with purity levels reaching up to 99%, minimizing the need for complex downstream processing.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, inexpensive solvents like water or ethanol, and avoids highly corrosive reagents, making it ideal for safe and scalable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methyl-2-(4-Methylphenyl)imidazo[1,2-a]pyridine-3-acetic Acid Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this cutting-edge synthesis technology to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 6-methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridine-3-acetic acid meets the highest industry standards. Our dedication to green chemistry and process safety aligns perfectly with the advantages offered by this patent, allowing us to provide a sustainable and reliable supply source for your Zolpidem production needs.
We invite you to engage with our technical procurement team to discuss how this innovative method can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify the quality and consistency of our output. Let us collaborate to drive efficiency and excellence in your pharmaceutical manufacturing operations.
