Industrial Scale Synthesis of 2-Aminomethyl-4-Cyano-Thiazole for Advanced Thrombin Inhibitors
The pharmaceutical industry constantly seeks robust, scalable pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for serine protease inhibitors. Patent CN1364164A introduces a groundbreaking methodology for the preparation of 2-aminomethyl-4-cyano-thiazole, a pivotal intermediate in the synthesis of potent thrombin inhibitors. This novel approach fundamentally shifts the paradigm from hazardous, low-yield laboratory protocols to a streamlined, industrially viable process. By leveraging the reaction between protected aminonitriles and cysteine esters, the inventors have unlocked a route that offers quantitative conversions in key steps, thereby addressing long-standing challenges in impurity control and process safety. For R&D directors and procurement specialists alike, this technology represents a significant leap forward in the reliable supply of high-purity pharmaceutical intermediates, ensuring that the downstream production of life-saving anticoagulants remains uninterrupted and economically feasible.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the thiazole ring substituted at the 4-position with electron-withdrawing groups has been fraught with significant technical and economic hurdles. Conventional literature describes methods relying on the reaction of thioamides with bromopyruvate derivatives, a pathway that often necessitates the use of Lawesson's reagent or the direct handling of hydrogen sulfide gas. From a process safety perspective, the utilization of H2S is deeply problematic for large-scale manufacturing due to its extreme toxicity and the stringent engineering controls required to prevent environmental release. Furthermore, the economic viability of using Lawesson's reagent diminishes rapidly upon scale-up, as the reagent is costly and generates substantial phosphorus-containing waste streams that complicate downstream purification. Alternative oxidative methods using manganese dioxide have shown only moderate yields, while other protocols involving bromochloroform and DBU were previously limited to gram-scale experiments, failing to demonstrate the robustness required for multi-kilogram or ton-scale production of active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in CN1364164A utilizes a convergent strategy that begins with the condensation of a protected aminonitrile and a cysteine ester. This initial cyclization step proceeds with remarkable efficiency, achieving yields exceeding 97% under mild conditions. The subsequent oxidative aromatization transforms the saturated thiazolidine ring into the desired aromatic thiazole system with near-quantitative conversion, utilizing accessible oxidants rather than exotic or hazardous reagents. This methodology not only eliminates the need for toxic gas handling but also simplifies the purification landscape, as crude products from early steps can often be telescoped directly into subsequent reactions without intermediate isolation. The ability to perform ammonolysis at high concentrations further enhances the space-time yield, making this route exceptionally attractive for commercial manufacturing where reactor throughput and operational simplicity are paramount drivers of profitability.
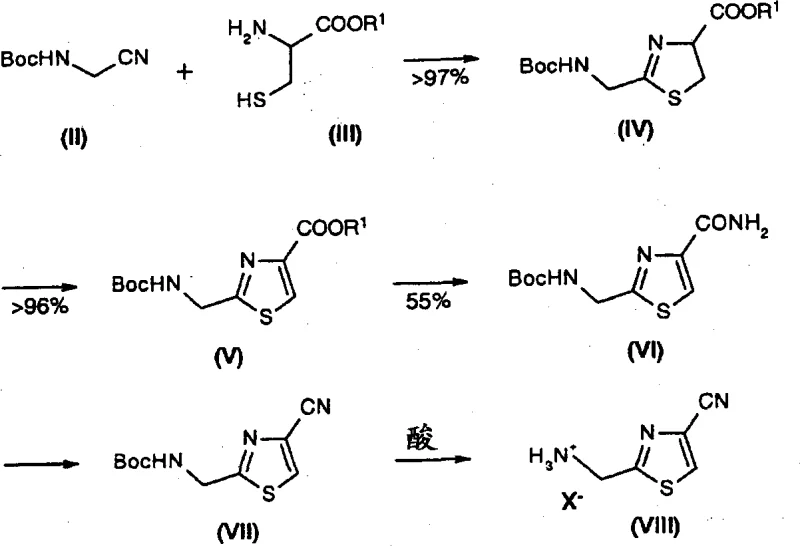
Mechanistic Insights into Oxidative Aromatization and Cyclization
The core chemical innovation lies in the efficient formation of the thiazolidine intermediate followed by its controlled oxidation. Mechanistically, the nucleophilic thiol group of the cysteine ester attacks the electrophilic carbon of the aminonitrile, facilitated by a base such as triethylamine in a polar protic or aprotic solvent system. This cyclization is thermodynamically favorable and proceeds to completion, effectively locking the stereochemistry and functionality required for the final drug molecule. Following this, the oxidative step employs a combination of DBU and bromotrichloromethane to effect dehydrogenation. This specific reagent combination is critical; it provides the necessary oxidation potential to aromatize the ring without degrading the sensitive nitrile or ester functionalities present on the scaffold. The reaction conditions are carefully tuned, typically operating between -5°C and 0°C during oxidant addition to manage exotherms, before warming to room temperature to ensure full conversion. This precise control over reaction parameters minimizes the formation of over-oxidized byproducts or ring-opened impurities, ensuring a clean impurity profile that simplifies the burden on analytical quality control teams.
