Advanced Manufacturing of High-Melting Fluoran Dyes via Stabilized N-Formyl Precursors
Advanced Manufacturing of High-Melting Fluoran Dyes via Stabilized N-Formyl Precursors
The global demand for high-performance thermal recording materials continues to drive innovation in the synthesis of fluoran-based leuco dyes. A pivotal advancement in this sector is detailed in patent CN1854201B, which discloses a robust method for preparing 3-dibutyl-amino-6-methyl-7-aniline fluorane, a critical component in thermosensitive paper coatings. This technology addresses long-standing challenges regarding raw material stability and reaction selectivity that have plagued conventional manufacturing routes. By introducing an N-formyl protection strategy on the diphenylamine precursor, the process achieves superior control over the electrophilic aromatic substitution, resulting in a direct pathway to the high-melting crystal form of the dye. For procurement specialists and R&D leaders, this represents a significant opportunity to optimize the supply chain for reliable dye intermediate suppliers while ensuring consistent quality in the final thermal paper products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
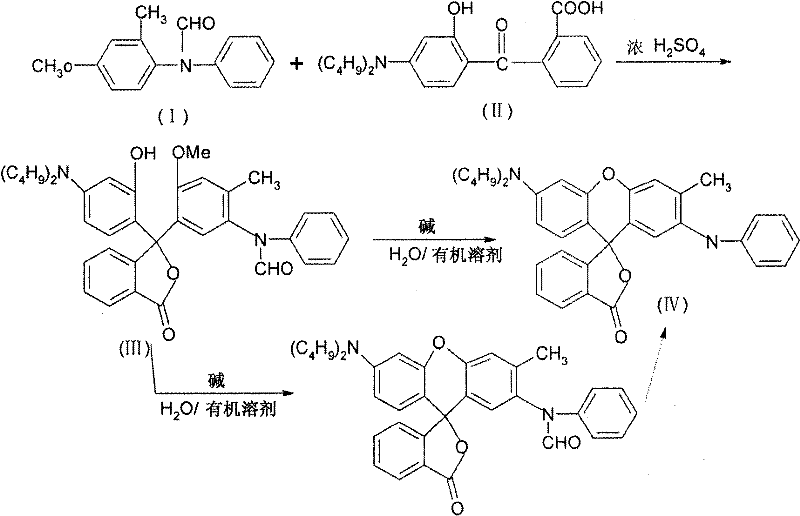
Historically, the synthesis of 3-dibutyl-amino-6-methyl-7-aniline fluorane relied on the condensation of unprotected 4-methoxy-2-methyldiphenylamine with 2-(4-dibutylamino-2-hydroxybenzoyl)benzoic acid. This traditional approach, documented in earlier patents such as US 5,110,952, suffers from inherent chemical instabilities. The unprotected amine moiety is highly susceptible to oxidation during storage and handling, leading to variable starting material quality that complicates process validation. Furthermore, during the acidic condensation phase, the free amine nitrogen can participate in undesirable side reactions with the carboxylic acid functionality of the benzoyl benzoic acid derivative. These competing pathways reduce the overall atom economy and generate complex impurity profiles that are difficult to remove. Consequently, conventional methods often yield a mixture of crystal modifications, requiring additional energy-intensive recrystallization steps to isolate the desired high-melting form, thereby inflating production costs and extending lead times.
The Novel Approach
The methodology outlined in CN1854201B introduces a strategic modification by utilizing N-formyl-4-methoxy-2-methyldiphenylamine as the nucleophilic partner. This N-formyl protected precursor exhibits markedly improved stability against oxidation, allowing for prolonged storage without significant degradation, which is a crucial advantage for inventory management. In the reaction matrix, the formyl group effectively masks the nucleophilicity of the nitrogen atom, directing the electrophilic attack exclusively to the aromatic ring and preventing intermolecular amidation side reactions. This enhancement in chemoselectivity translates directly to higher crude purity and simplified downstream processing. Moreover, the process elegantly integrates the deprotection step; the formyl group is spontaneously cleaved under the basic conditions required for the final lactonization, eliminating the need for a separate hydrolysis unit operation. This streamlined workflow not only boosts the isolated yield to approximately 83% but also ensures the direct formation of the thermodynamically stable high-melting polymorph.
Mechanistic Insights into Acid-Catalyzed Condensation and Lactonization
The core of this synthetic route lies in the precise control of electrophilic aromatic substitution within a highly acidic medium. In the presence of concentrated sulfuric acid, the 2-(4-dibutylamino-2-hydroxybenzoyl)benzoic acid undergoes protonation to generate a highly reactive acylium ion or a protonated carbonyl species. This electrophile attacks the electron-rich aromatic ring of the N-formyl-4-methoxy-2-methyldiphenylamine at the position ortho to the methoxy group, driven by the strong activating effect of the alkoxy substituent. The presence of the formyl group on the adjacent nitrogen is critical here; it reduces the electron density on the nitrogen, preventing it from acting as a competing nucleophile that could trap the activated carboxylic acid. This ensures that the reaction proceeds through the desired C-C bond formation to create the phthalide intermediate (Compound III) with high regioselectivity. The maintenance of temperature between 15-25°C during this exothermic condensation is vital to suppress potential sulfonation of the aromatic rings or decomposition of the acid-sensitive formyl group prior to the intended cyclization stage.
