Advanced Synthesis of Asymmetric Photoelectric Active Thiacalix[4]arene Receptors for High-End Sensing Applications
The landscape of supramolecular chemistry is undergoing a significant transformation driven by the demand for multi-functional receptor molecules capable of complex signal transduction. Patent CN107474078B introduces a groundbreaking methodology for the synthesis of an asymmetric photoelectric active thiacalix[4]arene receptor molecule, designated as Fc-TA-RB. This innovation addresses a critical limitation in current sensor technology by moving away from traditional symmetric designs towards a sophisticated asymmetric architecture. By sequentially introducing an electrochemical probe (ferrocene) and a fluorescent probe (rhodamine B) onto a thiacalix[4]arene platform, this technology enables dual-channel response detection. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal advancement in creating high-value intermediates for next-generation sensing devices, offering a reliable supramolecular receptor supplier pathway for advanced diagnostic and analytical applications.
![Chemical structure of the asymmetric photoelectric active thiacalix[4]arene receptor molecule Fc-TA-RB featuring ferrocene and rhodamine B groups](/insights/img/asymmetric-thiacalixarene-synthesis-pharma-supplier-20260308135036-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiacalix[4]arene-based receptor molecules has been dominated by symmetric design strategies. In these conventional approaches, researchers typically functionalize the lower rim hydroxyl groups at the 1,3-positions identically, resulting in molecules that possess structural symmetry. While these symmetric receptors are easier to synthesize, they suffer from a fundamental functional deficit: they generally contain only a single type of signal-responsive group, such as solely a fluorophore or solely an electroactive unit. This single-channel limitation restricts their ability to perform effective detection of specific substrates, particularly in complex environments where background noise or interference can obscure a single signal type. Furthermore, the lack of orthogonality in signal output makes it difficult to cross-verify results, limiting the reliability of these molecules in high-precision supermolecule recognition tasks and restricting their broader application in advanced analytical instrumentation.
The Novel Approach
The novel approach detailed in this patent fundamentally disrupts the status quo by employing an asymmetric design strategy that leverages the versatile modification sites of the thiacalix[4]arene scaffold. Instead of simultaneous or identical functionalization, the method utilizes a selective stepwise acylation reaction to install two chemically distinct probes onto the molecular platform. This results in the successful synthesis of the Fc-TA-RB molecule, which uniquely integrates a ferrocene group for electrochemical response and a rhodamine B group for fluorescence response. This dual-functionality lays the foundation for optical and electrical dual-channel response detection, allowing for the simultaneous monitoring of a target substrate through two independent physical mechanisms. For manufacturers of electronic chemical manufacturing components, this approach offers a pathway to produce high-purity OLED material precursors or sensor interfaces that provide redundant, high-fidelity data output, significantly enhancing the performance envelope of the final device.
Mechanistic Insights into Selective Stepwise Acylation
The core chemical innovation lies in the precise control of the acylation reactions to achieve asymmetry on a substrate that inherently possesses two chemically similar amine groups. The synthesis begins with p-tert-butylthiacalix[4]arene-1,3-diamine, which serves as the foundational scaffold. The first critical step involves the selective mono-acylation of one of the amine groups with ferrocenecarbonyl chloride. This reaction requires meticulous control over reaction parameters, specifically maintaining a temperature range of -10°C to 25°C, with an optimal window of -5°C to 10°C. The use of a base such as triethylamine in a dichloromethane solvent system facilitates the nucleophilic attack while the controlled stoichiometry (molar ratio of 1:1.2 to 1:1.5) ensures that the second amine group remains largely unreacted. This kinetic control is essential to prevent the formation of the symmetric bis-ferrocene byproduct, which would be difficult to separate and would defeat the purpose of the asymmetric design.
Following the isolation of the mono-acylated intermediate TA-Fc, the second stage of the synthesis introduces the optical component. The intermediate is reacted with rhodamine B acid chloride under similar basic conditions but at a slightly elevated temperature range of 0°C to 30°C. This step completes the asymmetric functionalization, attaching the bulky and highly conjugated rhodamine moiety to the remaining amine site. The mechanistic success of this route relies on the stability of the amide bond formed in the first step, which withstands the conditions of the second acylation. The resulting molecule, Fc-TA-RB, exhibits a complex impurity profile that must be managed through rigorous purification, typically involving column chromatography with specific solvent systems like ethyl acetate and petroleum ether. This mechanistic understanding is crucial for the commercial scale-up of complex polymer additives or fine chemical intermediates, ensuring that the final product meets the stringent purity specifications required for sensitive electrochemical applications.
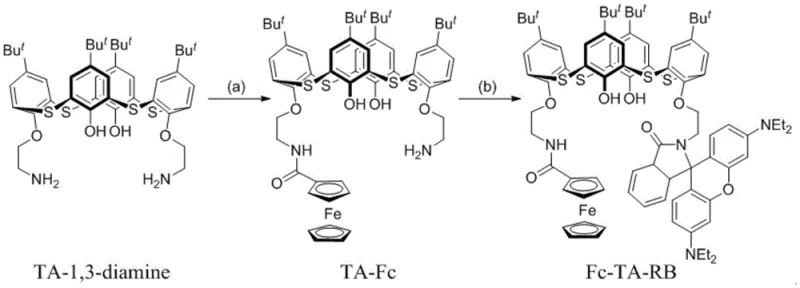
How to Synthesize Fc-TA-RB Efficiently
The synthesis of this high-value asymmetric receptor requires a disciplined approach to reaction engineering and purification to ensure the integrity of the dual-channel signaling capability. The process is defined by two distinct acylation events that must be isolated and purified individually to prevent cross-contamination of symmetric byproducts. The initial formation of the ferrocene-amide linkage sets the stage for the subsequent attachment of the rhodamine fluorophore, creating a molecule that bridges the gap between electrochemical and optical sensing modalities. Detailed operational parameters regarding solvent selection, base equivalents, and temperature gradients are critical for maximizing yield and minimizing the formation of difficult-to-remove impurities. For technical teams looking to implement this chemistry, the following guide outlines the standardized synthetic steps derived from the patent data to ensure reproducibility and quality.
