Advanced Synthesis of Poly-substituted 1,5-naphthyridines via Green Vitriol Catalysis for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, which serve as the backbone for numerous bioactive molecules. Patent CN101555248B introduces a significant advancement in this domain by disclosing a novel method for preparing poly-substituted 1,5-naphthyridine compounds. This technology leverages a cost-effective catalytic system utilizing green vitriol, also known as ferrous sulfate, to drive the cyclization of 3-aminopyridines with 2-alkenyl aldehydes or ketones. The strategic importance of this patent lies in its ability to generate diverse structural libraries of 1,5-naphthyridines, which are critical intermediates in the development of anti-malarial, anti-inflammatory, and anticancer agents. By shifting away from traditional precious metal catalysts or harsh oxidative conditions, this process offers a more sustainable and economically viable pathway for large-scale manufacturing. For R&D directors and procurement specialists, understanding the nuances of this synthesis route is essential for optimizing supply chains and reducing the overall cost of goods sold for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-naphthyridine derivatives has relied heavily on classical methodologies such as the Skraup, Doebner-von Miller, or Conrad-Limpach-Knorr reactions. While these methods have served the industry for decades, they are plagued by significant inherent limitations that hinder modern commercial production. A primary drawback is the requirement for extremely high reaction temperatures and harsh oxidative conditions, which often lead to poor selectivity and the formation of complex impurity profiles that are difficult to purge. Furthermore, these traditional routes exhibit a marked lack of substrate tolerance, particularly when dealing with amines bearing strong electron-withdrawing groups such as fluorine, nitro, or trifluoromethyl substituents. In many cases, the reaction simply fails to proceed or yields are unacceptably low, restricting the chemical space available for drug discovery teams. Additionally, the Skraup method often produces products with fixed substitution patterns, lacking the flexibility to introduce diverse functional groups at specific positions on the naphthyridine ring, thereby limiting the structural diversity required for modern SAR studies.
The Novel Approach
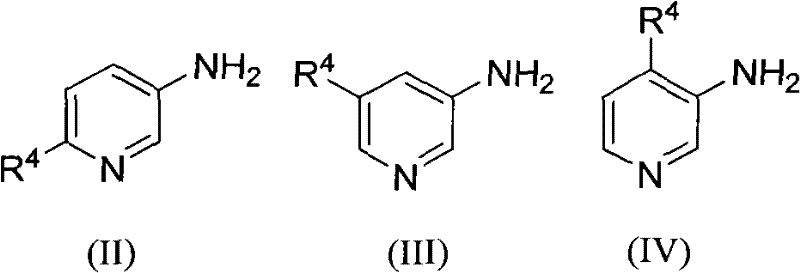
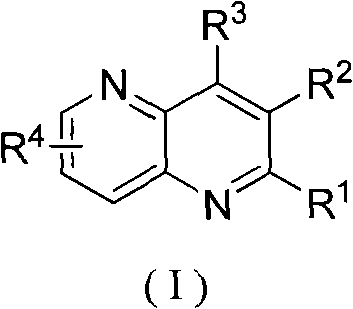
In stark contrast to the constraints of legacy chemistry, the method disclosed in patent CN101555248B offers a versatile and robust alternative that addresses these critical pain points. This novel approach utilizes a ferrous sulfate catalytic system in a sulfuric acid medium, enabling the efficient coupling of various 3-aminopyridines with 2-alkenyl aldehydes or ketones under heating reflux conditions. The beauty of this methodology lies in its exceptional substrate scope, allowing for the introduction of a wide array of substituents at the R1, R2, R3, and R4 positions, including alkyl, alkoxy, halogen, and carboxyl groups. This flexibility empowers medicinal chemists to access previously difficult-to-synthesize analogs, accelerating the lead optimization process. Moreover, the reaction conditions are relatively mild compared to traditional oxidative cyclizations, resulting in cleaner reaction profiles and simplified downstream processing. The ability to synthesize poly-substituted derivatives with high structural diversity makes this technology a powerful tool for building comprehensive compound libraries for biological screening.
Mechanistic Insights into FeSO4-Catalyzed Cyclization
The core of this technological breakthrough resides in the unique role of ferrous sulfate as a Lewis acid catalyst within the acidic reaction medium. During the reaction, the iron species likely coordinates with the carbonyl oxygen of the 2-alkenyl aldehyde or ketone, increasing its electrophilicity and facilitating the nucleophilic attack by the amino group of the 3-aminopyridine substrate. This activation lowers the energy barrier for the initial condensation step, which is often the rate-determining step in such heterocycle formations. Following the condensation, the acidic environment promotes a cascade of cyclization and dehydration events that ultimately close the second ring to form the stable 1,5-naphthyridine core. The use of sulfuric acid not only provides the necessary protons for catalysis but also serves as a solvent that can withstand the elevated temperatures required for reflux, typically ranging from 2 to 20 hours depending on the specific substrate reactivity. This prolonged heating ensures complete conversion of the starting materials, minimizing the presence of unreacted amines which can be challenging to separate from the final product.
From an impurity control perspective, this catalytic system offers distinct advantages over oxidative methods that often generate tarry byproducts. The reaction mechanism proceeds through a well-defined ionic pathway rather than a radical mechanism, which significantly reduces the formation of polymeric side products. The workup procedure described in the patent involves neutralizing the acidic reaction mixture to a pH of neutral, followed by filtration and extraction with organic solvents such as ethyl acetate. This straightforward isolation protocol allows for the efficient removal of inorganic iron salts and acidic residues, yielding a crude product that is amenable to further purification via column chromatography or distillation. The patent data indicates that the final purified products exhibit high structural fidelity, as confirmed by proton NMR spectroscopy, with yields ranging from 50% to 75%. This level of purity and consistency is crucial for pharmaceutical applications where strict impurity specifications must be met to ensure patient safety and regulatory compliance.
