Scalable Organocatalytic Production of High-Purity Huperzine A Key Intermediates for Global Pharma Supply Chains
Scalable Organocatalytic Production of High-Purity Huperzine A Key Intermediates for Global Pharma Supply Chains
The global demand for effective treatments for Alzheimer's disease continues to drive innovation in the synthesis of neuroprotective agents, particularly Huperzine A. A pivotal development in this field is documented in Chinese Patent CN101935302A, which discloses a highly efficient asymmetric synthesis method for key huperzine intermediates. This technology leverages small-molecule organocatalysis to construct the complex bridged ring system found in (-)-(5S,9R)-9,10-dihydro-2-methoxy-7-methyl-11-oxo-5,9-methylenecyclooctane[b]pyridine-5(6H)-carboxylate. Unlike traditional routes that rely on stoichiometric chiral auxiliaries or precious metal complexes, this approach utilizes a catalytic asymmetric Michael/Aldol tandem reaction. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this metal-free pathway is critical for securing a sustainable and cost-effective supply chain for neurological API manufacturing.
The structural complexity of the target molecule, characterized by its fused cyclooctane and pyridine rings with specific stereochemistry at the 5S and 9R positions, presents significant synthetic challenges. The patent outlines a strategy that not only addresses these stereochemical demands but also streamlines the overall process flow. By employing readily available starting materials such as methyl 5,6,7,8-tetrahydro-2-methyl-6-oxo-5-quinolinecarboxylate and substituted acroleins, the method achieves high yields and optical purity. This represents a substantial advancement in cost reduction in API manufacturing, as it bypasses the need for costly resolution steps often associated with racemic syntheses. The ability to produce this intermediate with high enantiomeric excess directly impacts the therapeutic efficacy of the final drug product, making this synthetic route a cornerstone for modern pharmaceutical production strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of huperzine intermediates has been plagued by inefficiencies related to stereocontrol and economic viability. Prior art, such as the methods described in J. Am. Chem. Soc. 1989, often relied on the formation of racemic mixtures followed by resolution, which inherently limits the maximum theoretical yield to 50% and generates significant waste. Furthermore, attempts to introduce asymmetry earlier in the sequence frequently involved the use of expensive transition metal catalysts, specifically palladium complexes paired with chiral phosphorus ligands, as noted in Tetrahedron: Asymmetry, 2001. These metal-based systems pose dual challenges: the high cost of the catalysts themselves and the stringent regulatory requirements for removing trace heavy metals from the final active pharmaceutical ingredient. For a commercial scale-up of complex pharmaceutical intermediates, the presence of palladium necessitates additional purification steps, such as scavenging or extensive chromatography, which increases both the lead time and the operational expenditure. Additionally, the sensitivity of some metal catalysts to air and moisture can complicate reactor operations, requiring specialized equipment and inert atmosphere handling that further escalates production costs.
The Novel Approach
The methodology presented in CN101935302A offers a transformative solution by shifting the paradigm from transition metal catalysis to organocatalysis. This novel approach utilizes chiral small molecules, such as prolinol derivatives, cinchona alkaloids, or thiourea-based catalysts, to induce asymmetry during the bond-forming events. The core transformation involves a tandem Michael/Aldol reaction that efficiently constructs the requisite carbon-carbon bonds and stereocenters in a single operational sequence. 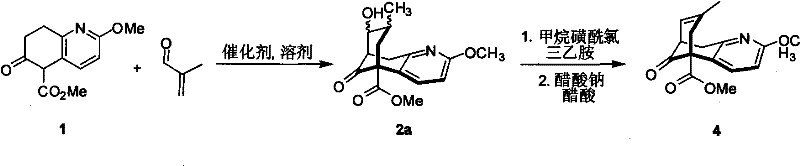 As illustrated in the reaction scheme, the process begins with the condensation of the quinoline derivative with a 2-substituted acrolein. This step is catalyzed by the chiral organic molecule in the presence of mild additives like sodium acetate or trifluoroacetic acid. The subsequent dehydration and cyclization steps proceed under mild conditions, avoiding the harsh reagents often required in metal-mediated processes. This shift not only enhances the safety profile of the manufacturing process but also significantly simplifies the downstream processing. The elimination of heavy metals means that the purification workflow is drastically simplified, leading to a more robust and economically attractive process for producing high-purity pharmaceutical intermediates at scale.
