Optimizing Raloxifene Intermediate Production: A Technical Analysis of Enhanced Synthetic Routes
Optimizing Raloxifene Intermediate Production: A Technical Analysis of Enhanced Synthetic Routes
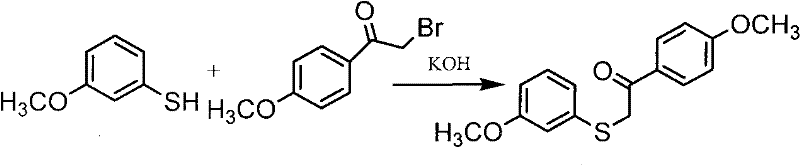
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical active pharmaceutical ingredient (API) precursors, particularly for high-value therapeutics like Raloxifene, a prominent selective estrogen receptor modulator. Patent CN101812002A introduces a significant technological advancement in the synthesis of 4-methoxy-alpha-[(3-methoxyphenyl)thio]acetophenone, a pivotal intermediate in the Raloxifene value chain. This proprietary methodology addresses longstanding inefficiencies in traditional condensation reactions by leveraging a refined ethanol-based solvent system and optimized thermal parameters. By shifting away from cryogenic conditions and heterogeneous reaction mixtures, this innovation offers a streamlined approach that enhances both chemical yield and operational safety. For R&D directors and process chemists, understanding the nuances of this improved protocol is essential for evaluating potential technology transfers or licensing opportunities that could redefine production standards for osteoporosis treatment intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key thioether intermediate has been plagued by operational complexities that hinder scalability and cost-efficiency. Prior art, including methodologies described in US Patents 6472531 and 5523416, relies heavily on biphasic systems involving water and ethanol, necessitating rigorous temperature control via ice-salt baths to manage exothermicity during the addition of solid alpha-bromo-4-methoxyacetophenone. This heterogeneous environment often leads to uneven mixing and incomplete dissolution of the solid halide, resulting in inconsistent reaction kinetics and the formation of stubborn impurities. Furthermore, the downstream processing associated with these legacy routes is notoriously resource-intensive, requiring multiple solvent exchanges, ethyl acetate extractions, and methanol refinements to achieve acceptable purity levels. These additional unit operations not only inflate manufacturing costs through increased solvent consumption but also extend cycle times and generate substantial volumes of hazardous waste, creating a significant burden on environmental compliance and supply chain logistics.
The Novel Approach
In stark contrast, the novel process delineated in the patent data utilizes a homogeneous reaction system that fundamentally alters the thermodynamic and kinetic landscape of the synthesis. By pre-dissolving the alpha-bromo ketone in ethanol and maintaining the reaction mixture at a mild temperature range of 35-40°C during reagent addition, the method ensures complete solubility and uniform molecular interaction from the outset. This elimination of solid-state diffusion barriers allows the nucleophilic substitution to proceed with exceptional thoroughness, effectively driving the reaction to completion without the need for aggressive cooling or extended reaction times. The resultant workflow simplifies isolation to a straightforward filtration and aqueous wash, bypassing the need for organic extraction solvents entirely. This paradigm shift not only accelerates production throughput but also drastically reduces the facility's solvent footprint, aligning perfectly with modern green chemistry principles and offering a compelling value proposition for cost-conscious procurement strategies.
Mechanistic Insights into Base-Catalyzed Nucleophilic Substitution
The core chemical transformation driving this synthesis is a classic base-catalyzed nucleophilic substitution, specifically an SN2-type displacement where the sulfur atom acts as the nucleophile. In this optimized system, potassium hydroxide serves as the base to deprotonate 3-methoxythiophenol, generating the highly reactive 3-methoxythiophenolate anion in situ. The choice of ethanol as the primary solvent is critical, as it effectively solvates both the organic thiol and the inorganic base, creating a pseudo-homogeneous medium that facilitates rapid ion pairing and collision frequency. Unlike aqueous systems where the nucleophile might be heavily solvated and less reactive, the ethanolic environment enhances the nucleophilicity of the thiolate species, allowing it to efficiently attack the electrophilic alpha-carbon of the bromoacetophenone derivative. This mechanistic efficiency is further bolstered by the controlled thermal input, which provides sufficient activation energy to overcome the transition state barrier without promoting thermal degradation or side reactions such as elimination.
From an impurity control perspective, the homogeneity of the reaction mixture plays a decisive role in defining the final quality profile. In heterogeneous systems, localized concentrations of base or unreacted halide can lead to over-alkylation or hydrolysis byproducts, which are difficult to separate due to similar polarity characteristics. However, the uniform distribution of reagents in this novel ethanol-based protocol ensures that the stoichiometry is maintained consistently throughout the reactor volume, minimizing the formation of bis-alkylated sulfides or hydrolyzed ketone byproducts. The high purity reported, often exceeding 99% by HPLC analysis directly after filtration, suggests that the reaction selectivity is exceptionally high. This implies that the activation energy for the desired substitution pathway is significantly lower than that of competing degradation pathways under these specific conditions, providing R&D teams with a robust platform for generating high-quality intermediates suitable for subsequent cyclization steps in the Raloxifene synthesis without requiring extensive chromatographic purification.