Furthermore, the transformation of the thiazole-4-carboxylate into the corresponding amide and subsequently the nitrile is achieved through a sequence of ammonolysis and dehydration. The ammonolysis step is surprisingly robust, functioning effectively even with crude ester inputs at high concentrations, which drastically reduces solvent consumption and waste generation. The use of alcoholic ammonia solutions allows for higher space-time yields compared to aqueous systems, which would require excessive volumes to achieve similar conversion rates. Finally, the dehydration using trifluoroacetic anhydride followed by BOC deprotection is executed with high fidelity, yielding the final hydrochloride salt with an HPLC purity of 99.4 area percent. This level of purity is achieved without recourse to chromatographic purification, relying instead on crystallization and phase separation techniques that are inherently scalable and cost-effective for industrial operations.
How to Synthesize 2-Aminomethyl-4-Cyano-Thiazole Efficiently
The synthesis of this critical intermediate is designed for operational simplicity and high throughput, minimizing the need for complex isolation steps between transformations. The process begins with the protection of aminoacetonitrile, followed by condensation with L-cysteine ethyl ester hydrochloride in the presence of a tertiary amine base. The resulting thiazolidine is then subjected to oxidative conditions to establish the aromatic core. Subsequent functional group manipulations convert the ester moiety into the final nitrile group while simultaneously removing the protecting group to reveal the free amine. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- React protected aminonitrile with cysteine ester in an inert solvent with a base to form the thiazolidine intermediate with quantitative conversion.
- Perform oxidative aromatization of the thiazolidine ring using DBU and bromotrichloromethane to generate the thiazole-4-carboxylate.
- Convert the ester to the amide via ammonolysis, followed by dehydration and deprotection to yield the final 2-aminomethyl-4-cyano-thiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material supply chain. By avoiding the need for specialized, hazardous reagents like hydrogen sulfide or Lawesson's reagent, manufacturers can source starting materials from a broader, more competitive vendor base, thereby reducing input costs and mitigating supply risk. The elimination of toxic gas handling infrastructure also translates to significant capital expenditure savings, as facilities do not require specialized scrubbing systems or high-level containment protocols. Moreover, the high space-time yields achieved through concentrated reaction conditions mean that existing reactor assets can produce more product in less time, effectively increasing capacity without the need for new equipment investments. This efficiency directly impacts the cost of goods sold, allowing for more competitive pricing in the global market for thrombin inhibitor intermediates.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of expensive and hazardous reagents. Traditional routes often incur high costs associated with the safe disposal of phosphorus waste from Lawesson's reagent or the engineering controls for H2S. By switching to cysteine esters and aminonitriles, the material costs are significantly lowered, and the waste treatment burden is reduced. Additionally, the ability to telescope steps without isolating intermediates reduces solvent usage and labor hours, leading to substantial overall cost savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commercially available, stable starting materials. Cysteine esters and protected aminonitriles are commodity chemicals with established supply chains, unlike specialized thioamides which may have limited suppliers. This diversity in sourcing options ensures that production schedules are not disrupted by single-source bottlenecks. Furthermore, the robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent flow of high-quality intermediate to downstream API manufacturing sites.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods. The avoidance of heavy metal oxidants like manganese dioxide and toxic gases aligns with modern green chemistry principles and strict environmental regulations. The process generates less hazardous waste, simplifying compliance reporting and disposal logistics. From a scalability perspective, the exothermic profiles are manageable, and the work-up procedures involve standard phase separations and crystallizations, making the transition from pilot plant to multi-ton commercial production seamless and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does this new synthesis method improve safety compared to traditional thiazole production?
A: Traditional methods often rely on hazardous hydrogen sulfide gas or expensive Lawesson's reagent. This patented route utilizes stable cysteine esters and aminonitriles, eliminating the need for toxic gas handling infrastructure and significantly reducing operational risks in large-scale manufacturing.
Q: What yields can be expected from this industrial process?
A: The process demonstrates exceptional efficiency, with the initial cyclization step achieving yields greater than 97%. The subsequent oxidation and functionalization steps maintain high conversion rates, resulting in an overall process that is highly suitable for cost-effective commercial production.
Q: Is this method scalable for multi-ton production of API intermediates?
A: Yes, the patent explicitly addresses industrial scalability. By avoiding difficult-to-handle reagents and utilizing standard solvents like methanol and dichloromethane, the process allows for high space-time yields and simplified work-up procedures, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminomethyl-4-Cyano-Thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-aminomethyl-4-cyano-thiazole meets the exacting standards required for anticoagulant drug synthesis. Our facility is equipped to handle the specific solvent systems and reaction conditions outlined in this technology, delivering a product that is ready for immediate integration into your API manufacturing workflow.
We invite you to engage with our technical procurement team to discuss how this optimized route can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us partner with you to secure a reliable, cost-effective supply chain for your critical thrombin inhibitor programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
