Advanced Catalyst-Free Synthesis of Coumarin Fluorochromes for Commercial Scale-up
Advanced Catalyst-Free Synthesis of Coumarin Fluorochromes for Commercial Scale-up
The development of high-performance fluorescent materials is critical for the advancement of optoelectronic devices, particularly in the realms of organic light-emitting diodes (OLEDs) and tunable laser systems. Patent CN101368004B introduces a groundbreaking synthetic methodology for producing coumarin fluorescent dyes, specifically targeting the structural Formula I derivatives. This innovation represents a significant departure from traditional multi-step syntheses by employing a direct, one-pot condensation reaction that eliminates the need for external catalysts. For R&D directors and procurement specialists in the electronic chemical sector, this patent offers a pathway to high-purity coumarin fluorochromes with reduced process complexity. The core reaction involves the reflux of ethyl cyanoacetate, 4-N,N-diethylaminosalicylaldehyde, and substituted o-aminophenols in a protonic polar solvent. By removing the dependency on harsh Lewis acids or transition metal catalysts, this method not only streamlines the manufacturing workflow but also inherently improves the purity profile of the final product, which is essential for applications requiring high fluorescence quantum efficiency and stability.
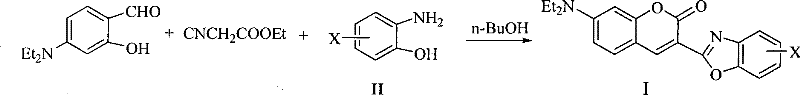
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of coumarin-based laser dyes and electroluminescent materials has been plagued by inefficient multi-step protocols that introduce significant operational burdens and cost inefficiencies. Traditional routes, such as those described in older US and German patents, often require the initial construction of the coumarin ring using strong acid catalysts like titanium tetrachloride (TiCl4) or benzoic acid in substantial quantities. These conventional methods necessitate rigorous purification steps to remove residual metal catalysts and acidic byproducts, which can degrade the optical properties of the dye and complicate waste treatment. Furthermore, alternative strategies involving the separate formation of benzoxazole rings prior to condensation add unnecessary synthetic steps, increasing the overall lead time and reducing the cumulative yield. The reliance on moisture-sensitive reagents and anhydrous conditions in these legacy processes further escalates the capital expenditure required for specialized reactor setups, making them less attractive for cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN101368004B revolutionizes the production landscape by enabling a direct, catalyst-free one-pot synthesis. This novel approach leverages the intrinsic reactivity of ethyl cyanoacetate and salicylaldehyde derivatives in a protic polar solvent environment, such as n-butanol or cyclohexanol, to drive the cyclization spontaneously under reflux conditions. By operating without added catalysts, the process inherently avoids the introduction of metallic impurities, thereby simplifying the downstream purification to a basic aqueous wash and filtration. This streamlined workflow not only reduces the consumption of auxiliary chemicals but also significantly shortens the production cycle, allowing for faster turnaround times. The ability to achieve yields ranging from 60% to 80% under such mild conditions demonstrates the robustness of this chemistry, positioning it as a superior alternative for the commercial scale-up of complex laser dyes where consistency and purity are paramount.
Mechanistic Insights into Catalyst-Free Condensation Cyclization
The mechanistic elegance of this synthesis lies in the concurrent formation of the coumarin and benzoxazole rings through a cascade of condensation and cyclization events driven solely by thermal energy in a protic solvent. The reaction initiates with a Knoevenagel condensation between the active methylene group of ethyl cyanoacetate and the aldehyde functionality of the 4-N,N-diethylaminosalicylaldehyde. In the absence of a catalyst, the protic solvent plays a crucial dual role: it acts as a heat transfer medium to maintain reflux temperatures and potentially stabilizes transition states through hydrogen bonding interactions. As the intermediate forms, the nucleophilic attack by the amino group of the o-aminophenol derivative triggers the closure of the benzoxazole ring, while the phenolic hydroxyl group participates in the lactonization to form the coumarin core. This concerted mechanism ensures that the reaction proceeds efficiently without the need for aggressive activation, minimizing side reactions such as polymerization or hydrolysis that are common in acid-catalyzed environments.
From an impurity control perspective, the absence of exogenous catalysts is a decisive factor in achieving the high purity required for electronic applications. In traditional catalytic routes, trace metals can act as quenchers for fluorescence or induce degradation over time, compromising the longevity of OLED devices. By eliminating these contaminants at the source, the new method produces a cleaner crude product that requires minimal post-reaction processing. The workup procedure described involves a simple alkaline wash to remove unreacted starting materials and soluble byproducts, followed by filtration. This simplicity translates directly to a narrower impurity profile, reducing the burden on analytical quality control teams who must verify that heavy metal levels meet stringent international standards for display and lighting materials. Consequently, this mechanistic advantage supports the production of high-purity coumarin fluorochromes suitable for high-end technological applications.