How to Synthesize 4-Methoxy-alpha-[(3-methoxyphenyl)thio]acetophenone Efficiently
Implementing this synthesis requires precise adherence to the thermal and concentration parameters established in the patent to maximize yield and purity. The process begins with the preparation of distinct solution streams to ensure immediate homogeneity upon mixing, avoiding the pitfalls of solid addition. Operators must carefully monitor the addition rate of the thiolate solution to the heated ketone stream to maintain the exotherm within the safe 35-40°C window, which is crucial for preventing solvent boil-off and ensuring consistent crystal formation upon cooling. The following guide outlines the standardized operational sequence derived from the patent examples, serving as a foundational reference for pilot plant scale-up and technology transfer activities.
- Dissolve alpha-bromo-4-methoxyacetophenone in ethanol and heat the mixture to a controlled temperature range of 35-40°C to ensure complete solubility and reaction readiness.
- Prepare a mixed solution of potassium hydroxide and 3-methoxythiophenol in ethanol, then add this mixture dropwise to the heated ketone solution under stirring.
- Maintain the reaction at room temperature for approximately 12 hours, followed by filtration, water washing, and drying to obtain the final high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this refined synthetic route translates directly into tangible operational efficiencies and risk mitigation. The elimination of cryogenic cooling requirements removes a significant energy load from the manufacturing process, reducing utility costs and freeing up specialized low-temperature reactor capacity for other campaigns. Furthermore, the simplification of the workup procedure—replacing multi-step solvent extractions with a single filtration and water wash—drastically cuts down on solvent procurement volumes and waste disposal fees. This reduction in material intensity not only lowers the variable cost per kilogram of the intermediate but also shortens the overall batch cycle time, allowing for increased asset utilization and faster response to market demand fluctuations. The robustness of the process also implies a lower risk of batch failure due to operational deviations, ensuring a more reliable and continuous supply of this critical pharmaceutical building block.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing train. By obviating the need for ethyl acetate extraction and methanol recrystallization, the manufacturer saves substantially on solvent purchase, recovery, and loss costs. Additionally, the high intrinsic purity of the crude product means that yield losses associated with purification steps are virtually eliminated, leading to a higher overall mass balance and better utilization of expensive starting materials like the bromo-ketone precursor. This lean manufacturing approach significantly improves the gross margin profile of the intermediate, making it a highly attractive option for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of commodity chemicals and standard operating conditions. Ethanol and potassium hydroxide are globally available bulk commodities with stable pricing and supply, reducing the risk of raw material shortages that can plague specialty reagent-dependent processes. The ability to run the reaction at near-ambient temperatures without specialized cooling infrastructure means that production can be easily transferred between different manufacturing sites or scaled up in existing multipurpose facilities without major capital expenditure. This flexibility ensures that supply continuity can be maintained even during periods of high demand or logistical disruptions, providing a strategic advantage for long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a markedly superior profile compared to traditional methods. The reduction in solvent usage directly correlates to a lower volatile organic compound (VOC) emission footprint, facilitating easier compliance with increasingly stringent environmental regulations. The aqueous waste stream generated from the washing step is significantly less complex to treat than mixed organic waste streams containing halogenated or aromatic solvents. Moreover, the high atom economy and yield of the reaction minimize the generation of solid chemical waste, aligning the manufacturing process with sustainability goals and reducing the liability associated with hazardous waste disposal, which is a critical factor for modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthetic methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: What are the primary advantages of the ethanol-based synthesis method over traditional protocols?
A: The ethanol-based method eliminates the need for cryogenic cooling (ice-salt baths) and complex solvent extraction steps. This results in a homogeneous reaction system that ensures higher conversion rates, simplifies post-reaction processing to mere filtration and washing, and significantly reduces solvent consumption and waste generation.
Q: How does this process impact the purity profile of the Raloxifene intermediate?
A: By facilitating a more thorough and homogeneous reaction environment, this process minimizes the formation of side-products and unreacted starting materials. The resulting crude product typically exhibits HPLC purity levels exceeding 99% after simple aqueous washing, removing the necessity for energy-intensive recrystallization or chromatographic purification steps.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to its operational simplicity. The absence of low-temperature requirements and the use of common, recoverable solvents like ethanol make it economically viable for ton-scale production, offering substantial improvements in throughput and environmental compliance compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxy-alpha-[(3-methoxyphenyl)thio]acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthetic routes like the one described in CN101812002A are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 4-methoxy-alpha-[(3-methoxyphenyl)thio]acetophenone meets the exacting standards required for downstream API synthesis. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth, high-yielding final drug substance manufacturing.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis can drive value in your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient process. We encourage you to contact us to obtain specific COA data from our recent pilot batches and to receive comprehensive route feasibility assessments tailored to your specific volume requirements. Let us collaborate to enhance the efficiency and reliability of your Raloxifene supply chain today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
