Advanced Copper-Catalyzed Synthesis of 1,2,4-Thiadiazole Derivatives for Commercial Scale-Up
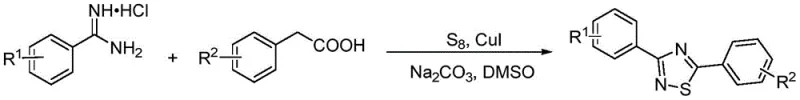
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and cost-effective synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is documented in Chinese patent CN111018807B, which details a robust method for synthesizing 1,2,4-thiadiazole derivatives. Unlike traditional approaches that often rely on expensive or unstable precursors, this invention leverages a direct oxidative coupling strategy between phenylacetic acid compounds and benzamidine hydrochloride compounds. The process utilizes elemental sulfur as an oxidant and a copper-based catalytic system, operating effectively in dimethyl sulfoxide (DMSO) at temperatures ranging from 130 to 140°C. This technological advancement is particularly relevant for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering high-purity heterocycles without the burden of complex multi-step syntheses. By streamlining the construction of the thiadiazole ring, this method addresses critical pain points in both research and development and large-scale manufacturing sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-thiadiazole derivatives has been fraught with significant challenges that hinder their widespread industrial application. Early methodologies, such as those reported by Haruki Eiichi in the late 1960s, required the use of N-chlorobenzamidine and phenylthioamide, necessitating the pre-synthesis of these specialized substrates which adds considerable time and cost to the overall process. Furthermore, more recent literature describes methods involving thioamides and fatty nitriles with iodine as an oxidant, or the use of benzyl bromides with lithium tert-butoxide. These conventional routes often suffer from harsh reaction conditions, the use of toxic solvents, and complicated post-treatment procedures that make purification difficult. The reliance on expensive starting materials and the generation of hazardous waste streams have traditionally restricted the commercial viability of these compounds, creating a bottleneck for procurement managers looking for cost reduction in API manufacturing.
The Novel Approach
The methodology outlined in patent CN111018807B represents a paradigm shift by utilizing readily available and inexpensive starting materials. The core innovation lies in the direct reaction between phenylacetic acid derivatives and benzamidine hydrochlorides in the presence of elemental sulfur and a copper catalyst. This approach eliminates the need for pre-functionalized sulfur-containing substrates, thereby simplifying the supply chain and reducing raw material costs significantly. The reaction conditions are notably milder compared to prior art, proceeding efficiently at 130-140°C without the need for exotic reagents. Moreover, the use of sodium carbonate as a base and DMSO as a solvent provides a stable reaction environment that supports a wide range of substrate substitutions. This versatility allows for the synthesis of various derivatives, including those with methyl, methoxy, and trifluoromethyl groups, ensuring high product selectivity and yield. For supply chain heads, this translates to a more resilient sourcing strategy for high-purity 1,2,4-thiadiazole derivatives.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The success of this synthetic route hinges on the intricate interplay between the copper catalyst and the elemental sulfur oxidant. Mechanistically, the reaction likely proceeds through a copper-mediated activation of the benzylic C-H bond of the phenylacetic acid, followed by an oxidative coupling with the amidine nitrogen. The elemental sulfur serves a dual role, acting as both the sulfur source for the heterocycle and the terminal oxidant to drive the cyclization forward. The copper species, whether CuI, CuBr, or Cu(OAc)2, facilitates the electron transfer processes necessary to form the C-N and C-S bonds that constitute the 1,2,4-thiadiazole ring. This catalytic cycle is highly efficient, requiring only a molar ratio of 0.1 to 0.2 relative to the substrate, which minimizes metal contamination in the final product. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters for specific substrate classes, ensuring that the electronic properties of substituents do not inhibit the catalytic turnover.
Impurity control is another critical aspect of this mechanism that benefits from the specific choice of reagents. The use of sodium carbonate as a mild base helps to neutralize the hydrochloride salt of the benzamidine without promoting side reactions such as hydrolysis or over-oxidation, which are common pitfalls in harsher alkaline media. The reaction temperature window of 130 to 140°C is carefully balanced to provide sufficient energy for the cyclization while preventing the decomposition of sensitive functional groups on the aromatic rings. Post-reaction processing involves standard extraction and column chromatography, which effectively removes residual copper salts and unreacted starting materials. This results in a final product with a clean impurity profile, meeting the stringent purity specifications required for pharmaceutical applications. The ability to tolerate diverse functional groups, such as trifluoromethyl and methoxy moieties, further demonstrates the robustness of this catalytic system against potential side reactions.