How to Synthesize Poly-substituted 1,5-naphthyridine Efficiently
Implementing this synthesis route in a commercial setting requires careful attention to the stoichiometry and reaction parameters outlined in the patent documentation. The process begins with the precise mixing of the 3-aminopyridine derivative and the alkenyl carbonyl component in a molar ratio typically ranging from 1:1.5 to 1:5.0, with a preferred ratio of 1:3.5 to drive the equilibrium towards product formation. The addition of the ferrous sulfate catalyst is critical, with a molar ratio relative to the aminopyridine of between 0.01 and 0.09, ensuring sufficient catalytic activity without excessive metal loading. The reaction is then heated under reflux in sulfuric acid for a duration of 2 to 20 hours, depending on the specific electronic nature of the substrates involved. Upon completion, the mixture is cooled to room temperature and carefully neutralized, followed by extraction and concentration to isolate the enriched material. For detailed standard operating procedures and specific safety guidelines regarding the handling of concentrated sulfuric acid and high-temperature reflux systems, please refer to the technical guide below.
- Mix 3-aminopyridines compound with 2-alkenyl aldehyde or 2-alkenyl ketone in a reactor.
- Add green vitriol (ferrous sulfate) as a catalyst and sulfuric acid, then carry out heating reflux reaction for 2 to 20 hours.
- Cool the system to room temperature, regulate pH to neutral, filter, extract, concentrate, and refine to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ferrous sulfate-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The primary driver for cost reduction is the replacement of expensive transition metal catalysts or specialized oxidants with green vitriol, a commodity chemical that is abundantly available and inexpensive on the global market. This substitution drastically lowers the raw material cost per kilogram of the final intermediate, directly improving the gross margin for the manufacturing process. Furthermore, the simplicity of the workup procedure, which relies on standard neutralization and extraction techniques rather than complex chromatographic separations at an early stage, reduces the consumption of solvents and processing time. These operational efficiencies translate into significant cost savings in terms of utility consumption and labor hours, making the process highly attractive for large-scale production campaigns where even marginal improvements in efficiency can yield substantial financial benefits.
- Cost Reduction in Manufacturing: The utilization of ferrous sulfate as a catalyst eliminates the need for costly precious metals such as palladium or rhodium, which are subject to volatile market pricing and supply constraints. By leveraging a base metal catalyst, the manufacturing process becomes immune to fluctuations in precious metal spot prices, ensuring stable and predictable production costs over the long term. Additionally, the high atom economy of the cyclization reaction minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. The ability to achieve respectable yields of 50% to 75% without the need for exotic reagents further enhances the economic viability of this route, allowing for competitive pricing strategies in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various 3-aminopyridines and 2-alkenyl aldehydes, are commercially available from multiple suppliers, mitigating the risk of single-source dependency. This diversity in the supply base ensures continuity of supply even in the event of disruptions at a specific vendor's facility. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures and production delays. For supply chain heads, this reliability is paramount, as it allows for more accurate forecasting and inventory planning, ensuring that downstream drug substance manufacturing schedules are met without interruption due to intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, as the use of sulfuric acid and reflux conditions is well-established in industrial chemical engineering, allowing for seamless transfer from laboratory to pilot and commercial scale. The absence of hazardous oxidants or unstable intermediates simplifies the safety profile of the plant, reducing the need for specialized containment equipment. From an environmental perspective, the use of iron salts is preferable to heavy metals, as iron is less toxic and easier to manage in wastewater treatment streams. This alignment with green chemistry principles facilitates regulatory approvals and enhances the sustainability profile of the manufacturing site, which is increasingly important for meeting the ESG goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN101555248B, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of this route for specific project requirements. The information covers aspects ranging from catalyst loading to substrate scope, offering a comprehensive overview of the technology's capabilities and limitations.
Q: What are the advantages of using ferrous sulfate in 1,5-naphthyridine synthesis?
A: Ferrous sulfate acts as an inexpensive and environmentally benign catalyst that facilitates the cyclization under reflux conditions, significantly reducing raw material costs compared to precious metal catalysts.
Q: Can this method accommodate diverse substrate structures?
A: Yes, the method allows for various substituents (R1-R4) including alkyl, alkoxy, halogen, and nitro groups, overcoming the limitations of traditional Skraup synthesis which often fails with electron-withdrawing groups.
Q: What is the typical yield range for this synthesis protocol?
A: According to the patent data, the productive rate of the target product after purification ranges from 50% to 75%, depending on the specific substrate and reaction conditions employed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Poly-substituted 1,5-naphthyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our technical team is well-versed in the nuances of heterocyclic chemistry, including the ferrous sulfate-catalyzed synthesis of 1,5-naphthyridines described in patent CN101555248B. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of material we deliver meets the highest industry standards. Our commitment to quality and reliability makes us the preferred choice for pharmaceutical companies seeking a trusted partner for complex intermediate manufacturing.
We invite you to collaborate with us to optimize your supply chain and achieve your cost reduction targets. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how our manufacturing capabilities can enhance your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of chemical expertise and production capacity that will accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
