Optimizing Ritodrine Hydrochloride Production for Commercial Scale-Up and High Purity Standards
The pharmaceutical landscape for anti-preterm labor medications demands rigorous adherence to purity standards and cost-effective manufacturing protocols to ensure global accessibility. Patent CN101239917B introduces a transformative methodology for the preparation of ritodrine hydrochloride, a critical beta-2-adrenoreceptor agonist used to inhibit uterine smooth muscle contraction. This technical insight report analyzes the patented synthesis route, which diverges significantly from traditional multi-step protections, offering a streamlined pathway that enhances both yield and operational simplicity. By leveraging a specific palladium-catalyzed hydrogenation strategy, the process addresses long-standing inefficiencies in the production of this vital pharmaceutical intermediate. For stakeholders evaluating reliable pharmaceutical intermediates supplier options, understanding the mechanistic advantages of this route is essential for strategic sourcing and long-term supply chain planning.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
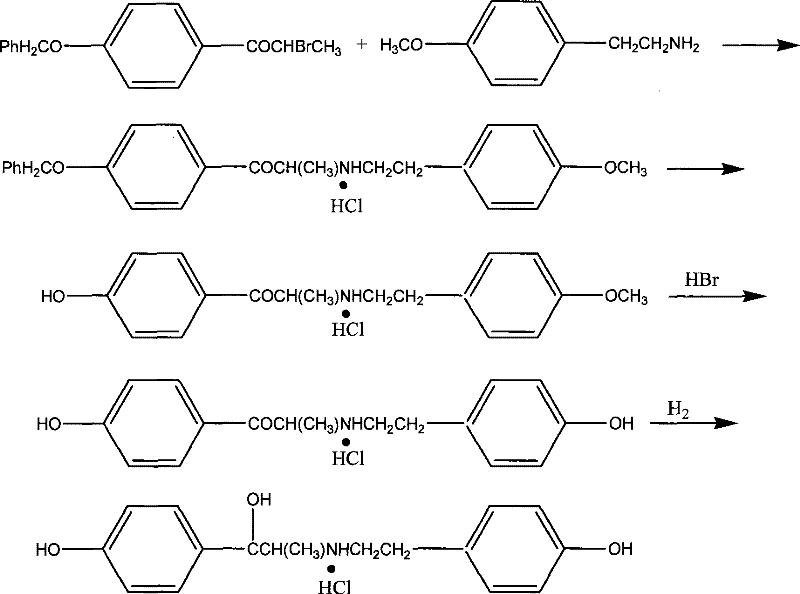
Historically, the synthesis of ritodrine hydrochloride has been plagued by inefficient reaction pathways that rely heavily on the protection and deprotection of phenolic hydroxyl groups. As illustrated in prior art methodologies, traditional routes often involve the condensation of hydroxyl-2-bromobenzene acetone with protected phenolic amines, necessitating complex sequential steps to manage reactivity. These conventional approaches typically result in overall yields that are dismally low, often hovering around merely 20%, which drastically inflates the cost of goods sold and creates significant waste disposal challenges. Furthermore, certain legacy methods depend on the use of controlled substances such as pseudoephedrine, introducing severe regulatory bottlenecks and supply chain vulnerabilities that can disrupt production schedules. The operational complexity of these older routes, characterized by lengthy reaction times and difficult purification processes, renders them economically unviable for modern large-scale cost reduction in API manufacturing initiatives.
The Novel Approach
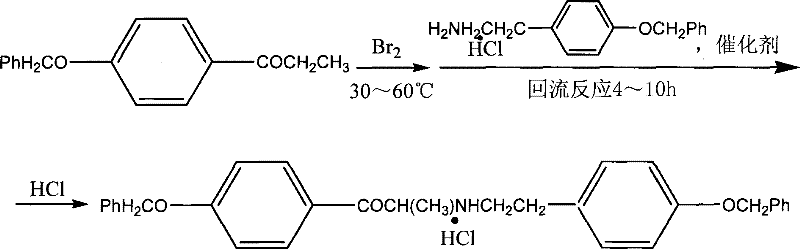
In stark contrast, the patented methodology outlined in CN101239917B employs a direct and robust synthetic strategy that bypasses the need for cumbersome protection groups. This novel approach utilizes p-benzyloxypropiophenone as a key starting material, which undergoes a streamlined bromination and subsequent amination reaction to form a stable intermediate. The elimination of multiple protection-deprotection cycles not only shortens the production cycle significantly but also reduces the consumption of auxiliary reagents and solvents. By focusing on a direct coupling mechanism followed by a single hydrogenation step, the process achieves a level of operational simplicity that is rarely seen in complex pharmaceutical synthesis. This efficiency translates directly into enhanced process reliability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where consistency and throughput are paramount.
Mechanistic Insights into Palladium-Catalyzed Hydrogenation and Coupling
The core of this technological breakthrough lies in the precise control of the catalytic environment during the formation of the key intermediate and the final reduction step. The synthesis begins with the reaction of p-benzyloxypropiophenone with elemental bromine in the presence of anhydrous aluminum trichloride, facilitating a highly selective alpha-bromination. This intermediate is then coupled with p-benzyloxyphenethylamine hydrochloride using a palladium-based catalyst system, such as palladium or bis-triphenylphosphine palladium chloride, under reflux conditions. The use of a mild base like sodium bicarbonate during this coupling phase ensures that the reaction proceeds without degrading the sensitive amine functionality, thereby preserving the structural integrity of the molecule. This careful orchestration of reagents prevents the formation of polymeric byproducts and ensures that the intermediate, 1-p-benzyloxyphenyl-2-(2-p-benzyloxyphenylethylamine)propanone hydrochloride, is obtained with a yield exceeding 70%.