- Perform selective mono-acylation of p-tert-butylthiacalix[4]arene-1,3-diamine with ferrocenecarbonyl chloride using triethylamine in dichloromethane at low temperature to form intermediate TA-Fc.
- Purify the intermediate TA-Fc via column chromatography using a dichloromethane and methanol solvent system to ensure high purity before the second step.
- React the purified TA-Fc intermediate with rhodamine B acid chloride in the presence of a base to complete the asymmetric functionalization, yielding the final Fc-TA-RB receptor.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this asymmetric synthesis route offers substantial strategic advantages for procurement managers and supply chain heads focused on cost reduction in electronic chemical manufacturing. The primary economic driver is the elimination of complex protection and deprotection sequences that are traditionally required to achieve asymmetry in diamine functionalization. By relying on kinetic control and selective stoichiometry rather than orthogonal protecting groups, the process significantly reduces the number of unit operations, thereby lowering solvent consumption, waste generation, and overall processing time. This streamlined workflow translates directly into improved cost efficiency and a reduced environmental footprint, aligning with modern green chemistry initiatives and regulatory compliance standards without compromising the structural complexity of the final product.
- Cost Reduction in Manufacturing: The synthetic route utilizes readily available starting materials such as p-tert-butylthiacalix[4]arene-1,3-diamine and common acyl chlorides, which are commercially accessible and do not require custom synthesis. The avoidance of expensive protecting group chemistry and the use of standard solvents like dichloromethane and triethylamine further drive down the bill of materials. Additionally, the high selectivity achieved through temperature control minimizes the loss of valuable ferrocene and rhodamine derivatives to side reactions, ensuring that raw material utilization is optimized. This efficiency creates a robust margin structure that allows for competitive pricing in the high-performance sensor market while maintaining profitability.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions enhances the resilience of the supply chain against disruptions. Unlike processes that depend on exotic catalysts or specialized reagents with long lead times, this method utilizes reagents that are widely stocked by global chemical suppliers. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and stoichiometries, also reduces the risk of batch failures due to minor process variations. This reliability ensures consistent delivery schedules for downstream clients who depend on these specialized intermediates for the production of critical sensing devices and analytical instruments.
- Scalability and Environmental Compliance: The stepwise nature of the synthesis allows for easy scalability from laboratory benchtop to pilot plant and eventually to commercial production volumes. Each step can be optimized independently, and the purification methods, while currently described as column chromatography in the patent, can be adapted to crystallization or preparative HPLC for larger scales to improve throughput. Furthermore, the process generates manageable waste streams primarily consisting of amine salts and organic solvents, which can be effectively treated or recycled. This scalability supports the growing demand for high-purity supramolecular receptors in emerging fields like IoT sensors and biomedical diagnostics, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the Fc-TA-RB receptor molecule. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of this technology for potential partners. Understanding the nuances of the asymmetric design and the purification requirements is essential for evaluating the suitability of this intermediate for specific end-use applications. We encourage technical teams to review these details to assess how this innovation can be integrated into their existing development pipelines.
Q: What is the primary advantage of the asymmetric design in this thiacalixarene receptor?
A: The asymmetric design allows for the integration of two distinct signal transduction mechanisms—electrochemical (ferrocene) and fluorescent (rhodamine B)—on a single molecular platform. This enables dual-channel response detection, significantly improving the reliability and accuracy of substrate recognition compared to traditional symmetric receptors that rely on a single signal type.
Q: How is selectivity achieved during the first acylation step?
A: Selectivity is achieved through precise control of reaction stoichiometry and temperature. By maintaining the reaction temperature between -10°C and 25°C and carefully controlling the molar ratio of ferrocenecarbonyl chloride to the diamine substrate, the process favors mono-substitution, preventing the formation of symmetric bis-acylated byproducts.
Q: What are the key purification challenges for this molecule?
A: The primary challenge lies in separating the desired asymmetric product from unreacted starting materials and potential symmetric byproducts. The patent specifies the use of gradient column chromatography, utilizing dichloromethane/methanol for the intermediate and ethyl acetate/petroleum ether for the final product, to achieve the high purity required for sensitive electrochemical and optical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fc-TA-RB Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of asymmetric thiacalixarene receptors in the field of advanced sensing and supramolecular recognition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like Fc-TA-RB can be manufactured with the consistency and quality required for industrial deployment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the dual-channel functionality of these receptors, guaranteeing that every batch meets stringent purity specifications. We understand that the transition from patent to production requires not just chemical expertise but also a deep commitment to process safety and regulatory compliance.
We invite R&D directors and procurement leaders to collaborate with us to unlock the full commercial potential of this technology. By leveraging our process optimization capabilities, we can help you achieve significant cost reductions and accelerate your time-to-market for next-generation sensor technologies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your supply chain goals and drive innovation in your product portfolio.
