Advanced Spirofluorene Platinum Complexes for High-Efficiency OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer superior efficiency and stability for next-generation display technologies. Patent CN115651029A introduces a groundbreaking class of spirofluorene-biphenyl-azaacridine-based quadridentate cyclometalated platinum (II) complexes designed to overcome the limitations of traditional phosphorescent emitters. This innovation addresses the critical need for high-purity OLED material suppliers who can deliver compounds with narrow emission spectra and reduced shoulder peaks. By leveraging a unique 6/5/6 fused ring system, this technology enables precise control over photophysical properties, ensuring high thermal stability and efficient luminescence essential for commercial display and illumination applications. The strategic integration of spirofluorene units provides the necessary steric bulk to suppress concentration quenching, a common failure mode in planar metal complexes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphorescent platinum (II) complexes often suffer from significant efficiency losses due to planar molecular structures that facilitate detrimental intermolecular interactions. In standard designs, the flat geometry allows molecules to stack closely together, leading to pi-pi accumulation and strong platinum-platinum interactions in the solid state. These interactions create non-radiative decay pathways that drastically reduce luminescent efficiency and broaden the emission spectrum, resulting in poor color purity for display applications. Furthermore, conventional ligands often lack the rigidity required to prevent molecular deformation during operation, which increases non-radiative transitions and lowers the overall quantum yield. The inability to effectively manage triplet excitons without heavy aggregation limits the practical lifespan and performance consistency of devices using these older generation materials in high-brightness environments.
The Novel Approach
The novel approach detailed in the patent utilizes a rational ligand design incorporating a spirofluorene-biphenyl-azaacridine framework to fundamentally alter the molecular topology. By introducing the bulky spirofluorene group, the synthesis creates a three-dimensional structure that physically separates adjacent molecules, effectively suppressing the pi-pi stacking that plagues planar analogues. This structural modification not only enhances the rigidity of the molecule through the biphenyl linkage but also fine-tunes the HOMO and LUMO energy levels for optimal charge transport. The result is a phosphorescent complex that maintains high efficiency even at high concentrations, with a narrow emission spectrum ideal for wide color gamut displays. This method represents a significant leap forward in cost reduction in electronic chemical manufacturing by improving material utilization rates and device longevity.
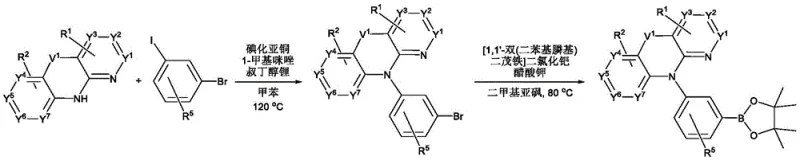
Mechanistic Insights into Spirofluorene-Stabilized Cyclometalation
The core mechanism driving the superior performance of these materials lies in the precise orchestration of steric and electronic effects within the quadridentate ligand system. The spirofluorene moiety acts as a rigid spacer that locks the molecular conformation, preventing the structural relaxation that typically dissipates energy as heat rather than light. This rigidity ensures that the excited state energy is channeled efficiently into radiative decay, maximizing the internal quantum efficiency towards the theoretical limit of 100%. Additionally, the electron-donating or withdrawing nature of substituents on the azaacridine ring allows for fine-tuning of the emission wavelength without compromising the stability of the platinum-carbon bonds. This level of control is crucial for achieving the specific color coordinates required by major panel manufacturers for red, green, and blue sub-pixels in full-color displays.
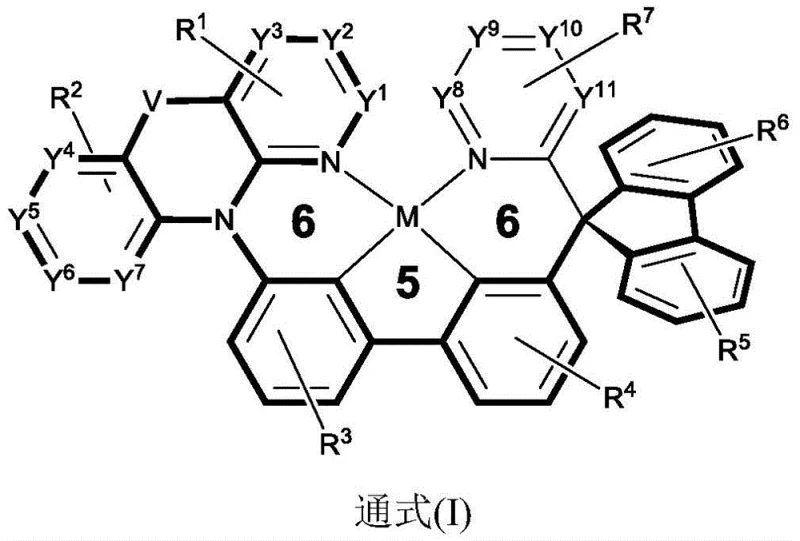
Impurity control is another critical aspect where this mechanistic design offers substantial advantages over conventional syntheses. The robust nature of the 6/5/6 fused ring system minimizes the formation of side products during the cyclometalation step, which is often the most challenging phase of platinum complex synthesis. By reducing the presence of structural isomers and unreacted intermediates, the downstream purification process becomes significantly more straightforward and cost-effective. This purity is paramount for R&D directors focusing on the杂质谱 (impurity profile) of materials, as trace impurities can act as trap sites for charge carriers, leading to premature device failure. The synthetic route ensures that the final product meets stringent purity specifications required for high-end commercial OLED panels without requiring excessive chromatographic separation steps.
