Revolutionizing Herbicide Intermediate Production: High-Yield Synthesis of 2-Chloro-Sulfonyl-3-Methyl Benzoate
Revolutionizing Herbicide Intermediate Production: High-Yield Synthesis of 2-Chloro-Sulfonyl-3-Methyl Benzoate
The global demand for high-efficiency sulfonylurea herbicides, such as Triflusulfuron-methyl, necessitates a robust and economically viable supply chain for their critical precursors. Patent CN101786972A introduces a transformative methodology for the preparation of 2-chloro-sulfonyl-3-methyl benzoate, a pivotal intermediate in this agricultural sector. This innovation addresses long-standing inefficiencies in traditional manufacturing by consolidating a previously cumbersome three-step sequence into a streamlined, one-pot diazotization and sulfonyl chlorination protocol. By leveraging acetic acid as a dual-purpose solvent and reactant medium, the process achieves yields exceeding 70%, a substantial improvement over historical benchmarks. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize purity profiles while drastically reducing the cost of goods sold (COGS) through simplified unit operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, specifically those disclosed in documents like US5157119, rely on a convoluted three-step synthetic route that imposes severe penalties on both operational efficiency and overall yield. The conventional process initiates with the reaction of 2-hydroxy-3-methyl methyl benzoate with dimethylthiocarbamoyl chloride, a reagent that is not only prohibitively expensive but also presents significant handling hazards and supply chain volatility. Following this initial substitution, the intermediate must undergo a thermal rearrangement in orthodichlorobenzene under reflux conditions for approximately 18 hours, a step that consumes vast amounts of energy and introduces high-boiling solvent removal challenges. The final conversion to the target sulfonyl chloride requires chlorination with chlorine gas, adding another layer of safety complexity and corrosion risk to the plant infrastructure. Cumulatively, these discrete steps result in a total recovery rate of no more than 52%, meaning nearly half of the valuable starting material is lost to waste streams, purification losses, and side reactions.
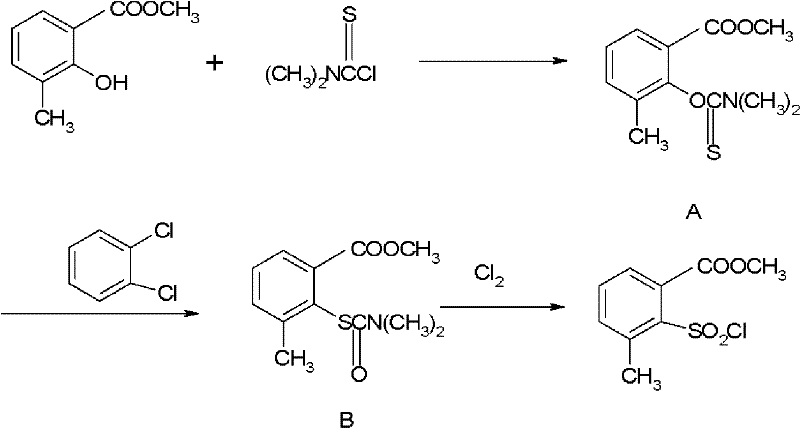
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct diazotization strategy starting from the readily available 2-amino-3-methyl benzoate. This method elegantly bypasses the need for the costly thiocarbamoyl protection and subsequent high-temperature rearrangement entirely. The process operates under mild conditions, maintaining temperatures between 10°C and 25°C during the critical sulfonyl chlorination phase, which significantly lowers energy consumption compared to the refluxing orthodichlorobenzene required in the old method. By employing sulfur dioxide gas in the presence of a copper chloride catalyst within an acetic acid medium, the transformation proceeds with high selectivity and minimal byproduct formation. This consolidation of reaction steps not only simplifies the equipment footprint required for production but also enhances the safety profile by eliminating the handling of hazardous high-temperature rearrangement intermediates. The result is a robust, scalable process that delivers yields consistently above 70%, effectively doubling the material throughput compared to legacy technologies.
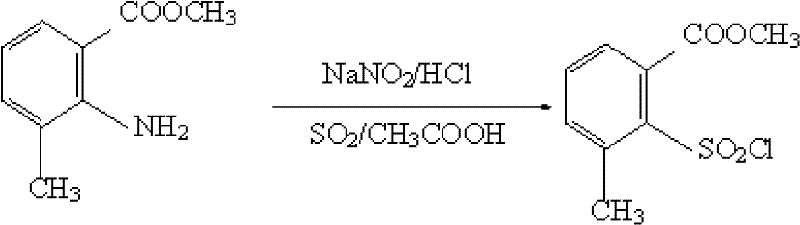
Mechanistic Insights into CuCl2-Catalyzed Diazotization and Sulfonyl Chlorination
The core of this technological breakthrough lies in the precise control of the diazonium intermediate and its subsequent capture by sulfur dioxide in a catalytic cycle mediated by copper species. Initially, the 2-amino-3-methyl benzoate undergoes diazotization with sodium nitrite and concentrated hydrochloric acid in an acetic acid solvent at low temperatures (-10°C to 5°C), forming a stable diazonium salt. This low-temperature control is critical to prevent the premature decomposition of the diazonium species into phenols or other hydrolysis byproducts, ensuring high fidelity in the subsequent substitution. The use of acetic acid is particularly advantageous as it provides a polar environment that stabilizes the ionic diazonium species while remaining compatible with the subsequent introduction of gaseous sulfur dioxide. Unlike aqueous systems where hydrolysis competes aggressively, the acetic acid medium suppresses water activity, thereby directing the reaction pathway exclusively towards the desired sulfonyl chloride formation.
