Advanced Terpyridine Supramolecular Photocatalysts: Scaling Efficient CO2 Reduction Technologies
The global energy landscape is undergoing a critical transformation, driven by the urgent necessity to mitigate carbon emissions and secure sustainable fuel sources. In this context, semiconductor photocatalysis represents a frontier technology capable of converting solar energy directly into chemical fuels, specifically through the reduction of carbon dioxide (CO2) into value-added products like carbon monoxide (CO) and methane (CH4). A significant breakthrough in this domain is detailed in patent CN115414965A, which discloses a novel preparation method for a bromine-containing terpyridine conjugated supramolecular photocatalyst. This innovation addresses the longstanding inefficiencies of multi-component catalytic systems by introducing a single-component architecture that functions simultaneously as a photosensitizer and a catalyst. By leveraging a straightforward dissolution-precipitation protocol, the technology achieves high crystallinity and a robust built-in electric field, essential parameters for driving efficient charge separation. For industrial stakeholders, this represents a pivotal shift towards more streamlined, cost-effective, and scalable photochemical manufacturing processes that do not rely on scarce precious metal resources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient photocatalytic systems for CO2 reduction has been hindered by the complexity of constructing effective light-harvesting and catalytic units. Traditional approaches typically involve the physical or chemical integration of distinct components, such as Ruthenium-based polypyridyl complexes acting as photosensitizers and Cobalt oxime complexes serving as the actual catalysts. This dual-component strategy introduces significant engineering challenges, particularly regarding the interfacial contact between the sensitizer and the catalyst. Efficient electron transfer requires precise调控 (regulation) of the interface to minimize energy loss during the migration of photogenerated electrons from the excited state of the sensitizer to the active site of the catalyst. Furthermore, the reliance on precious metals like Ruthenium imposes severe economic constraints and supply chain vulnerabilities, as these materials are subject to geopolitical volatility and high market prices. The synthetic procedures for these multi-component assemblies are often laborious, requiring multiple purification steps and stringent control over stoichiometry, which complicates the transition from laboratory discovery to commercial production.
The Novel Approach
The methodology outlined in patent CN115414965A offers a transformative alternative by utilizing a single-component terpyridine-based supramolecular assembly. Instead of mixing disparate chemical entities, this approach relies on the self-organization of 4'-(4-bromophenyl)-2,2':6',2''-terpyridine molecules into a highly ordered crystalline structure. Through a simple dissolution-precipitation technique, these molecules stack via strong π-π interactions, creating a continuous pathway for electron transport that mimics the properties of inorganic semiconductors. This structural integrity eliminates the need for complex interface engineering, as the light absorption and catalytic reduction occur within the same continuous lattice. The inclusion of the bromine substituent is a critical design feature that enhances the internal electric field, thereby facilitating the separation of electron-hole pairs more effectively than conventional organic photocatalysts. This simplification of the material architecture not only reduces the synthetic burden but also inherently improves the stability and reproducibility of the photocatalytic performance, making it a superior candidate for industrial adoption.
Mechanistic Insights into Krohnke Synthesis and Supramolecular Assembly
The foundation of this advanced photocatalytic material lies in the precise synthesis of the core building block, 4'-(4-bromophenyl)-2,2':6',2''-terpyridine, achieved through the classic Krohnke pyridine synthesis method. This reaction involves the condensation of p-bromobenzaldehyde with two equivalents of 2-acetylpyridine in the presence of ammonium acetate or aqueous ammonia and a base such as potassium hydroxide. The reaction mechanism proceeds through the formation of an α,β-unsaturated ketone intermediate, which subsequently undergoes cyclization with ammonia to form the central pyridine ring, linking the three aromatic units into a rigid, planar terpyridine scaffold. The choice of the para-bromo substituent on the central phenyl ring is not arbitrary; it serves a dual purpose of modifying the electronic properties of the molecule and providing a handle for potential further functionalization. The resulting conjugated system possesses an extended π-electron cloud, which is essential for absorbing visible light and generating the excitons required for photocatalysis. The purity and structural integrity of this intermediate are paramount, as any defects in the molecular structure can disrupt the subsequent self-assembly process and degrade the semiconductor properties of the final material.
