Advanced Biocatalytic Synthesis of Statin Intermediates Using Engineered KdAKR Mutants for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and high-purity synthetic routes for third-generation statins has led to significant advancements in biocatalytic engineering. Patent CN116103255A, published in May 2023, introduces a groundbreaking aldehyde-ketone reductase (KdAKR) mutant system specifically designed for the asymmetric synthesis of tert-butyl 6-chloro-(3R,5S)-dihydroxyhexanoate. This compound serves as a critical chiral precursor for rosuvastatin and pitavastatin, two of the most potent lipid-lowering agents globally. The patent details a sophisticated protein engineering strategy that transforms a naturally occurring enzyme from Kluyveromyces dobzhanskii into an industrial-grade biocatalyst capable of operating under rigorous commercial conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift from traditional chemical synthesis to high-efficiency biomanufacturing, offering substantial improvements in stereoselectivity, catalytic activity, and thermal stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-chloro-(3R,5S)-dihydroxyhexanoate tert-butyl ester has relied heavily on chemical catalysis, typically involving Claisen condensation followed by asymmetric reduction using sodium borohydride. While chemically feasible, this conventional pathway suffers from inherent inefficiencies that pose significant challenges for large-scale manufacturing. The chemical reduction step often requires harsh reaction conditions, including extreme temperatures and pressures, which drive up energy consumption and operational costs. Furthermore, chemical catalysts frequently struggle to achieve the stringent stereochemical control required for third-generation statins, often resulting in mixtures of diastereomers that necessitate complex and costly downstream purification processes. The use of stoichiometric reducing agents like sodium borohydride also generates substantial chemical waste, creating environmental compliance burdens and increasing the overall carbon footprint of the manufacturing process. These limitations collectively hinder the ability to achieve cost reduction in API manufacturing while maintaining the high purity standards demanded by regulatory agencies.
The Novel Approach
In stark contrast, the novel biocatalytic approach disclosed in CN116103255A leverages the power of directed evolution to overcome the natural limitations of wild-type enzymes. By introducing specific point mutations at six key amino acid positions (W297H, Y296W, Y28A, I63L, L58I, and G223P), the inventors have created the KdAKR-M6 mutant, which exhibits extraordinary catalytic performance. This engineered enzyme operates under mild physiological conditions (30-35°C, pH 7.0), significantly reducing energy requirements and equipment stress. The process utilizes a coupled enzyme system with glucose dehydrogenase (EsGDH) to regenerate the essential NADPH cofactor in situ, eliminating the need for expensive external cofactor addition. This self-sustaining cycle not only lowers material costs but also simplifies the reaction setup, making it highly amenable to commercial scale-up of complex pharmaceutical intermediates. The result is a streamlined, environmentally friendly process that delivers optical purity exceeding 99.5% deP, effectively eliminating the need for difficult chiral separations.
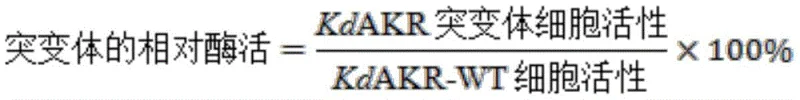
Mechanistic Insights into KdAKR-M6 Multi-Site Mutagenesis
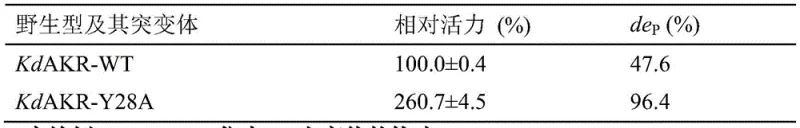
The dramatic improvement in catalytic efficiency observed in the KdAKR-M6 mutant is rooted in precise structural modifications that optimize the enzyme's active site and overall stability. Through homology modeling and molecular docking, the inventors identified that the wild-type enzyme's active pocket entrance was suboptimal for the bulky synthetic substrate. The introduction of the W297H and Y296W mutations alters the steric environment and electronic properties of the binding pocket, facilitating better substrate orientation and tighter binding affinity. Simultaneously, the Y28A and I63L mutations reduce steric hindrance, allowing for faster turnover rates. Perhaps most critically for industrial application, the L58I and G223P mutations introduce rigidifying effects that enhance the protein's structural integrity against thermal denaturation. This rational design strategy ensures that the enzyme maintains its conformational stability even at elevated substrate concentrations and prolonged reaction times, which is essential for consistent batch-to-batch quality in GMP manufacturing environments.
