Advanced Solvent-Free Synthesis of Bipyridyl Dimalononitrile Derivatives for High-Value Applications
Introduction to Patent CN102993085A: A Breakthrough in Green Synthesis
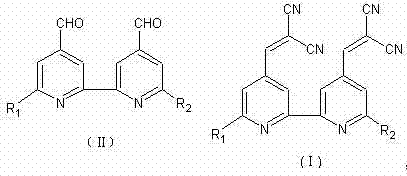
The chemical industry is constantly seeking methods to reduce environmental impact while maintaining high efficiency, and patent CN102993085A presents a significant advancement in this domain. This patent details a novel synthesis method for 2,2'-(2,2'-bipyridyl-4,4'-dimethyl)dimalononitrile and its substitutes, which are critical intermediates in the production of advanced functional materials. Traditionally, the synthesis of such conjugated systems relies heavily on organic solvents and amine catalysts, creating substantial downstream processing burdens. However, the technology disclosed in this patent utilizes a solvent-free and catalyst-free approach, leveraging the unique physical properties of malononitrile to drive the reaction forward. By operating at temperatures between 45°C and 160°C, the process achieves exceptional conversion rates without the need for external catalytic agents. This represents a paradigm shift for manufacturers of reliable pharmaceutical intermediate supplier networks who are under increasing pressure to adopt greener chemistry principles. The ability to produce high-value bipyridine derivatives with minimal waste generation positions this technology as a cornerstone for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formation of carbon-carbon double bonds in complex heterocyclic systems like bipyridines has been dominated by the classical Knoevenagel condensation. Standard protocols typically require the use of organic solvents such as ethanol or toluene, coupled with basic catalysts like piperidine or ammonium acetate. While effective on a small laboratory scale, these conventional methods introduce severe inefficiencies when translated to commercial production. The presence of catalysts necessitates rigorous purification steps to remove trace amines, which can be detrimental to the stability of the final electronic or pharmaceutical product. Furthermore, the reliance on volatile organic solvents creates significant safety hazards regarding flammability and toxicity, alongside the heavy financial burden of solvent recovery and waste disposal. In many cases, the solubility issues of the starting bipyridyl dialdehydes in common solvents lead to heterogeneous reaction conditions, resulting in inconsistent yields and prolonged reaction times that hamper throughput.
The Novel Approach
The methodology outlined in patent CN102993085A elegantly circumvents these traditional bottlenecks by employing a neat reaction strategy where malononitrile serves a dual purpose. Not only does it act as the active methylene component in the condensation, but in its molten state, it also functions as the reaction medium. This eliminates the need for any additional solvent, thereby drastically reducing the volume of waste generated per kilogram of product. The absence of a catalyst removes the risk of catalyst-derived impurities, simplifying the quality control profile for the final API intermediate. By adjusting the molar ratio of bipyridyl dialdehyde to malononitrile between 1:2.5 and 1:8, the system ensures that the solid aldehyde starting material is fully dissolved and reacted. This innovative approach not only streamlines the operational workflow but also aligns perfectly with the goals of cost reduction in electronic chemical manufacturing, where purity and process simplicity are paramount drivers of value.
Mechanistic Insights into Solvent-Free Knoevenagel Condensation
The core of this synthetic breakthrough lies in the unique reactivity of malononitrile under thermal conditions without external activation. In a typical Knoevenagel mechanism, a base catalyst deprotonates the active methylene group to form a nucleophilic carbanion, which then attacks the carbonyl carbon of the aldehyde. In this solvent-free system, the thermal energy provided at 45-160°C is sufficient to overcome the activation barrier, potentially facilitated by the inherent acidity of the methylene protons in malononitrile and the electrophilicity of the pyridine-substituted aldehydes. The electron-withdrawing nature of the pyridine rings enhances the reactivity of the aldehyde groups, making them more susceptible to nucleophilic attack even in the absence of a strong base. As the reaction proceeds, the formation of the conjugated double bond system extends the pi-electron network, which is crucial for the photoelectric properties mentioned in the patent background. This mechanistic pathway ensures that the reaction proceeds cleanly to the desired dimalononitrile product without significant side reactions such as polymerization or Michael additions, which are common pitfalls in solution-phase chemistry.
Controlling impurities in this system is inherently easier due to the lack of extraneous reagents. In catalyzed reactions, side products often arise from the interaction of the catalyst with the substrate or solvent, leading to complex impurity profiles that are difficult to separate. Here, the primary byproduct is water, which is easily removed during the workup or drives the equilibrium forward if managed correctly. The patent specifies that the reaction mixture becomes turbid as the product forms, indicating precipitation from the molten malononitrile matrix. This phase separation acts as a driving force for the reaction, pulling the equilibrium towards the product side according to Le Chatelier's principle. Consequently, the crude product obtained after cooling and water addition is already of high purity, requiring only a simple recrystallization step to meet stringent specifications. This intrinsic purity control mechanism is a major advantage for producing high-purity OLED material precursors where trace metallic or amine contaminants can quench luminescence.
