Advanced Synthesis of Styrene Phosphonic Acid: A Strategic Upgrade for Global Supply Chains
The chemical landscape for producing functionalized organophosphorus compounds is undergoing a significant transformation, driven by the urgent need for safer and more efficient manufacturing protocols. Patent CN1254340A introduces a groundbreaking methodology for the formation of 1-phenylvinyl-1-phosphonic acid, commonly known as Styrene Phosphonic Acid (SPA), which addresses critical limitations inherent in legacy production techniques. This intellectual property outlines a sophisticated three-step sequence involving the reaction of phosphorous acid with acetophenone, followed by acylation and subsequent elimination, offering a robust alternative to the hazardous phosphorus trichloride route. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards sustainable chemistry that minimizes toxic by-product formation while enhancing overall process controllability. The strategic implementation of Lewis acid catalysts and immobilized acidic systems allows for precise temperature management and superior impurity profiles, making it an ideal candidate for high-value applications in pharmaceutical intermediates and advanced coating technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Styrene Phosphonic Acid has relied heavily on the reaction between phosphorus trichloride, water, and acetophenone, a pathway fraught with substantial operational hazards and inefficiencies. This traditional method suffers from inherently low yields of isolatable product due to the formation of numerous undesirable by-products that complicate downstream purification efforts. Furthermore, the reaction requires the meticulous and careful addition of water to the reactants to mitigate severe foaming problems and excessive heat development, which poses significant safety risks in large-scale reactors. When phosphorus trichloride reacts with water, it inevitably generates hydrogen chloride gas, a corrosive by-product that necessitates complex removal systems such as vacuum stripping or nitrogen purging to prevent equipment damage and ensure operator safety. These additional engineering controls drastically increase the production cost of SPA and create bottlenecks in supply chain continuity, rendering the conventional process economically unviable for modern, high-throughput manufacturing environments seeking cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel approach detailed in the patent utilizes a much safer and more atom-economical pathway starting with phosphorous acid and acetophenone. This innovative route proceeds through the formation of a stable hydroxy adduct intermediate, which is subsequently reacted with acetic anhydride to form an acyl derivative before undergoing a final elimination step. By shifting the feedstock from phosphorus trichloride to phosphorous acid, the process completely eliminates the generation of hydrogen chloride gas, thereby removing the need for expensive gas scrubbing systems and reducing the environmental footprint of the facility. The use of acetic anhydride as a dehydrating and acylating agent allows for milder reaction conditions, typically ranging from 25°C to 55°C during the initial stages, which significantly lowers energy consumption and thermal stress on the reactor infrastructure. This methodological shift not only enhances the safety profile of the operation but also streamlines the workflow, enabling a more reliable styrene phosphonic acid supplier to meet rigorous delivery schedules without the interruptions caused by hazardous waste handling.
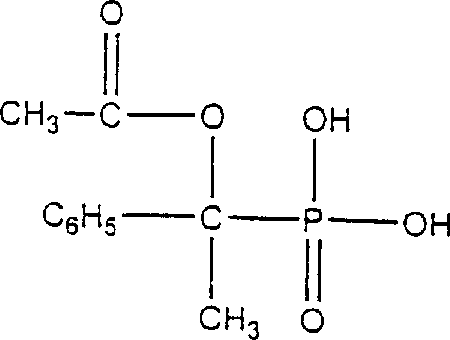
Mechanistic Insights into Lewis Acid-Catalyzed Adduct Formation
The core of this synthetic breakthrough lies in the precise mechanistic control exerted during the initial condensation of phosphorous acid and acetophenone, facilitated by the presence of a Lewis acid catalyst. Typical catalysts employed in this system include metal chlorides and acetates such as zinc chloride, zinc acetate, or aluminum chloride, which activate the carbonyl group of the acetophenone towards nucleophilic attack by the phosphorous acid. This activation lowers the energy barrier for the formation of the hydroxy adduct intermediate, allowing the reaction to proceed efficiently at moderate temperatures between 25°C and 55°C. The choice of catalyst is critical; for instance, zinc acetate has been shown to provide superior selectivity compared to uncatalyzed conditions, minimizing the formation of polymeric side products. The reaction can be conducted in the absence or presence of suitable organic solvents like acetic acid or tetrahydrofuran, providing flexibility in process design. This controlled environment ensures that the stoichiometry is maintained effectively, leading to a higher concentration of the desired intermediate and setting the stage for high-yield conversion in subsequent steps.
Following the formation of the hydroxy adduct, the mechanism proceeds through an acylation step where the intermediate reacts exothermically with acetic anhydride to generate the acyl derivative. This step is crucial for preparing the molecule for the final elimination, as the acetyl group serves as a good leaving group in the presence of an acid catalyst. The subsequent deacylation step involves treating this acyl derivative with an acidic catalyst, preferably an immobilized acid such as acidic clay or ion exchange resin, to remove the acetic acid moiety and generate the vinyl double bond. The use of immobilized catalysts is particularly advantageous because the acid moiety is fixed to a support matrix, allowing it to be easily filtered and removed from the final reaction mixture without requiring neutralization washes that generate salt waste. This mechanistic elegance results in a final product structure that is highly pure, as evidenced by the ability to recrystallize the SPA from solvents like chloroform to remove trace impurities such as unreacted phosphorous acid or Diels-Alder dimers.
