Advanced Catalytic Synthesis of Avatrombopag Impurity for Commercial API Manufacturing
The pharmaceutical industry's relentless pursuit of therapeutic efficacy demands rigorous control over impurity profiles, particularly for complex small molecules like Avatrombopag. Patent CN113185510B introduces a transformative methodology for synthesizing a critical N-oxide impurity, addressing longstanding challenges in selectivity and yield that have plagued conventional oxidation protocols. This technical breakthrough leverages manganese dioxide as a heterogeneous catalyst to facilitate the conversion of the parent compound into its N-oxide derivative with exceptional precision. By shifting away from harsh, uncontrolled oxidation environments, this approach ensures the generation of high-purity reference standards essential for regulatory compliance and quality control. The structural integrity of the target molecule, characterized by its complex thiazole and piperazine moieties, is preserved while achieving the specific oxidative modification required for comprehensive safety assessments. 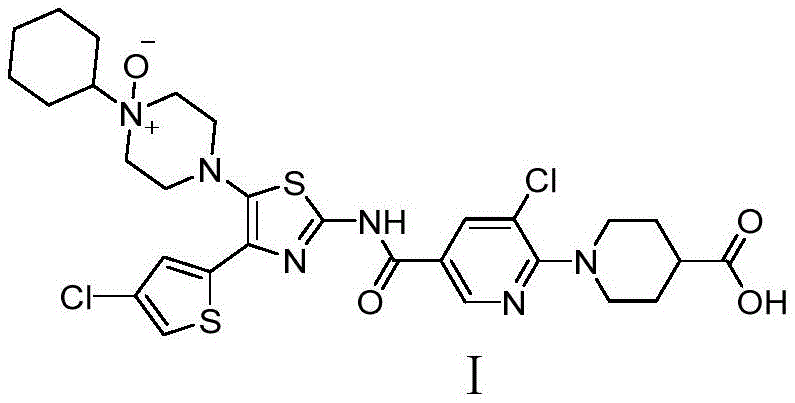
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-oxide impurities in thrombopoietin receptor agonists has relied on stoichiometric oxidants without catalytic enhancement, leading to inefficient process dynamics. Traditional methods often necessitate reflux conditions in organic solvents, which not only consume substantial energy but also increase the risk of thermal degradation of sensitive functional groups within the molecular scaffold. Without a selective catalyst, the oxidant must be added slowly and incrementally to prevent runaway reactions, drastically extending processing time and complicating operational workflows. Furthermore, the lack of catalytic direction frequently results in over-oxidation or non-specific side reactions, generating a complex mixture of byproducts that are difficult to separate. These inefficiencies culminate in suboptimal yields, often remaining below 60%, and necessitate resource-intensive purification techniques such as silica gel chromatography which are impractical for large-scale manufacturing. 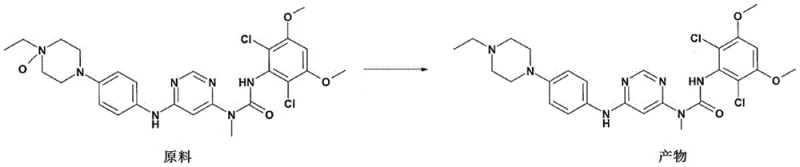
The Novel Approach
In stark contrast, the novel methodology detailed in the patent utilizes manganese dioxide to orchestrate a highly selective oxidation under significantly milder conditions. This catalytic system allows for the one-time addition of the oxidant, eliminating the need for tedious dropwise control and reducing the potential for human error during operation. The reaction proceeds efficiently at temperatures between 40°C and 60°C, avoiding the thermal stress associated with reflux and preserving the stability of the chloro-thienyl and pyridine components. By optimizing the molar ratios of the catalyst and oxidant, the process minimizes waste generation and streamlines the downstream workup to a simple pH adjustment and crystallization. This paradigm shift from brute-force oxidation to catalytic precision results in yields exceeding 90%, providing a robust and reliable pathway for producing high-quality impurity standards.