As illustrated in the reaction scheme, the process begins with the condensation of the quinoline derivative with a 2-substituted acrolein. This step is catalyzed by the chiral organic molecule in the presence of mild additives like sodium acetate or trifluoroacetic acid. The subsequent dehydration and cyclization steps proceed under mild conditions, avoiding the harsh reagents often required in metal-mediated processes. This shift not only enhances the safety profile of the manufacturing process but also significantly simplifies the downstream processing. The elimination of heavy metals means that the purification workflow is drastically simplified, leading to a more robust and economically attractive process for producing high-purity pharmaceutical intermediates at scale.
Mechanistic Insights into Organocatalytic Asymmetric Michael/Aldol Tandem Reaction
The success of this synthesis hinges on the precise stereochemical control exerted by the organocatalyst during the initial C-C bond formation. The mechanism typically involves the activation of the nucleophile or electrophile through hydrogen bonding or iminium/enamine catalysis, depending on the specific catalyst class employed. For instance, prolinol-derived catalysts (Type a in the patent) likely operate via an enamine mechanism, where the catalyst forms a transient covalent bond with the carbonyl substrate, lowering the LUMO energy and directing the attack of the electrophile from a specific face. 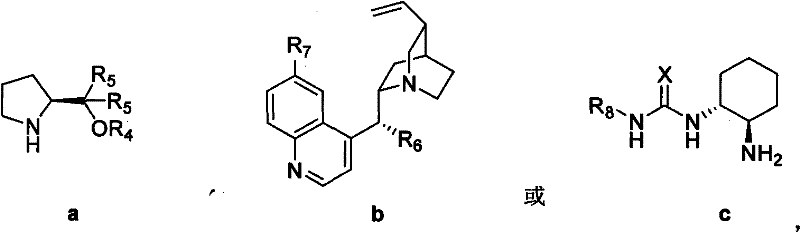 Conversely, thiourea-based catalysts (Type c) function primarily through dual hydrogen-bonding activation, stabilizing the transition state and organizing the reactants in a chiral environment. The patent details three distinct classes of catalysts (a, b, and c), each offering tunable steric and electronic properties to optimize the enantioselectivity for different substrates. The choice of additive, such as acetic acid or benzoic acid, plays a crucial role in proton transfer steps and stabilizing the zwitterionic intermediates formed during the tandem sequence. Understanding these mechanistic subtleties allows process chemists to fine-tune reaction conditions, such as solvent polarity and temperature, to maximize the diastereomeric and enantiomeric ratios. This level of control is essential for ensuring that the impurity profile of the intermediate remains within the strict specifications required for GMP manufacturing.
Conversely, thiourea-based catalysts (Type c) function primarily through dual hydrogen-bonding activation, stabilizing the transition state and organizing the reactants in a chiral environment. The patent details three distinct classes of catalysts (a, b, and c), each offering tunable steric and electronic properties to optimize the enantioselectivity for different substrates. The choice of additive, such as acetic acid or benzoic acid, plays a crucial role in proton transfer steps and stabilizing the zwitterionic intermediates formed during the tandem sequence. Understanding these mechanistic subtleties allows process chemists to fine-tune reaction conditions, such as solvent polarity and temperature, to maximize the diastereomeric and enantiomeric ratios. This level of control is essential for ensuring that the impurity profile of the intermediate remains within the strict specifications required for GMP manufacturing.
Impurity control is another critical aspect where this organocatalytic route excels. In metal-catalyzed reactions, side products often arise from beta-hydride elimination or competing coordination modes of the metal center. In contrast, organocatalytic pathways tend to generate fewer metallic impurities and offer cleaner reaction profiles. The patent reports that the reaction can be conducted in a variety of common organic solvents, including THF, dichloromethane, and toluene, providing flexibility in process optimization. The subsequent conversion of the beta-keto ester intermediate to the final unsaturated product involves a mesylation followed by elimination. This two-step sequence is highly selective, minimizing the formation of regioisomers or over-elimination byproducts. The ability to achieve enantiomeric excess (ee) values up to 89% directly from the catalytic step demonstrates the high fidelity of the stereochemical induction. For quality assurance teams, this implies a more predictable and controllable process, reducing the risk of batch failures due to out-of-specification chirality.
How to Synthesize (-)-(5S,9R)-9,10-dihydro-2-methoxy-7-methyl-11-oxo-5,9-methylenecyclooctane[b]pyridine-5(6H)-carboxylate Efficiently
The practical execution of this synthesis involves a carefully orchestrated sequence of reactions that balance reactivity with selectivity. The initial tandem reaction requires a molar ratio of the quinoline starting material to the acrolein derivative ranging from 1:2 to 1:12, with the catalyst loading typically between 10% to 60% relative to the substrate. The reaction is generally performed at temperatures ranging from 0°C to reflux, with reaction times extending from 3 to 5 days to ensure complete conversion. Following the isolation of the beta-keto ester intermediate, the subsequent functionalization steps involve standard organic transformations that are amenable to scale-up.
