Advanced Synthesis of 2-Bromo-5-p-toluenesulfonyl-5H-pyrrolopyrazine for Commercial Scale-Up
The pharmaceutical industry continuously demands more efficient and safer pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as critical building blocks for kinase inhibitors and other targeted therapies. Patent CN113603693B, published in August 2022, introduces a groundbreaking preparation method for 2-bromo-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine, a vital fine chemical raw material and medical intermediate. This innovation addresses long-standing challenges in the synthesis of pyrrolopyrazine derivatives by replacing hazardous reagents with mild alkaline conditions and simplifying the purification workflow. The core breakthrough lies in the strategic use of mild alkali reagents, such as sodium tert-butoxide, which facilitates a controlled cyclization and subsequent sulfonylation without the extreme risks associated with traditional strong bases. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process safety and economic viability. By fundamentally altering the reaction landscape, this patent enables the production of high-purity pyrrolopyrazine derivatives with markedly improved yields and reduced operational complexity.
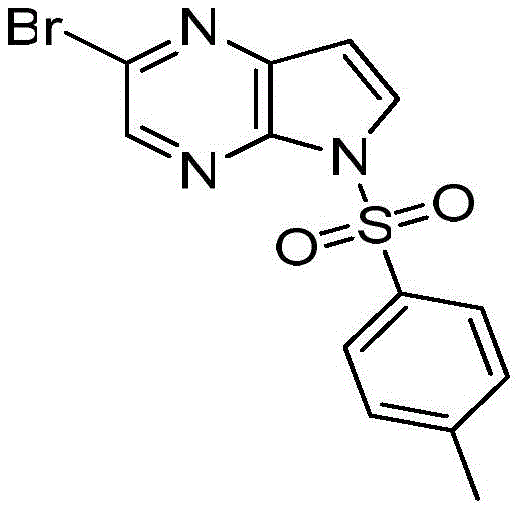
The molecular architecture of 2-bromo-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine (CAS: 1201186-54-0) is characterized by a fused pyrrolopyrazine core substituted with a bromine atom and a p-toluenesulfonyl group. This specific substitution pattern is crucial for downstream coupling reactions in drug discovery, making the availability of this intermediate a key bottleneck in many development pipelines. The patent details a three-step one-pot strategy that begins with the dissolution of 3-[(trimethylsilyl)ethynyl]-5-pyrazine-2-amine in N,N-dimethylacetamide (DMAc). The introduction of the mild base initiates an intramolecular cyclization, forming the pyrrole ring fused to the pyrazine. Subsequent addition of p-toluenesulfonyl chloride at room temperature installs the protecting group, stabilizing the nitrogen atom for further functionalization. This streamlined approach not only enhances the chemical integrity of the final product but also aligns perfectly with the goals of cost reduction in fine chemical manufacturing by minimizing solvent exchanges and unit operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar pyrrolopyrazine scaffolds has been plagued by significant safety hazards and inefficiencies that hinder commercial scale-up. Traditional protocols frequently rely on sodium hydride (NaH) as the base for cyclization, a reagent known for its pyrophoric nature and potential to generate hydrogen gas, posing severe explosion risks in large reactors. Furthermore, these older methods often suffer from poor selectivity, leading to a complex mixture of byproducts that are difficult to separate. To achieve the necessary purity for pharmaceutical applications, manufacturers are typically forced to employ silica gel column chromatography, a technique that is notoriously expensive, time-consuming, and difficult to scale beyond kilogram quantities. The reliance on such labor-intensive purification not only inflates the cost of goods sold (COGS) but also extends the production lead time, creating bottlenecks for supply chain heads who require consistent and rapid delivery of high-purity intermediates. Additionally, the harsh conditions associated with strong bases can degrade sensitive functional groups, further reducing overall yield and complicating the impurity profile.