How to Synthesize 2,2'-(2,2'-Bipyridyl-4,4'-dimethyl)dimalononitrile Efficiently
Implementing this synthesis route requires precise control over temperature and stoichiometry to maximize yield and minimize raw material waste. The process begins with the careful weighing of the bipyridyl dialdehyde derivative and an excess of malononitrile, ensuring the molar ratio falls within the optimal range of 1:2.5 to 1:8. The mixture is then heated under stirring, where the malononitrile melts and dissolves the aldehyde, creating a homogeneous liquid phase that facilitates efficient molecular collision. Maintaining the temperature between 45°C and 160°C is critical; too low a temperature may result in incomplete dissolution and slow kinetics, while excessive heat could lead to the volatilization of malononitrile or decomposition of sensitive functional groups. Once the reaction is complete, indicated by TLC analysis or the cessation of water evolution, the mixture is quenched into water.
- Mix bipyridyl dialdehyde compounds with excess malononitrile (molar ratio 1: 2.5 to 1:8) in a reaction vessel.
- Heat the mixture to a temperature range of 45-160°C and stir for 1-6 hours to facilitate the condensation reaction without external solvents.
- Quench the reaction by adding water, filter the precipitate, wash with aqueous ethanol, and recrystallize from DMF or DMSO to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free technology offers transformative benefits that extend far beyond simple yield improvements. The elimination of organic solvents and catalysts fundamentally alters the cost structure of the manufacturing process, removing entire unit operations such as solvent distillation, catalyst filtration, and hazardous waste treatment. This simplification translates directly into lower operational expenditures and a reduced carbon footprint, which is increasingly important for multinational corporations aiming to meet sustainability targets. Furthermore, the robustness of the reaction conditions allows for greater flexibility in sourcing raw materials, as the process is less sensitive to the moisture content or grade of solvents that would otherwise be required. This resilience enhances supply chain reliability, ensuring consistent production schedules even when facing fluctuations in the availability of high-grade chemical reagents.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in raw material and waste management costs. By removing the need for expensive organic solvents and amine catalysts, the bill of materials is significantly streamlined. Additionally, the energy consumption associated with heating and cooling large volumes of solvent is eliminated, leading to substantial utility savings. The simplified workup procedure, which involves merely adding water and filtering, reduces labor hours and equipment usage time, allowing for higher throughput in existing facilities without the need for capital-intensive expansion. These cumulative efficiencies result in a much more competitive cost position for the final bipyridine derivatives.
- Enhanced Supply Chain Reliability: Relying on a process that uses fewer distinct chemical inputs reduces the complexity of the supply chain. There is no longer a dependency on specific grades of solvents or specialized catalysts that might have long lead times or single-source suppliers. Malononitrile and bipyridyl aldehydes are commodity chemicals with established supply lines, ensuring continuity of supply. The simplified logistics of not having to transport and store large quantities of flammable solvents also reduces regulatory burdens and insurance costs, making the overall supply chain more agile and responsive to market demands for rapid delivery of critical intermediates.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges related to heat transfer and mixing, but this neat reaction method mitigates many of those risks. The absence of solvent volume means reactors can be charged with a higher density of reactants, effectively increasing the capacity of existing vessels. From an environmental perspective, the process generates minimal hazardous waste, primarily consisting of aqueous washings that are easier to treat than solvent-laden effluents. This ease of compliance with environmental regulations facilitates faster permitting for new production lines and reduces the risk of shutdowns due to non-compliance, securing long-term production stability for high-volume contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making.
Q: What are the advantages of the solvent-free method for bipyridyl dimalononitrile synthesis?
A: The solvent-free method eliminates the need for volatile organic compounds and expensive catalysts like piperidine. This significantly reduces waste treatment costs, simplifies the workup procedure to a simple water precipitation, and achieves high yields (over 95%) without complex purification steps.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process is highly scalable. Since malononitrile acts as both a reactant and a solvent at elevated temperatures, the reaction mixture remains manageable. The simple post-treatment involving water addition and filtration makes it ideal for large-scale manufacturing without requiring specialized solvent recovery infrastructure.
Q: What is the purity profile of the resulting dimalononitrile derivatives?
A: The method produces high-purity products, often exceeding 95% yield directly after recrystallization. The absence of catalyst residues means fewer impurities related to amine salts or metal complexes, which is critical for applications in sensitive electronic materials or pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-(2,2'-Bipyridyl-4,4'-dimethyl)dimalononitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting green chemistry technologies like the one described in patent CN102993085A. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that consistency is key in the supply of complex intermediates, which is why our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for every batch. Whether your application requires these bipyridine derivatives for pharmaceutical synthesis or advanced electronic materials, our commitment to quality and process optimization ensures that you receive a product that meets the highest international standards.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this solvent-free method for your supply chain. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you optimize your sourcing strategy and secure a reliable supply of high-performance chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