- Perform an asymmetric Michael/Aldol tandem reaction between methyl 5,6,7,8-tetrahydro-2-methyl-6-oxo-5-quinolinecarboxylate and 2-substituted acrolein using a chiral organocatalyst (prolinol, cinchona, or thiourea derivative) in organic solvent at 0°C to reflux for 3-5 days.
- Isolate the beta-keto ester intermediate (2a/2b) and optionally cyclize using a base if the open-chain aldehyde form (2b) is obtained, ensuring high diastereoselectivity.
- Convert the hydroxyl group of the intermediate to a mesylate using methanesulfonyl chloride and triethylamine, followed by elimination in acetic acid/sodium acetate to yield the final unsaturated key intermediate with up to 89% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology offers compelling strategic advantages beyond mere technical feasibility. The most significant benefit lies in the drastic simplification of the supply chain for raw materials. By replacing expensive palladium catalysts and specialized chiral ligands with commercially available organocatalysts, the volatility associated with precious metal pricing is completely eliminated. This stability in raw material costs translates directly into more predictable budgeting and long-term pricing contracts for the final intermediate. Furthermore, the absence of heavy metals removes the bottleneck of metal scavenging and testing, which often delays batch release in traditional processes. This streamlined workflow enhances the overall throughput of the manufacturing facility, allowing for faster response times to market demands.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. Traditional methods relying on palladium incur high costs not only for the catalyst purchase but also for the recovery and disposal of metal-containing waste streams. By utilizing organocatalysts, which are generally less expensive and easier to handle, the direct material cost of the synthesis is significantly lowered. Additionally, the simplified workup procedures, which often involve basic aqueous washes and standard chromatography rather than specialized metal removal resins, reduce the consumption of auxiliary materials and solvents. This holistic reduction in operational complexity leads to substantial cost savings per kilogram of produced intermediate, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available reagents. Organocatalysts are typically stable solids that do not require the stringent storage conditions (e.g., inert atmosphere, low temperature) often mandated for sensitive metal complexes. This ease of handling reduces the risk of supply disruptions caused by reagent degradation or logistical challenges. Moreover, the versatility of the solvent system, which accommodates a wide range of organic solvents from alcohols to hydrocarbons, provides flexibility in sourcing. If a specific solvent faces supply constraints, the process can often be adapted to use an alternative without compromising yield or purity. This adaptability ensures a continuous and reliable supply of the critical huperzine intermediate, safeguarding the production schedules of downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this green chemistry approach aligns perfectly with modern sustainability goals. The elimination of toxic heavy metals reduces the environmental footprint of the manufacturing process, simplifying waste treatment and disposal compliance. The patent demonstrates that the reaction can be successfully scaled to gram levels with maintained efficiency, indicating a clear path toward kilogram and tonnage production. The high atom economy of the tandem reaction, combined with the potential for catalyst recycling in some organocatalytic systems, further enhances the sustainability profile. For companies aiming to meet rigorous environmental standards and reduce their carbon footprint, adopting this synthetic route represents a proactive step towards greener pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What represents the primary advantage of this organocatalytic route over traditional methods?
A: The primary advantage is the elimination of expensive palladium catalysts and chiral phosphorus ligands required in previous methods, significantly reducing raw material costs while simplifying the removal of heavy metal impurities.
Q: What level of optical purity can be achieved with this synthesis method?
A: The patent data indicates that the process can achieve enantiomeric excess (ee) values up to 89%, with specific catalyst systems demonstrating ranges between 65-88% depending on the substrate and conditions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes relatively cheap organocatalysts and standard organic solvents, and the patent explicitly demonstrates successful gram-level preparation, indicating strong potential for scale-up to kilogram or tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-(5S,9R)-9,10-dihydro-2-methoxy-7-methyl-11-oxo-5,9-methylenecyclooctane[b]pyridine-5(6H)-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving neurological therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of the huperzine intermediate described in CN101935302A or optimization of an existing route, our CDMO capabilities are designed to accelerate your project timelines while maintaining the highest standards of quality and compliance.
We invite you to engage with our technical procurement team to discuss how our advanced organocatalytic platforms can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique project needs. Let us collaborate to bring safer, more effective, and affordable treatments for Alzheimer's disease to patients worldwide through superior chemical manufacturing excellence.