How to Synthesize Spirofluorene Platinum Complex Efficiently
The synthesis of these high-performance emitters follows a logical progression starting from readily available raw materials to ensure supply chain continuity. The process begins with the construction of the spirofluorene core through lithiation and cyclization, followed by coupling with the nitrogen-containing heterocycle to form the complete tetradentate ligand. Detailed standardized synthesis steps see the guide below, which outlines the specific reaction conditions, stoichiometry, and purification methods validated in the patent examples. This structured approach allows for reproducible results across different batches, a key requirement for industrial scale-up. The use of common catalysts like palladium tetrakis and standard solvents ensures that the process can be adapted to existing manufacturing infrastructure without needing specialized equipment modifications.
- Synthesize the spirofluorene-biphenyl intermediate via lithiation and nucleophilic addition followed by acid-catalyzed cyclization.
- Perform Suzuki-Miyaura coupling between the brominated spirofluorene derivative and the azaacridine boronic ester to form the tetradentate ligand.
- React the purified ligand with platinum dichloride in benzonitrile at elevated temperatures to achieve the final cyclometalated phosphorescent complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this spirofluorene-based technology translates into tangible operational benefits beyond mere performance metrics. The synthetic route avoids the use of exotic or highly unstable reagents, relying instead on robust cross-coupling chemistry that is well-understood in the fine chemical industry. This reliability significantly reduces the risk of batch failures and supply disruptions, ensuring a steady flow of materials for continuous production lines. Moreover, the enhanced thermal stability of the final complex means that less material is wasted during the vacuum deposition process, directly contributing to substantial cost savings in material consumption. The ability to produce high-purity material with fewer purification steps also shortens the overall manufacturing cycle time, enhancing responsiveness to market demands.
- Cost Reduction in Manufacturing: The elimination of complex purification stages required for planar complexes leads to a more streamlined production workflow. By preventing aggregation through molecular design, the need for extensive chromatographic separation is reduced, lowering solvent usage and waste disposal costs. This qualitative improvement in process efficiency drives down the overall cost of goods sold without compromising the quality of the final electronic chemical. The robust yield observed in the patent examples suggests that scaling this process will maintain economic viability even at large volumes.
- Enhanced Supply Chain Reliability: The reliance on standard organic synthesis transformations such as Suzuki coupling ensures that raw material sourcing is not dependent on single-source suppliers of niche intermediates. This diversification of the supply base mitigates the risk of geopolitical or logistical bottlenecks that often plague the specialty chemical sector. Furthermore, the stability of the intermediates allows for safer storage and transportation, reducing the likelihood of degradation during transit. This reliability is critical for maintaining just-in-time inventory systems used by major display manufacturers.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reaction conditions that can be safely translated from laboratory glassware to industrial reactors. The reduction in hazardous waste generation, due to higher selectivity and fewer purification steps, aligns with increasingly strict environmental regulations governing chemical manufacturing. This compliance reduces the regulatory burden on production facilities and supports corporate sustainability goals. The process demonstrates a clear path for commercial scale-up of complex organometallic compounds while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spirofluorene-biphenyl-azaacridine technology in OLED production. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation to ensure accuracy. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their product roadmaps. The focus is on practical applicability, performance consistency, and long-term viability in commercial devices.
Q: How does the spirofluorene structure improve OLED efficiency?
A: The bulky spirofluorene group introduces significant steric hindrance, preventing intermolecular pi-pi stacking and platinum-platinum interactions that typically cause luminescence quenching in planar Pt complexes.
Q: What is the thermal stability of these platinum complexes?
A: The rigid 6/5/6 fused ring structure combined with the spirofluorene moiety enhances thermal stability, making the material suitable for vacuum evaporation processes in OLED device fabrication.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the synthesis relies on standard cross-coupling reactions like Suzuki coupling and conventional cyclometalation, which are well-established and scalable in industrial chemical manufacturing settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirofluorene Platinum Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced emissive materials play in the future of display technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity phosphorescent emitter meets the exacting standards of the global electronics industry. Our commitment to quality assurance means that you can rely on us for consistent material performance across your entire production volume.
We invite you to contact our technical procurement team to discuss how we can support your specific material requirements. Request a Customized Cost-Saving Analysis to understand how switching to this optimized synthetic route can impact your bottom line. We are ready to provide specific COA data and route feasibility assessments to help you evaluate the potential of this technology for your next-generation OLED projects. Partner with us to secure a stable supply of cutting-edge electronic chemicals that drive innovation.