The Novel Approach
In stark contrast, the methodology disclosed in CN113603693B offers a transformative solution by utilizing mild alkali reagents such as sodium tert-butoxide, potassium carbonate, or cesium carbonate. This shift in reagent choice fundamentally changes the reaction profile, allowing the cyclization to proceed smoothly at moderate temperatures ranging from 40°C to 100°C. The mildness of these bases significantly reduces the formation of unwanted side products, resulting in a cleaner reaction crude that is amenable to simple workup procedures. Instead of resorting to column chromatography, the new process employs a straightforward quench into a dilute sodium bicarbonate solution followed by filtration and recrystallization. This change eliminates the need for vast quantities of silica gel and organic solvents typically used in chromatography, directly translating to substantial cost savings and a reduced environmental footprint. The ability to isolate the product through crystallization also ensures a more consistent particle size and purity profile, which is critical for downstream processing in API manufacturing. Consequently, this novel approach not only mitigates safety risks but also dramatically enhances the economic feasibility of producing this valuable intermediate on a multi-ton scale.
Mechanistic Insights into Mild Base-Catalyzed Cyclization and Sulfonylation
The success of this synthesis hinges on the precise mechanistic interplay between the trimethylsilyl-protected alkyne precursor and the mild base in a polar aprotic solvent like DMAc. Upon heating, the mild base deprotonates the amine or activates the alkyne moiety, triggering a 5-endo-dig or similar cyclization pathway that constructs the pyrrole ring fused to the pyrazine core. The use of sodium tert-butoxide is particularly effective because it provides sufficient basicity to drive the cyclization without promoting excessive decomposition or polymerization of the sensitive heterocyclic intermediates. The reaction temperature is carefully controlled between 40°C and 100°C to optimize the kinetics of ring closure while maintaining thermal stability. Once the cyclic intermediate is formed, the system is cooled to room temperature before the addition of p-toluenesulfonyl chloride. This sequential addition is crucial; adding the sulfonylating agent at lower temperatures prevents competitive side reactions and ensures selective N-sulfonylation of the newly formed pyrrole nitrogen. The molar ratio of the base to the amine precursor is maintained between 1.0:1.0 and 1.5:1.0, ensuring complete conversion without a large excess of reagents that would complicate the workup. This controlled mechanistic pathway is the key to achieving the high molar yields reported in the patent examples.
Impurity control is another critical aspect where this mechanism excels, directly addressing the concerns of R&D directors regarding product quality. In conventional harsh base methods, over-reaction or degradation often leads to tarry byproducts that co-elute with the desired product. By employing mild conditions and a specific quenching protocol using 0.5-10% wt sodium bicarbonate solution, the new process effectively neutralizes acidic byproducts and unreacted sulfonyl chloride without hydrolyzing the sensitive bromo-pyrrolopyrazine core. The subsequent recrystallization step, utilizing a mixed solvent system of ethyl acetate and petroleum ether, leverages the solubility differences between the target molecule and any remaining minor impurities. This physical purification method is highly effective at removing trace organics and inorganic salts, resulting in a final product with exceptional purity suitable for direct use in subsequent cross-coupling reactions. The structural integrity of the final compound is confirmed through rigorous analytical techniques, including 1H-NMR and 13C-NMR, which show distinct signals corresponding to the methyl group of the tosyl moiety and the characteristic protons of the fused ring system, validating the efficacy of this impurity control strategy.
How to Synthesize 2-Bromo-5-p-toluenesulfonyl-5H-pyrrolopyrazine Efficiently
Implementing this synthesis route requires careful attention to the sequence of addition and temperature control to maximize yield and safety. The process begins by charging a reactor with N,N-dimethylacetamide and the amine precursor, followed by the slow addition of the mild base under heating. Once the cyclization is complete, indicated by the consumption of starting material, the mixture must be cooled before introducing the sulfonyl chloride to prevent exothermic runaway. The final isolation involves a simple aqueous workup and crystallization, making it highly adaptable for both pilot and commercial plants. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Dissolve 3-[(trimethylsilyl)ethynyl]-5-pyrazine-2-amine in N,N-dimethylacetamide (DMAc) and react with a mild alkali reagent like sodium tert-butoxide at 40-100°C for 0.5-2 hours.
- Cool the reaction system to room temperature, add p-toluenesulfonyl chloride, and maintain at room temperature for 0.5-2 hours to complete the sulfonylation.