Following the isolation of the phthalide intermediate, the transformation to the final fluorane structure involves a base-mediated intramolecular cyclization. Upon treatment with a strong base such as sodium hydroxide or potassium carbonate in a polar organic solvent, the phenolic hydroxyl group generated (or unmasked) acts as a nucleophile. It attacks the lactone carbonyl or the adjacent electrophilic center, closing the xanthene ring system characteristic of fluoran dyes. Simultaneously, the highly basic environment (pH ≥ 11) facilitates the hydrolysis of the N-formyl protecting group. This tandem cyclization-deprotection sequence is kinetically favorable and thermodynamically driven by the formation of the conjugated, planar fluorane system. The result is the expulsion of the formate species and the establishment of the secondary amine linkage in the final product, yielding the target 3-dibutyl-amino-6-methyl-7-aniline fluorane with a purity exceeding 99.5%.
How to Synthesize 3-Dibutyl-Amino-6-Methyl-7-Aniline Fluorane Efficiently
The operational protocol for this synthesis is designed for scalability and safety, leveraging standard industrial unit operations. The process begins with the controlled addition of reactants into concentrated sulfuric acid, requiring efficient heat exchange systems to maintain the critical 15-25°C window. Following the condensation period, the reaction mass is quenched into cold water, inducing precipitation of the intermediate phthalide, which is then filtered and washed to remove residual acid. The subsequent cyclization step utilizes common organic solvents such as toluene, ethylene glycol, or chlorobenzene mixed with aqueous base, allowing for flexible solvent recovery strategies. Detailed standardized operating procedures regarding stoichiometry, agitation rates, and specific workup parameters are essential for reproducibility.
- Condense N-formyl-4-methoxy-2-methyldiphenylamine with 2-(4-dibutylamino-2-hydroxybenzoyl)benzoic acid in concentrated sulfuric acid at 15-25°C.
- Quench the reaction mixture into cold water to precipitate the phthalide intermediate (Compound III).
- Reflux the intermediate in an organic solvent with aqueous base (pH ≥ 11) to effect cyclization and simultaneous deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this N-formyl protection strategy offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the stabilization of the supply chain for raw materials. Unlike the unprotected amine used in legacy processes, which requires careful handling and rapid turnover to avoid oxidative degradation, the N-formyl precursor is robust and shelf-stable. This stability reduces the risk of batch-to-batch variability caused by raw material aging, thereby minimizing production delays and quality deviations. Furthermore, the elimination of side reactions means that less raw material is wasted on impurity formation, directly contributing to cost reduction in thermosensitive material manufacturing. The higher yield per batch implies that fewer tons of starting materials need to be purchased and transported to achieve the same output of finished dye, optimizing logistics costs and warehouse utilization.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous reagents often required to manage the instability of unprotected amines in older methods. By preventing side reactions through electronic protection, the consumption of the valuable benzoic acid derivative is minimized, as it is not consumed by competing amidation pathways. Additionally, the spontaneous removal of the formyl group during the cyclization step removes the requirement for a dedicated hydrolysis reactor and the associated utility costs (steam, water, time) for a separate processing stage. This consolidation of steps leads to substantial operational expenditure savings and a reduced carbon footprint per kilogram of product manufactured.
- Enhanced Supply Chain Reliability: The use of a stable, storable intermediate (Compound I) decouples the synthesis of the dye from the immediate production of the amine precursor. Manufacturers can stockpile the N-formyl amine during periods of low demand without fear of spoilage, creating a buffer against upstream supply disruptions. This inventory flexibility allows for more consistent production scheduling and shorter lead times for customers requiring high-purity fluoran coupler materials. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility quality or operator technique, further ensuring a steady flow of goods to the market.
- Scalability and Environmental Compliance: The synthesis avoids the use of heavy metal catalysts or exotic reagents, relying instead on commodity chemicals like sulfuric acid and sodium hydroxide. This simplifies waste stream management, as the effluent primarily consists of inorganic salts and organic solvents that can be readily treated or recycled. The direct formation of the high-melting crystal form eliminates the need for solvent-intensive recrystallization cycles typically used to convert low-melting polymorphs, significantly reducing volatile organic compound (VOC) emissions. This alignment with green chemistry principles facilitates easier regulatory compliance and supports the sustainability goals of downstream paper manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this chemistry into their existing production lines or sourcing strategies. The answers are derived directly from the experimental data and comparative examples provided in the patent literature, ensuring accuracy and relevance for industrial application.
Q: Why is the N-formyl protected amine preferred over the unprotected amine in fluorane synthesis?
A: The N-formyl group protects the amine nitrogen from oxidation and unwanted side reactions with the benzoic acid derivative during the acidic condensation step, significantly improving selectivity and final product purity.
Q: Does the deprotection of the formyl group require a separate reaction step?
A: No, the patent data indicates that the formyl group is spontaneously removed during the basic cyclization (reflux with NaOH/KOH), eliminating the need for a dedicated hydrolysis step and reducing processing time.
Q: What crystal form of the dye does this process produce?
A: This method directly yields the high-melting crystal modification (179-186°C range), which is superior for thermal recording applications due to better color density and background stability compared to low-melting forms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Dibutyl-Amino-6-Methyl-7-Aniline Fluorane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply continuity in the specialty chemicals sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of thermosensitive dye meets the exacting standards required for high-performance thermal paper applications. Our commitment to process excellence allows us to deliver commercial scale-up of complex organic dyes with reliability and speed.
We invite you to collaborate with us to optimize your supply chain for thermal recording materials. By leveraging our expertise in advanced organic synthesis, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project milestones and quality expectations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
