Advanced 2-Fluorophenyloxymethane Compounds for High-Performance Liquid Crystal Displays
Advanced 2-Fluorophenyloxymethane Compounds for High-Performance Liquid Crystal Displays
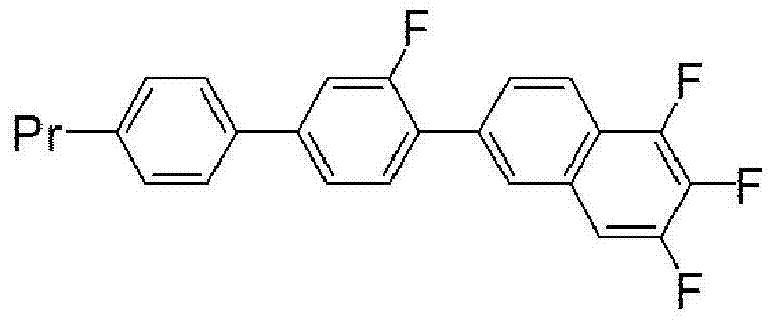
The rapid evolution of display technology demands materials that offer superior electro-optical performance, specifically regarding response speed and stability across wide temperature ranges. Patent CN103717563A introduces a novel class of compounds featuring a 2-fluorophenyloxymethane structure, designed to address the critical limitations of conventional liquid crystal materials. These compounds are engineered to exhibit low viscosity while maintaining a positive dielectric constant anisotropy, which is essential for modern IPS and VA mode displays. The innovation lies in the specific molecular architecture that prevents crystallization upon cooling, a common failure mode in high-performance mixtures. By integrating a methyleneoxy linkage (-CH2O-) within a fluorinated aromatic framework, the invention achieves a delicate balance between fluidity and intermolecular interaction. This technical breakthrough provides a robust foundation for next-generation liquid crystal compositions used in everything from automotive panels to high-definition televisions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of liquid crystal materials has been plagued by a trade-off between viscosity and solubility. Traditional approaches often relied on direct bonding of ring structures to minimize viscosity, but this frequently resulted in compounds with high crystallinity that would precipitate out of the mixture at lower operating temperatures. Alternatively, introducing linking groups to improve solubility often led to a significant increase in rotational viscosity, thereby slowing down the response time of the display element. Furthermore, achieving a high positive dielectric anisotropy typically required adding polar compounds in high concentrations, which exacerbated the risk of phase separation and crystallization. These inherent drawbacks limited the operational temperature range and reliability of the final display devices, necessitating complex mixture formulations that were costly and difficult to manufacture consistently.
The Novel Approach
The methodology disclosed in CN103717563A circumvents these issues by strategically placing fluorine atoms and utilizing a methyleneoxy bridge to disrupt molecular packing without sacrificing electronic properties. This novel approach allows for the creation of compounds that remain in a stable nematic phase over a broad temperature spectrum, effectively eliminating the risk of crystallization during device operation. The synthesis pathway is designed to be modular, enabling the production of a series of analogs with varying alkyl chain lengths to fine-tune physical properties without redesigning the entire synthetic route. This flexibility is crucial for customizing liquid crystal mixtures for specific client applications, ensuring that the material performs optimally regardless of the display mode or environmental conditions. The result is a versatile platform technology that enhances both the performance and manufacturability of advanced electronic chemicals.
Mechanistic Insights into Etherification and Suzuki Coupling
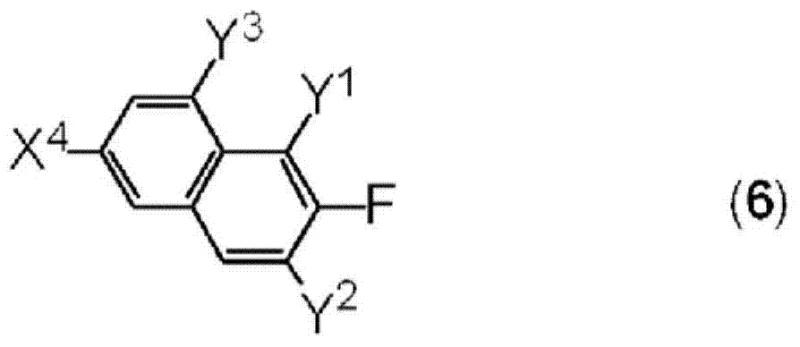
The core of this synthesis relies on a convergent strategy that builds the molecular complexity in distinct, high-yielding stages. The initial phase involves the formation of the ether linkage, which can be achieved either through a Mitsunobu reaction using azodicarboxylates and phosphines or via nucleophilic substitution with a base. This step is critical as it establishes the central scaffold of the molecule, connecting the fluorinated phenol moiety with the benzyl component. Subsequent transformation of the halogenated intermediate into a boronic acid derivative prepares the molecule for the final carbon-carbon bond-forming event. The precision required in these steps ensures that the regiochemistry of the fluorine substituents is preserved, which is vital for the resulting dielectric properties. Careful control of reaction temperatures, often ranging from cryogenic conditions for lithiation to reflux for coupling, minimizes side reactions and byproduct formation.