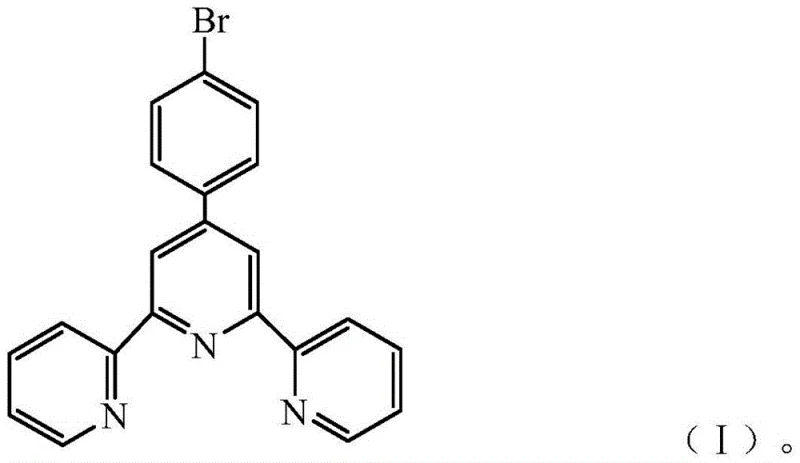
Following the synthesis of the molecular precursor, the critical step involves the supramolecular self-assembly via a solvent exchange mechanism. The pure terpyridine solid is first dissolved in a benign organic solvent such as dimethylformamide (DMF), ethylene glycol methyl ether, or tetrahydrofuran (THF), where the molecules exist as free-moving species. Upon the dropwise addition of water, which acts as a poor solvent, the solubility of the terpyridine molecules decreases drastically, forcing them out of the solution phase. However, rather than precipitating as an amorphous powder, the molecules organize themselves into highly crystalline rod-like structures driven by intermolecular π-π stacking interactions and halogen bonding involving the bromine atoms. This ordered aggregation extends the conjugation of the π-electrons across adjacent molecules, effectively creating a 'molecular wire' that allows for cross-ring electron transport. The resulting built-in electric field, amplified by the dipole moment of the bromine substituents, drives the photogenerated electrons and holes in opposite directions, preventing recombination and ensuring that the electrons are available at the surface to reduce CO2. This mechanism highlights how subtle changes in processing conditions, such as solvent ratios and stirring times, can dictate the macroscopic morphology and microscopic electronic properties of the catalyst.
How to Synthesize 4'-(4-bromophenyl)-2,2':6',2''-terpyridine Efficiently
The practical implementation of this technology requires adherence to specific operational parameters to ensure the formation of the desired rod-like supramolecular morphology. The process begins with the rigorous purification of the Krohnke reaction product to remove any unreacted aldehydes or ketones that could act as impurities during the assembly phase. Once the high-purity terpyridine solid is obtained, it is dispersed in the chosen organic solvent at a specific mass-to-volume ratio, typically around 500mg per 7-14mL of solvent, to ensure complete dissolution without excessive dilution. The subsequent addition of water must be controlled carefully; adding too little water may fail to induce precipitation, while adding it too rapidly can lead to amorphous aggregation rather than crystalline growth. The mixture is then subjected to prolonged stirring, typically ranging from 3 to 10 hours, to allow the nuclei to grow into the characteristic micrometer-scale rods. Detailed standardized synthesis steps, including specific temperatures, reaction times, and workup procedures, are provided in the technical guide below to assist R&D teams in replicating these results accurately.
- Synthesize the core 4'-(4-bromophenyl)-2,2': 6',2''-terpyridine molecule using the Krohnke method by reacting p-bromobenzaldehyde and 2-acetylpyridine with ammonia and KOH in ethanol.
- Dissolve the purified terpyridine solid in a benign organic solvent such as DMF, ethylene glycol methyl ether, or THF to form a transparent solution at room temperature.