Following the isolation of the intermediate, the final transformation involves a pressurized catalytic hydrogenation that is critical for establishing the final stereochemistry and purity profile of the drug substance. The reaction is conducted using a palladium on carbon catalyst under controlled hydrogen pressure ranging from 1.0 to 20 atm and temperatures between 20 to 80 degrees Celsius. This specific window of operating conditions is engineered to reduce the ketone functionality to the corresponding alcohol without causing over-reduction or hydrogenolysis of the benzyl protecting groups prematurely. The result is a final product with a purity greater than 99.00%, as confirmed by HPLC analysis, demonstrating the robustness of the impurity control mechanism. Such high purity is essential for meeting the stringent regulatory requirements for high-purity ritodrine hydrochloride intended for human therapeutic use.
How to Synthesize Ritodrine Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and safety. The process is divided into two main stages: the formation of the benzyloxy-protected intermediate and the subsequent hydrogenation to the final active pharmaceutical ingredient. Operators must maintain precise temperature controls during the bromination step to prevent exothermic runaways, while the hydrogenation phase requires specialized high-pressure equipment to ensure safe handling of hydrogen gas. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are critical for reproducibility and are outlined in the technical guide below for qualified manufacturing partners.
- React p-benzyloxypropiophenone with bromine and aluminum trichloride, followed by coupling with p-benzyloxyphenethylamine hydrochloride using a palladium catalyst.
- Isolate the intermediate 1-p-benzyloxyphenyl-2-(2-p-benzyloxyphenylethylamine)propanone hydrochloride through acidification and crystallization.
- Perform pressurized catalytic hydrogenation using palladium on carbon at controlled temperatures to yield final Ritodrine Hydrochloride with purity exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their vendor networks. The primary advantage lies in the drastic simplification of the manufacturing process, which inherently reduces the operational burden and the potential for batch failures. By eliminating the need for multiple protection and deprotection steps, the process significantly lowers the consumption of raw materials and utilities, leading to a more favorable cost structure without compromising on quality. This efficiency allows suppliers to offer more competitive pricing models while maintaining healthy margins, which is a key factor in cost reduction in API manufacturing. Furthermore, the reliance on readily available starting materials rather than controlled substances ensures a stable and uninterrupted supply chain, mitigating the risk of regulatory delays.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis route eliminates several expensive and time-consuming unit operations found in conventional methods. By removing the need for complex protection group chemistry, the process reduces the volume of solvents and reagents required, which directly lowers waste treatment costs and raw material expenditure. The higher yields achieved at each step mean that less starting material is needed to produce the same amount of final product, effectively amplifying the value of every kilogram of input. This cumulative efficiency results in substantial cost savings that can be passed down through the supply chain, enhancing the overall economic viability of the drug product.
- Enhanced Supply Chain Reliability: One of the most significant risks in pharmaceutical manufacturing is the dependency on controlled or hard-to-source precursors. This method utilizes p-benzyloxypropiophenone, a commodity chemical that is widely available from multiple global sources, thereby diversifying the supply base and reducing single-source dependency. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates and ensuring that production schedules are met without unexpected interruptions.
- Scalability and Environmental Compliance: The simplicity of the reaction design makes it highly amenable to scale-up from laboratory to commercial production volumes without the need for specialized or exotic equipment. The reduction in the number of chemical steps inherently lowers the environmental footprint of the manufacturing process by decreasing the total amount of chemical waste generated. Additionally, the use of catalytic hydrogenation is a well-established green chemistry technique that avoids the use of stoichiometric reducing agents which often produce heavy metal waste. This alignment with environmental compliance standards facilitates smoother regulatory approvals and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of ritodrine hydrochloride using this advanced methodology. These answers are derived directly from the patent data and technical specifications to provide clarity on process capabilities and quality standards. Understanding these details is vital for partners evaluating the feasibility of integrating this intermediate into their broader pharmaceutical portfolios.
Q: How does this novel synthesis route improve yield compared to conventional methods?
A: Conventional methods often suffer from yields around 20% due to complex protection and deprotection steps. This patented route utilizes a direct coupling and hydrogenation strategy, achieving intermediate yields greater than 70% and final product yields exceeding 50%, significantly enhancing material efficiency.
Q: What are the purity specifications achievable with this manufacturing process?
A: The process is designed to minimize impurity formation through controlled reaction conditions and specific catalyst selection. HPLC analysis of the final product demonstrates purity levels greater than 99.00%, meeting stringent requirements for pharmaceutical intermediates.
Q: Does this method rely on controlled substances or difficult-to-source raw materials?
A: Unlike some prior art methods that utilize controlled precursors like pseudoephedrine, this synthesis starts from readily available p-benzyloxypropiophenone. This ensures a stable supply chain and eliminates regulatory hurdles associated with controlled substance procurement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ritodrine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes in the modern pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and risk-mitigated. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to verify that every batch of ritodrine hydrochloride meets the highest international standards. Our capability to implement complex catalytic processes like the one described in CN101239917B demonstrates our dedication to innovation and quality excellence in the fine chemical sector.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for this essential anti-preterm labor medication. By leveraging our technical expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