How to Synthesize 3,5-Diphenyl-1,2,4-thiadiazole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, precise adherence to the stoichiometric ratios and reaction conditions described in the patent is essential. The process begins with the dissolution of the reactants in DMSO, followed by the sequential addition of the catalyst, oxidant, and base. Maintaining the temperature within the specified range is critical for achieving optimal conversion rates. The detailed standardized synthesis steps provided below outline the exact procedure for preparing the model compound, 3,5-diphenyl-1,2,4-thiadiazole, which serves as a benchmark for evaluating the efficiency of the method.
- Dissolve phenylacetic acid compounds and benzamidine hydrochloride compounds in DMSO solvent at a molar ratio of 1-2: 1.
- Add elemental sulfur powder as oxidant, sodium carbonate as base, and a copper catalyst (CuI, CuBr, or Cu(OAc)2) to the mixture.
- Stir the reaction mixture at 130-140°C for 18-24 hours, then cool, extract, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic method offers substantial benefits that directly impact the bottom line and operational efficiency of chemical manufacturing enterprises. The primary advantage lies in the drastic simplification of the raw material portfolio; by switching to commodity chemicals like phenylacetic acid and benzamidine hydrochloride, companies can mitigate the risks associated with sourcing specialized intermediates. This shift not only lowers the direct material costs but also enhances supply chain reliability by reducing dependence on single-source suppliers for exotic reagents. Furthermore, the elimination of complex pre-synthesis steps shortens the overall production lead time, allowing for faster response to market demands. For procurement managers, this means a more predictable cost structure and the ability to negotiate better terms due to the availability of bulk starting materials.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of inexpensive catalysts and oxidants. Elemental sulfur is one of the cheapest oxidants available, and copper salts are significantly more affordable than precious metal catalysts like palladium or platinum often used in cross-coupling reactions. Additionally, the low catalyst loading (0.1-0.2 equivalents) reduces the cost per kilogram of the final product. The simplified workup procedure, which avoids complex distillation or crystallization steps initially, further lowers utility and labor costs. These factors combine to deliver significant cost reduction in API manufacturing, making the final thiadiazole derivatives more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more stable supply chain. Since the reagents are stable and widely available, there is minimal risk of production delays due to material shortages. The tolerance of the reaction to various substituents means that a single platform technology can be used to produce a wide library of derivatives, reducing the need for multiple distinct manufacturing lines. This flexibility allows supply chain heads to consolidate production and manage inventory more effectively. Moreover, the use of DMSO, a common industrial solvent, ensures that solvent supply is never a bottleneck, unlike specialized fluorinated solvents that might be subject to regulatory or availability constraints.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of hazardous reagents like lithium tert-butoxide or gaseous chlorine. The use of solid sulfur powder and aqueous-compatible workup steps simplifies waste management and aligns with green chemistry principles. The reduced toxicity of the reagent mix lowers the burden on environmental health and safety (EHS) teams, facilitating easier regulatory approval for new facilities. The high atom economy of incorporating sulfur directly into the ring structure minimizes waste generation. Consequently, this method supports the commercial scale-up of complex heterocycles while maintaining a strong commitment to environmental compliance and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations.
Q: What are the key advantages of this 1,2,4-thiadiazole synthesis method?
A: The method utilizes cheap and readily available starting materials like phenylacetic acid and benzamidine hydrochloride. It operates under mild conditions (130-140°C) with high selectivity and avoids the need for pre-synthesized complex substrates found in older methods.
Q: Which catalysts are suitable for this oxidative cyclization?
A: The patent specifies copper salts such as Copper(I) iodide (CuI), Copper(I) bromide (CuBr), or Copper(II) acetate (Cu(OAc)2) as effective catalysts, typically used in low molar ratios relative to the substrate.
Q: Is this process scalable for industrial production?
A: Yes, the process uses inexpensive reagents like sulfur powder and sodium carbonate, and the workup involves standard extraction and chromatography, making it highly suitable for commercial scale-up of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Thiadiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111018807B for developing next-generation therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle copper-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest industry standards. We understand that consistency and quality are paramount for pharmaceutical intermediates, and our dedicated technical team is ready to adapt this robust chemistry to your specific project requirements.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your pipeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs, helping you optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target 1,2,4-thiadiazole derivatives. Let us help you accelerate your development timeline and secure a reliable supply of high-quality intermediates for your global operations.