- Induce supramolecular self-assembly by dropwise addition of water (poor solvent) to the organic solution, stirring for 3-10 hours to precipitate the white photocatalyst solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition to this single-component terpyridine photocatalyst offers substantial strategic advantages over traditional metal-complex systems. The most immediate benefit is the drastic simplification of the raw material portfolio. By eliminating the need for expensive and supply-constrained precious metals like Ruthenium or Iridium, manufacturers can decouple their production costs from the volatile commodities market. The primary feedstocks—p-bromobenzaldehyde and 2-acetylpyridine—are commodity chemicals produced at massive scales for the pharmaceutical and agrochemical industries, ensuring a stable and reliable supply chain with predictable pricing. Furthermore, the synthetic route avoids the use of toxic heavy metal catalysts, which simplifies waste management protocols and reduces the regulatory burden associated with hazardous material handling and disposal. This alignment with green chemistry principles not only lowers operational expenditures but also enhances the environmental profile of the final product, a key metric for modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of costly noble metal precursors and the simplification of the synthesis workflow. Traditional photocatalytic systems often require multi-step ligand synthesis and metal coordination under inert atmospheres, which demand specialized equipment and high energy inputs. In contrast, the terpyridine supramolecular assembly is achieved through a room-temperature dissolution-precipitation process that utilizes common solvents like ethanol and water. This reduction in process complexity translates directly to lower capital expenditure (CAPEX) and operational expenditure (OPEX), as the reaction does not require high-pressure reactors or cryogenic cooling systems. Additionally, the high crystallinity of the product minimizes the need for extensive downstream purification, further streamlining the manufacturing timeline and reducing solvent consumption.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-performance materials is critical for maintaining production continuity in the fine chemical sector. The reliance on earth-abundant elements (Carbon, Hydrogen, Nitrogen, Bromine) ensures that the production of this photocatalyst is not susceptible to the geopolitical risks that often plague the supply chains of Platinum Group Metals (PGMs). The raw materials are widely available from multiple global suppliers, reducing the risk of single-source dependency. Moreover, the robustness of the supramolecular structure implies a longer shelf-life and greater stability during storage and transportation compared to sensitive metal-organic complexes that may degrade upon exposure to moisture or light. This inherent stability reduces inventory losses and ensures that the material retains its catalytic activity from the point of manufacture to the point of use.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to the ton scale often introduces unforeseen engineering challenges, but the physics of this self-assembly process suggests excellent scalability. The dissolution-precipitation mechanism is fundamentally a mixing and crystallization operation, unit processes that are well-understood and easily scaled in standard chemical reactors. The use of water as the anti-solvent is particularly advantageous from an environmental standpoint, as it reduces the volume of organic waste generated during the isolation step. The resulting solid product can be recovered via simple filtration and drying, avoiding the need for energy-intensive distillation or chromatography. This ease of scale-up, combined with a reduced environmental footprint, positions this technology as a compliant and future-proof solution for meeting the growing global demand for renewable fuel technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this terpyridine-based photocatalytic technology. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, offering clarity on performance metrics, structural properties, and application potential. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this material into existing CO2 conversion workflows or developing new photochemical reactor designs.
Q: What distinguishes this terpyridine photocatalyst from traditional metal-complex systems?
A: Unlike traditional systems that require separate photosensitizers (like Ruthenium complexes) and catalysts (like Cobalt oximes), this patent describes a single-component supramolecular assembly. The terpyridine assembly acts as both the light harvester and the catalytic active site, eliminating the need for complex interface engineering and expensive precious metals.
Q: How does the bromine substituent enhance photocatalytic performance?
A: The bromine substituent (-Br) influences the molecular dipolarity, which helps regulate the built-in electric field within the supramolecular crystal lattice. This strong internal field promotes the efficient separation of photogenerated electron-hole pairs, significantly boosting the reduction potential and overall catalytic activity compared to non-halogenated analogs.
Q: Is the dissolution-precipitation method suitable for industrial scale-up?
A: Yes, the process relies on simple solvent exchange (adding water to an organic solution) and standard filtration. It avoids high-pressure reactors or complex vacuum deposition techniques, making it highly amenable to batch processing and large-scale manufacturing with minimal equipment modification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-(4-bromophenyl)-2,2':6',2''-terpyridine Supplier
The commercialization of next-generation photochemical materials requires a partner with deep expertise in process development and quality assurance. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the Krohnke synthesis and subsequent self-assembly steps to ensure batch-to-batch consistency, adhering to stringent purity specifications required for high-performance electronic and energy materials. We understand that the efficiency of a photocatalyst is heavily dependent on its crystalline morphology and defect density; therefore, our rigorous QC labs employ advanced characterization techniques to verify the structural integrity of every lot before shipment. Whether you are developing pilot-scale CO2 reduction reactors or researching new optoelectronic applications, our infrastructure is designed to support your transition from R&D to full-scale manufacturing.
We invite you to collaborate with us to unlock the potential of this sustainable technology. By leveraging our supply chain capabilities, you can secure a stable source of high-quality terpyridine intermediates while minimizing your procurement risks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your project's economic viability. Let us help you engineer a more sustainable and profitable future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