Furthermore, the mechanism of stereoselectivity enhancement is attributed to the precise positioning of the catalytic tetrad (Tyrosine, Histidine, Aspartic Acid, and Lysine) relative to the prochiral ketone group of the substrate. The mutated residues create a chiral environment that strictly favors the formation of the (3R,5S) configuration, suppressing the formation of the unwanted (3S,5S) epimer. This high fidelity is crucial because even minor impurities can affect the efficacy and safety profile of the final statin drug. The patent data indicates that the specific activity of the M6 mutant is 48.60 times higher than that of the wild-type enzyme, a magnitude of improvement that directly translates to reduced catalyst loading and shorter reaction cycles. This mechanistic robustness provides R&D teams with the confidence to scale the process from laboratory benchtop to multi-ton production without fearing loss of selectivity or activity.
How to Synthesize Tert-Butyl 6-Chloro-(3R,5S)-Dihydroxyhexanoate Efficiently
The implementation of this biocatalytic route involves a well-defined sequence of strain construction, fermentation, and bioconversion steps that are fully compatible with existing pharmaceutical infrastructure. The process begins with the transformation of E. coli BL21(DE3) host cells with recombinant plasmids encoding the optimized KdAKR-M6 and the cofactor-regenerating EsGDH. Following induction with IPTG, the wet biomass is harvested and utilized directly as a whole-cell catalyst, avoiding the complexity and cost of enzyme purification. The bioconversion is conducted in a phosphate buffer system where the substrate is fed at high concentrations, leveraging the mutant's enhanced tolerance to substrate inhibition. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results.
- Construct recombinant E. coli BL21(DE3) strains harboring the KdAKR-M6 mutant gene and glucose dehydrogenase (EsGDH) gene via plasmid transformation and induction.
- Cultivate the engineered bacteria in LB medium with kanamycin, induce expression with IPTG at 28°C, and harvest wet cells via centrifugation.
- Perform bioconversion by mixing wet cells with substrate (200g/L) and glucose (300g/L) in phosphate buffer at 35°C for 8 hours to achieve complete conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the KdAKR-M6 technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from chemical to enzymatic synthesis fundamentally alters the cost structure of producing this key statin intermediate. By eliminating the need for hazardous chemical reducing agents and the associated waste treatment protocols, manufacturers can achieve significant cost savings in raw materials and environmental compliance. The mild reaction conditions also reduce the demand for specialized high-pressure or cryogenic equipment, lowering capital expenditure (CAPEX) for new production lines. Moreover, the high space-time yield of 224.6 g/L/d means that existing reactor volumes can produce significantly more product in less time, effectively increasing asset utilization and reducing the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The enzymatic process drastically simplifies the production workflow by removing multiple chemical protection and deprotection steps often required in traditional synthesis. The use of whole-cell biocatalysts eliminates the expensive enzyme purification stage, while the in-situ cofactor regeneration system removes the need for purchasing stoichiometric amounts of NADPH. These factors combine to substantially lower the variable cost per kilogram of the final product, providing a competitive edge in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: The robustness of the KdAKR-M6 mutant ensures consistent performance across large-scale batches, minimizing the risk of production failures or off-spec material that could disrupt supply continuity. The high substrate loading capacity of 200 g/L allows for concentrated reaction mixtures, which reduces the volume of solvent and water required for processing. This efficiency translates to smaller logistics footprints for raw material delivery and waste removal, streamlining the overall supply chain operation and enhancing resilience against market fluctuations.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal hazardous waste compared to borohydride-based chemical reduction. This aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. The scalability is proven by the rapid reaction kinetics, where complete conversion is achieved within 8 hours, allowing for high-throughput manufacturing schedules. This operational efficiency ensures that suppliers can meet surging demand for statin precursors without compromising on quality or delivery timelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented enzyme technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these specifics is vital for making informed decisions about process adoption and supplier qualification.
Q: What are the key advantages of the KdAKR-M6 mutant over the wild-type enzyme?
A: The KdAKR-M6 mutant exhibits a 48.60-fold increase in catalytic activity and a 4°C improvement in thermal stability (T50) compared to the wild-type, while maintaining strict stereoselectivity (deP > 99.5%).
Q: How does this biocatalytic route compare to traditional chemical synthesis for statin intermediates?
A: Unlike traditional chemical methods requiring harsh conditions and sodium borohydride reduction, this enzymatic process operates under mild conditions (35°C, pH 7.0) with higher stereoselectivity and eliminates heavy metal waste.
Q: What is the maximum substrate loading capacity demonstrated in this patent?
A: The process supports a high substrate loading of up to 200g/L of tert-butyl 6-chloro-(5S)-hydroxy-3-carbonylhexanoate, achieving complete conversion within 8 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl 6-Chloro-(3R,5S)-Dihydroxyhexanoate Supplier
As the global demand for high-efficacy statins continues to rise, securing a stable supply of high-quality chiral intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced biocatalytic technologies like the KdAKR-M6 system to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and reliability. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of tert-butyl 6-chloro-(3R,5S)-dihydroxyhexanoate meets the highest international standards for chirality and chemical purity.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative enzymatic solutions. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced biocatalytic capabilities can become a strategic asset for your organization's growth and competitiveness in the global pharmaceutical market.