- Quench the mixture into 0.5-10% wt sodium bicarbonate solution, filter the solid, recrystallize from ethyl acetate/petroleum ether, and dry to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers compelling advantages that extend far beyond simple chemical yield improvements. The elimination of hazardous reagents like sodium hydride removes a major safety liability, potentially lowering insurance costs and simplifying regulatory compliance for storage and handling. Furthermore, the replacement of silica gel column chromatography with recrystallization represents a massive reduction in consumable costs and waste generation. Silica gel is expensive and creates significant solid waste disposal issues, whereas recrystallization solvents can often be recovered and recycled. This shift drastically simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor hours required per batch. Consequently, the overall production cycle time is shortened, enabling faster turnaround times for custom synthesis orders and improving the agility of the supply chain in response to market demands. These factors collectively contribute to a more robust and cost-effective supply model for this critical intermediate.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the complete removal of column chromatography, which is traditionally one of the most expensive steps in fine chemical production due to the high cost of silica gel and the large volumes of solvents required. By optimizing the reaction to produce a clean crude product that can be purified via simple filtration and recrystallization, the material costs are significantly lowered. Additionally, the use of mild, commercially available bases like sodium tert-butoxide or potassium carbonate is more economical and easier to handle than specialized or hazardous reagents. The higher molar yields, consistently exceeding 83%, mean that less raw material is wasted, further driving down the cost per kilogram of the final API intermediate. This efficiency allows suppliers to offer more competitive pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain reliability is greatly enhanced by the simplicity and safety of the new protocol. Processes that rely on dangerous reagents often face interruptions due to strict safety audits or transportation restrictions on hazardous materials. By switching to mild conditions, the manufacturing process becomes more resilient to regulatory changes and logistical hurdles. The use of common solvents like DMAc, ethyl acetate, and petroleum ether ensures that raw material sourcing is stable and not subject to the volatility of niche chemical markets. Moreover, the robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality material for downstream customers. This predictability is essential for pharmaceutical companies planning long-term production schedules for their own drug candidates.
- Scalability and Environmental Compliance: Scalability is a inherent feature of this design, as the removal of chromatography removes the primary bottleneck for scaling heterocyclic synthesis. Crystallization is a standard unit operation in the chemical industry that scales linearly from grams to tons, unlike chromatography which becomes impractical at large scales. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates. From an environmental perspective, the reduction in solvent usage and the elimination of silica gel waste align with green chemistry principles. The simplified workup generates less hazardous waste, reducing the burden on waste treatment facilities and lowering the environmental compliance costs for the manufacturer. This sustainability angle is increasingly important for multinational corporations aiming to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-bromo-5-p-toluenesulfonyl-5H-pyrrolopyrazine. These answers are derived directly from the technical specifications and beneficial effects outlined in patent CN113603693B, providing clarity on how this new method outperforms legacy technologies. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their development pipelines.
Q: Why is the new preparation method safer than conventional routes?
A: Conventional methods often utilize hazardous reagents like sodium hydride (NaH), which pose significant explosion and fire risks. The patented process replaces these with mild alkali reagents such as sodium tert-butoxide or potassium carbonate, drastically reducing operational hazards and making the process suitable for large-scale industrial environments.
Q: How does this process improve purification efficiency?
A: Traditional synthesis often requires tedious and expensive silica gel column chromatography to remove byproducts. This novel method optimizes reaction conditions to minimize byproduct formation, allowing for simple filtration and recrystallization (e.g., using ethyl acetate/petroleum ether) to achieve high purity, thereby significantly lowering production costs and time.
Q: What yields can be expected from this synthesis route?
A: According to the experimental data in patent CN113603693B, the optimized process consistently achieves molar yields ranging from 83.4% to 86.9%. This high efficiency, combined with the elimination of complex purification steps, ensures a robust supply chain for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-5-p-toluenesulfonyl-5H-pyrrolopyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial production requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly move from clinical trials to market launch. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-bromo-5-p-toluenesulfonyl-5H-pyrrolopyrazine meets the highest international standards. We are committed to implementing the latest process innovations, such as the mild-base cyclization route described in CN113603693B, to deliver superior value to our global partners.
We invite you to collaborate with us to leverage these advanced synthesis technologies for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can reduce your overall COGS. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your supply chain goals.