How to Synthesize Coumarin Fluorescent Dyes Efficiently
Implementing this synthesis protocol requires precise control over stoichiometry and reaction parameters to maximize yield and reproducibility. The patent specifies a molar ratio of ethyl cyanoacetate to aldehyde to aminophenol derivative ideally at 1:1:1, though slight excesses of the aldehyde or amine (up to 1.2 equivalents) can be tolerated to drive the equilibrium forward. The choice of solvent is critical, with n-butanol identified as the optimal medium due to its boiling point and polarity, which facilitate the necessary reflux conditions for 7 to 9 hours. Detailed standardized operating procedures regarding temperature ramping, agitation speeds, and specific workup concentrations are essential for transferring this laboratory success to pilot plant operations. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the technical breakdown below.
- Mix ethyl cyanoacetate, 4-N,N-diethylaminosalicylaldehyde, and o-aminophenol derivatives in a protic polar solvent like n-butanol.
- Heat the mixture to reflux temperature and maintain stirring for 7 to 9 hours to complete the cyclization.
- Cool the reaction to room temperature, treat with dilute NaOH solution to remove impurities, then filter and dry the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of expensive and hazardous catalysts like titanium tetrachloride removes a significant cost center associated with both raw material acquisition and hazardous waste disposal. Furthermore, the simplified one-pot nature of the reaction reduces the requirement for multiple reactor vessels and intermediate isolation steps, effectively lowering the capital intensity of the production line. This process intensification allows manufacturers to produce larger batches within the same timeframe, enhancing overall throughput without proportional increases in operational expenditure. The use of common, commercially available solvents like n-butanol ensures supply chain resilience, mitigating the risk of delays associated with specialty chemical shortages.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of catalyst costs and the associated neutralization and disposal fees. Traditional methods often require stoichiometric or near-stoichiometric amounts of Lewis acids, which are not only pricey but also generate corrosive waste streams that demand expensive treatment infrastructure. By shifting to a thermal, catalyst-free regime, the variable cost per kilogram of the final dye is significantly reduced. Additionally, the simplified workup involving basic washing and filtration consumes fewer utilities and labor hours compared to complex chromatographic purifications or multi-stage extractions, leading to substantial operational savings that improve the overall margin profile of the product line.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethyl cyanoacetate, salicylaldehyde derivatives, and standard alcohols creates a robust supply chain foundation. Unlike specialized catalysts that may have limited suppliers or long lead times, these raw materials are produced globally in vast quantities, ensuring consistent availability. This diversification of sourcing options protects the manufacturing schedule from disruptions caused by single-source dependencies. Moreover, the mild reaction conditions reduce the wear and tear on reactor equipment, decreasing maintenance downtime and ensuring a more predictable production calendar. This reliability is crucial for meeting the just-in-time delivery expectations of downstream clients in the fast-paced electronics and display industries.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of exothermic hazards typically associated with strong acid additions. The reflux operation is thermally manageable and easily controlled in large-scale reactors, facilitating a smooth transition from pilot runs to full-scale production. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly strict global regulations on industrial emissions and effluent discharge. The process generates primarily organic waste that is easier to treat or incinerate compared to heavy metal-laden sludge, simplifying compliance reporting and reducing the environmental footprint of the manufacturing facility, which is a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on yield expectations, scalability, and purity standards. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the practical advantages of the catalyst-free approach while acknowledging the specific operational parameters required for success.
Q: What are the primary advantages of this catalyst-free synthesis method?
A: The primary advantage is the elimination of expensive and hazardous catalysts like TiCl4 or benzoic acid, which simplifies the purification process and reduces heavy metal contamination risks in the final electronic grade material.
Q: Can this process be scaled for industrial production of laser dyes?
A: Yes, the process utilizes standard reflux conditions in common solvents like n-butanol and involves simple filtration workups, making it highly suitable for large-scale commercial manufacturing without complex equipment requirements.
Q: What is the typical yield range for these coumarin derivatives?
A: According to the patent data, the general yield for this synthetic route ranges from 60% to 80%, depending on the specific substituents on the aminophenol ring and the solvent choice.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Fluorescent Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for advanced electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to market reality is seamless. We are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of coumarin fluorescent dye meets the exacting standards required for OLED and laser applications. Our commitment to technical excellence allows us to optimize processes like the one described in CN101368004B, delivering high-quality intermediates that empower your downstream innovation.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing capabilities can enhance your product competitiveness. Let us help you secure a reliable supply of high-performance fluorescent materials that drive the next generation of optoelectronic devices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