Mechanistic Insights into MnO2-Catalyzed N-Oxidation
The core of this technological advancement lies in the unique interaction between the manganese dioxide surface and the tertiary amine nitrogen within the piperazine ring. Manganese dioxide acts as a Lewis acid catalyst, activating the m-chloroperoxybenzoic acid oxidant and facilitating the transfer of oxygen to the nucleophilic nitrogen center with high regioselectivity. This mechanism prevents the oxidation of other susceptible sites, such as the thiazole sulfur or the pyridine nitrogen, which are common pitfalls in non-catalyzed systems. The heterogeneous nature of the catalyst also aids in the ease of separation, as the solid manganese species can be removed via filtration prior to the final crystallization step. Such mechanistic control ensures that the impurity profile remains clean, with the target N-oxide being the predominant species formed during the reaction cycle. 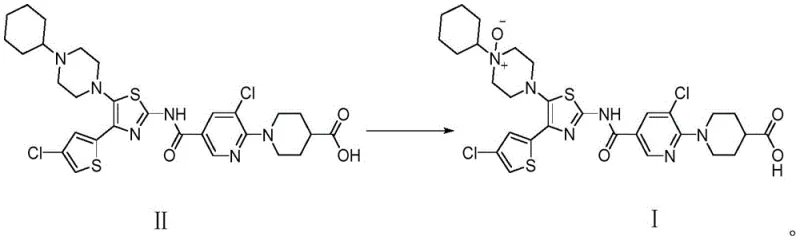
Furthermore, the reaction environment is meticulously tuned to maintain a basic pH, typically around 12, using sodium hydroxide to ensure the solubility of the starting material and the stability of the intermediate species. This alkaline condition is crucial for deprotonating intermediate states and driving the equilibrium towards the desired N-oxide product. The solvent system, comprising a mixture of ethanol and water, provides an optimal balance of polarity to dissolve both the organic substrate and the inorganic base while maintaining the suspension of the catalyst. Careful control of the reaction time, typically between 2 to 6 hours, ensures complete conversion without allowing sufficient time for secondary decomposition pathways to emerge. This comprehensive understanding of the reaction kinetics allows for precise scaling from laboratory benchtop to pilot plant operations without loss of fidelity.
How to Synthesize Avatrombopag Impurity Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize the benefits of the catalytic system. The process begins with the dissolution of the precursor in the optimized solvent blend, followed by the precise addition of reagents to initiate the catalytic cycle. Detailed standard operating procedures regarding temperature ramping and agitation speeds are critical to maintaining the suspension of the manganese dioxide and ensuring uniform heat transfer throughout the reaction mass. For a complete breakdown of the standardized synthesis steps, please refer to the guide below.
- Dissolve the precursor compound II in a mixed solvent system of ethanol and water with sodium hydroxide to establish basic conditions.
- Introduce manganese dioxide catalyst and m-chloroperoxybenzoic acid oxidant at controlled molar ratios under mild heating.
- Maintain reaction temperature between 40-60°C, followed by pH adjustment and crystallization to isolate the high-purity target impurity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this catalytic innovation offers substantial advantages in terms of cost structure and supply reliability for pharmaceutical intermediates. The elimination of complex purification steps like column chromatography significantly reduces the consumption of silica gel and organic solvents, leading to direct material cost savings and reduced hazardous waste disposal fees. Additionally, the shortened reaction time and simplified workup procedure enhance throughput capacity, allowing manufacturers to respond more agilely to fluctuating market demands for reference standards. The robustness of the process also minimizes the risk of batch failures, ensuring a consistent supply of critical impurities needed for regulatory filings and quality assurance testing.
- Cost Reduction in Manufacturing: The transition to a catalytic process inherently lowers the stoichiometric burden of expensive oxidants and reduces energy consumption by operating at lower temperatures. By avoiding the need for extensive chromatographic purification, the overall processing time is drastically shortened, which translates to lower labor costs and higher equipment utilization rates. The simplicity of the isolation procedure further reduces the requirement for specialized consumables, contributing to a leaner and more cost-effective manufacturing model that supports competitive pricing strategies.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as manganese dioxide and m-chloroperoxybenzoic acid mitigates the risk of raw material shortages that can plague specialized synthetic routes. The process tolerance to minor variations in reaction conditions ensures high batch-to-b一致性 consistency, reducing the likelihood of supply disruptions caused by out-of-specification results. This reliability is paramount for maintaining continuous validation streams and ensuring that drug development timelines are not compromised by the unavailability of critical impurity standards.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup align well with green chemistry principles, reducing the environmental footprint associated with volatile organic compound emissions. The process is designed for scalability, allowing for seamless transition from kilogram to multi-ton production scales without the need for specialized high-pressure or cryogenic equipment. This adaptability ensures that supply can be scaled up rapidly to meet commercial launch requirements while maintaining strict adherence to environmental safety regulations and waste management protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific Avatrombopag impurity. These insights are derived directly from the patented technology and practical manufacturing experience to assist decision-makers in evaluating the feasibility of this route. Understanding these aspects is crucial for integrating this intermediate into your broader quality control and supply chain strategies.
Q: Why is manganese dioxide preferred over traditional oxidants for this impurity synthesis?
A: Manganese dioxide offers superior selectivity and allows for milder reaction conditions compared to uncatalyzed reflux methods, significantly reducing byproduct formation and simplifying post-treatment purification steps.
Q: What is the expected purity profile of the synthesized Avatrombopag impurity?
A: The optimized catalytic process consistently achieves purity levels exceeding 99%, meeting stringent requirements for reference standards and impurity profiling in regulatory submissions.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process eliminates the need for strict oxidant addition control and hazardous reflux conditions, making it highly suitable for safe and efficient commercial scale-up operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avatrombopag Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the successful development and commercialization of novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Avatrombopag impurity meets the highest international standards for analytical reference materials. Our commitment to technical excellence allows us to deliver complex intermediates that support your regulatory submissions and quality assurance programs effectively.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our catalytic manufacturing platform can enhance your operational efficiency and reduce overall procurement costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