The final assembly of the target molecule is accomplished through a palladium-catalyzed cross-coupling reaction, specifically a Suzuki-Miyaura coupling, which joins the boronic acid intermediate with a fluorinated naphthalene derivative. This step is pivotal for extending the conjugated system and introducing the rigid core necessary for liquid crystallinity. The use of transition metal catalysts such as tetrakis(triphenylphosphine)palladium(0) facilitates this union under relatively mild conditions, preserving the sensitive functional groups present in the structure. Impurity control is managed through rigorous workup procedures, including aqueous washes to remove metal residues and chromatographic purification to isolate the desired isomer. This mechanistic pathway ensures high structural fidelity, which directly translates to consistent batch-to-batch performance in the final liquid crystal mixture.
How to Synthesize 2-Fluorophenyloxymethane Derivatives Efficiently
The synthesis of these high-value electronic chemicals requires a disciplined approach to reaction engineering and purification to meet the stringent standards of the display industry. The process begins with the preparation of key intermediates, such as fluorinated phenols and benzyl halides, which are then coupled to form the ether backbone. Following this, the introduction of the boron functionality enables the final coupling with the naphthalene core. Each step is optimized for yield and purity, utilizing standard laboratory and plant equipment to ensure scalability. The detailed standardized synthesis steps see the guide below for specific reaction conditions and stoichiometry.
- Perform etherification between a fluorinated phenol and a benzyl halide or alcohol using a base or dehydration condensing agent to form the ether intermediate.
- Convert the halogenated ether intermediate into a boronic acid or ester derivative via lithiation or Grignard reaction followed by quenching with a borate ester.
- Execute a Suzuki-Miyaura coupling reaction between the boronic acid derivative and a fluorinated naphthalene triflate using a palladium catalyst and base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthetic route offers significant strategic benefits beyond mere technical performance. The reliance on commercially available starting materials, such as substituted phenols and benzyl alcohols, mitigates the risk of raw material shortages and price volatility. The synthetic steps utilize well-established chemical transformations that do not require exotic reagents or specialized high-pressure equipment, facilitating easier technology transfer between manufacturing sites. This accessibility ensures a more resilient supply chain capable of responding quickly to fluctuations in market demand for display panels. Furthermore, the modularity of the synthesis allows for the production of multiple variants from a common set of intermediates, optimizing inventory management and reducing overall working capital requirements.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences in favor of a convergent synthesis strategy significantly streamlines the production process. By avoiding the need for expensive protecting group strategies or harsh reaction conditions that require specialized containment, the overall cost of goods sold is substantially reduced. The use of catalytic amounts of palladium, which can potentially be recovered and recycled, further contributes to long-term cost efficiency. Additionally, the high selectivity of the coupling reactions minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification processes and lowering solvent consumption.
- Enhanced Supply Chain Reliability: The robustness of the described chemical pathway ensures consistent output quality, which is critical for maintaining uninterrupted production lines for LCD manufacturers. Since the reagents and catalysts are standard commodities in the fine chemical industry, sourcing is not dependent on single-source suppliers or geopolitically sensitive regions. This diversification of the supply base enhances the security of supply, reducing the likelihood of delays due to logistical bottlenecks. The ability to scale the reaction from gram to kilogram quantities without significant re-optimization further guarantees that supply can grow in tandem with customer demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like THF and toluene that are manageable within standard industrial waste treatment frameworks. The avoidance of highly toxic reagents or extreme conditions simplifies compliance with environmental health and safety regulations. Waste streams are primarily organic and can be treated using conventional incineration or recovery methods, minimizing the environmental footprint of the manufacturing operation. This alignment with green chemistry principles not only reduces regulatory risk but also aligns with the sustainability goals of major electronics consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 2-fluorophenyloxymethane derivatives. The answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on performance metrics and handling requirements. Understanding these details is essential for integrating these materials into existing formulation workflows and ensuring optimal device performance.
Q: What is the primary advantage of the 2-fluorophenyloxymethane structure in liquid crystals?
A: The structure offers a unique balance of low viscosity and high dielectric constant anisotropy, preventing crystallization while maintaining fast response times in display elements.
Q: How is the purity of the final liquid crystal compound ensured during synthesis?
A: Purity is maintained through rigorous purification steps including silica gel column chromatography and recrystallization from solvents like ethanol or hexane after each critical reaction stage.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the process utilizes standard reagents and conditions such as THF solvents and palladium catalysts, which are well-established for scale-up in fine chemical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluorophenyloxymethane Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-fluorophenyloxymethane intermediate meets the exacting standards required for high-end electronic materials. Our commitment to quality assurance means that you can trust our materials to perform consistently in your final liquid crystal formulations.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your business goals. Let us be your trusted partner in delivering the high-performance materials that power the next generation of display technology.