Following the formation of the diazonium salt, the introduction of sulfur dioxide gas in the presence of cupric chloride initiates a radical-based substitution mechanism akin to a Meerwein arylation or a modified Sandmeyer-type reaction. The copper catalyst facilitates the single-electron transfer processes necessary to convert the diazonium group into a sulfonyl chloride moiety. The reaction temperature is carefully maintained between 10°C and 25°C to balance the kinetics of gas absorption with the stability of the reactive intermediates. This mechanistic pathway avoids the formation of the sulfonamide or sulfonic acid byproducts often seen in less controlled environments. Furthermore, the workup procedure involving dichloromethane extraction allows for the efficient separation of the organic product from the inorganic copper salts and acidic residues, resulting in a crude product of high purity that requires minimal downstream processing to meet stringent pharmaceutical or agrochemical specifications.
How to Synthesize 2-Chloro-Sulfonyl-3-Methyl Benzoate Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the diazotizing agents and the rate of sulfur dioxide addition to maximize yield and safety. The patent outlines a specific protocol where the weight ratio of the amine to sodium nitrite is optimized between 1.1:1 and 3.2:1, ensuring complete conversion of the amine without excessive nitrite waste. The detailed standardized synthetic steps below provide a roadmap for laboratory validation and pilot-scale translation, emphasizing the critical control points for temperature and reagent addition rates. Operators should note that the exothermic nature of the diazotization requires efficient cooling capabilities, while the sulfonyl chlorination step demands effective gas scrubbing systems to handle excess sulfur dioxide. Adhering to these parameters ensures the reproducibility of the high yields reported in the patent examples.
- Prepare the diazotization solution by reacting 2-amino-3-methyl benzoate with sodium nitrite and concentrated hydrochloric acid in acetic acid at -10 to 5 degrees Celsius.
- Introduce sulfur dioxide gas into a separate acetic acid solution containing a copper chloride catalyst at 10 to 25 degrees Celsius.
- Add the diazotization solution to the sulfur dioxide mixture, allow reaction to complete, and isolate the product via dichloromethane extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend far beyond simple yield improvements. The elimination of dimethylthiocarbamoyl chloride, a specialized and costly reagent, removes a significant bottleneck in the raw material supply chain, replacing it with commodity chemicals like sodium nitrite and sulfur dioxide that are globally available and price-stable. This shift fundamentally alters the cost structure of the intermediate, allowing for significant margin expansion or competitive pricing strategies in the final herbicide market. Moreover, the reduction from three distinct reaction vessels and isolation steps to a streamlined one-pot process drastically reduces the manufacturing cycle time, enhancing the agility of the supply chain to respond to seasonal demand fluctuations in the agrochemical sector.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the unit operations and the substitution of expensive reagents with commodity chemicals. By removing the need for the high-boiling solvent orthodichlorobenzene and the costly thiocarbamoyl chloride, the variable cost per kilogram of product is substantially lowered. Additionally, the increase in yield from roughly 52% to over 70% means that less raw material is required to produce the same amount of finished goods, effectively reducing the material cost burden by nearly one-third without any negotiation with suppliers. The energy savings from avoiding an 18-hour reflux step further contribute to a leaner manufacturing cost profile, making the final product highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Relying on a three-step process with specialized intermediates creates multiple points of failure in the supply chain, where a delay in one step halts the entire production line. The new one-pot method mitigates this risk by consolidating the synthesis, thereby reducing the lead time for high-purity agrochemical intermediates significantly. The raw materials, specifically 2-amino-3-methyl benzoate, sodium nitrite, and sulfur dioxide, are standard industrial chemicals with robust global supply networks, ensuring continuity of supply even during market disruptions. This reliability allows downstream herbicide manufacturers to maintain lower safety stock levels, freeing up working capital and improving overall inventory turnover rates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the new process offers a cleaner profile that simplifies waste treatment and compliance reporting. The avoidance of chlorination with elemental chlorine gas in the final step reduces the generation of hazardous chlorinated organic byproducts and minimizes the risk of toxic gas releases. Furthermore, the use of acetic acid, which is easier to recover and recycle compared to orthodichlorobenzene, aligns with modern green chemistry principles and reduces the volume of hazardous waste requiring disposal. This environmental efficiency not only lowers disposal costs but also future-proofs the manufacturing site against increasingly stringent environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diazotization-based synthesis for 2-chloro-sulfonyl-3-methyl benzoate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on process robustness and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this intermediate.
Q: What are the primary advantages of the new diazotization method over the conventional three-step synthesis?
A: The new method reduces the process from three complex steps to a single one-pot reaction, increasing total yield from less than 52% to over 70% while eliminating expensive reagents like dimethylthiocarbamoyl chloride.
Q: Why is acetic acid used as the solvent in this sulfonyl chlorination process?
A: Acetic acid serves as an ideal polar protic solvent that stabilizes the reactive diazonium intermediate and facilitates the solubility of both the organic amine substrate and the inorganic reagents involved in the catalytic cycle.
Q: Is this process suitable for large-scale industrial production of herbicide intermediates?
A: Yes, the process utilizes low-cost, readily available raw materials and operates at mild temperatures (10-25°C), making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-Sulfonyl-3-Methyl Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation herbicides depends on the availability of high-quality, cost-effective intermediates produced via sustainable routes. Our technical team has extensively analyzed the diazotization pathway described in CN101786972A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to full industrial maturity. We are committed to delivering this critical intermediate with stringent purity specifications and rigorous QC labs testing to ensure it meets the exacting standards of global agrochemical formulators. Our facility is equipped to handle the specific gas-liquid reaction dynamics of sulfur dioxide chlorination safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for Triflusulfuron-methyl production. By leveraging our expertise in process intensification, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your transition to this superior synthetic method is seamless and commercially rewarding.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