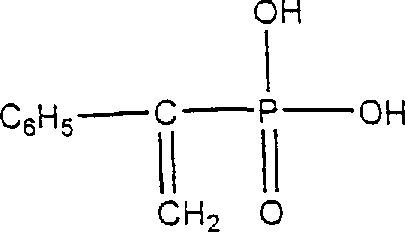
How to Synthesize Styrene Phosphonic Acid Efficiently
The practical execution of this synthesis requires careful attention to thermal management and catalyst selection to maximize yield and purity. The process begins with the mixing of stoichiometric amounts of phosphorous acid and acetophenone in a reactor equipped with agitation and temperature control, followed by the addition of a catalytic amount of a Lewis acid such as zinc acetate. Once the hydroxy adduct is formed, acetic anhydride is added dropwise to manage the exotherm, after which the mixture is heated to facilitate acylation. The final transformation involves the addition of a solid acid catalyst and heating to induce elimination, followed by filtration and solvent removal. For a comprehensive understanding of the specific parameters, reagent ratios, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized technical guide below.
- React stoichiometric phosphorous acid with acetophenone in the presence of a Lewis acid catalyst like zinc acetate at 25-55°C to form a hydroxy adduct intermediate.
- Treat the hydroxy adduct with acetic anhydride to generate the acyl derivative, managing the exothermic reaction carefully.
- Perform deacylation using an immobilized acid catalyst at 70-100°C to eliminate acetic acid and yield the final Styrene Phosphonic Acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring material availability. The elimination of phosphorus trichloride from the supply chain removes a volatile and highly regulated raw material, replacing it with phosphorous acid and acetic anhydride, which are generally more stable and easier to source in bulk quantities globally. This shift significantly reduces the logistical complexity associated with transporting hazardous corrosives and mitigates the risk of supply disruptions caused by regulatory changes or transportation restrictions on dangerous goods. Furthermore, the simplified workup procedure, which relies on filtration of solid catalysts rather than complex gas scrubbing and neutralization steps, translates directly into reduced operational expenditures and shorter batch cycle times. These factors collectively contribute to a more resilient supply chain capable of sustaining long-term production contracts without the volatility associated with legacy chemical processes.
- Cost Reduction in Manufacturing: The transition to this novel pathway eliminates the need for expensive engineering controls required to handle hydrogen chloride gas, such as specialized vacuum systems and corrosion-resistant piping, leading to substantial capital expenditure savings. By utilizing immobilized acid catalysts that can be filtered and potentially regenerated, the process reduces the consumption of consumable reagents and minimizes the volume of aqueous waste streams that require treatment. The higher selectivity of the Lewis acid-catalyzed reaction also means that less raw material is lost to by-product formation, improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. These cumulative efficiencies drive down the total cost of ownership for the manufacturing process, making it highly competitive in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: Sourcing phosphorous acid and acetophenone is generally more straightforward and less prone to geopolitical or regulatory bottlenecks compared to phosphorus trichloride, which is often subject to strict monitoring due to its dual-use potential. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures that production can continue reliably even if minor variations in utility supply occur. Additionally, the ability to recycle unreacted phosphorous acid, as demonstrated in the patent examples, further secures the supply of key starting materials and reduces dependency on fresh feedstock deliveries. This reliability is critical for maintaining continuous operations in large-scale facilities where downtime can result in significant financial losses.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful transition from gram-scale experiments to multi-hundred-gram batches without loss of efficiency or control. The use of solid acid catalysts and the absence of gaseous by-products simplify the scale-up process, as there is no need to engineer complex gas-liquid separation units for larger reactors. From an environmental standpoint, the reduction in hazardous waste generation and the potential for solvent recovery align perfectly with increasingly stringent global environmental regulations. This compliance advantage future-proofs the manufacturing asset, ensuring that the facility remains operational and profitable even as environmental standards become more rigorous over time.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines or for assessing the quality of the final product.
Q: What are the primary safety advantages of the new SPA synthesis route compared to the traditional PCl3 method?
A: The novel route eliminates the use of phosphorus trichloride and water, thereby avoiding the generation of hazardous hydrogen chloride gas and severe foaming issues associated with the conventional process.
Q: How does the use of immobilized acid catalysts impact the purification process?
A: Immobilized catalysts such as acidic clays or ion exchange resins can be easily filtered off from the reaction mixture, significantly simplifying downstream processing and reducing solvent consumption.
Q: What is the typical purity profile achievable with this crystallization method?
A: Through recrystallization from solvents like chloroform, the process can achieve high purity levels, effectively removing unreacted phosphorous acid and Diels-Alder dimer impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Styrene Phosphonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global fine chemicals market. Our team of expert chemists has extensively analyzed the pathways described in CN1254340A and possesses the technical capability to implement this superior route for the commercial production of Styrene Phosphonic Acid. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by the pharmaceutical and advanced materials industries.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume and quality requirements. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer and more efficient process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and drive long-term value for your organization.
