Advanced Fluorination Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The global demand for fluorinated organic molecules continues to surge across the pharmaceutical and agrochemical sectors, driven by the unique metabolic stability and lipophilicity that fluorine atoms impart to bioactive compounds. Patent CN102153430A introduces a transformative methodology for the preparation of fluorinated aromatic organic compounds, specifically targeting the conversion of aldehyde groups into difluoromethyl moieties. This innovation centers on the utilization of 2,2-difluoro-1,3-benzodioxol as a highly efficient fluorinating agent, addressing critical limitations found in legacy technologies. By leveraging this stable liquid reagent, manufacturers can achieve superior control over reaction parameters while mitigating the severe safety hazards associated with gaseous fluorine sources. The technical breakthrough described herein offers a robust pathway for producing high-purity pharmaceutical intermediates, ensuring that supply chains remain resilient against regulatory and operational disruptions. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is essential for optimizing synthetic routes and securing a competitive edge in the fine chemical market.
The implementation of this technology represents a significant paradigm shift in how fluorinated aromatic organic compounds are synthesized on an industrial scale. Traditional methods often rely on hazardous gases or expensive, unstable reagents that complicate process safety and increase operational expenditures. In contrast, the approach detailed in CN102153430A utilizes a reagent that is commercially available and exhibits excellent thermal stability, with a boiling point of 129°C and a flash point of 32°C. This physical profile allows for precise dosing and reaction monitoring without the need for specialized cryogenic or high-pressure containment systems. Furthermore, the versatility of this method extends to a wide range of aromatic aldehydes, accommodating various substituents such as halogens, nitro groups, and alkyl chains. As a reliable agrochemical intermediate supplier or partner in drug development, adopting this chemistry can streamline the production of key building blocks, ultimately reducing lead time for high-purity intermediates and enhancing overall project velocity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fluorination of organic substrates has been plagued by significant technical and safety challenges that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical ingredients. Common fluorinating agents such as sulfur tetrafluoride (SF4) are toxic gases with extremely low boiling points, making them notoriously difficult to handle and requiring elaborate engineering controls to prevent leaks and exposure. Similarly, inorganic fluorides like potassium fluoride often exhibit low reactivity, necessitating harsh conditions that can degrade sensitive functional groups on the aromatic ring. Other reagents, such as diethylaminosulfur trifluoride (DAST), while effective, are expensive and thermally unstable, posing explosion risks during storage and usage. Moreover, many nitrogen-containing fluorinating reagents suffer from poor atom economy, where only half of the fluorine atoms in the reagent molecule are incorporated into the product, leading to substantial waste generation and increased raw material costs. These factors collectively create bottlenecks in manufacturing, driving up the cost reduction in electronic chemical manufacturing and other high-value sectors.
The Novel Approach
The methodology disclosed in patent CN102153430A overcomes these historical barriers by employing 2,2-difluoro-1,3-benzodioxol, a stable liquid that eliminates the risks associated with gaseous fluorine sources. This reagent is neither explosive nor highly volatile, allowing for safer transportation and storage within standard chemical warehouses. The reaction proceeds under relatively mild conditions, typically between 20°C and 160°C, which preserves the integrity of sensitive substrates and minimizes the formation of unwanted byproducts. Crucially, this new process route improves the utilization rate of fluorine, ensuring that a higher proportion of the expensive fluorine atoms end up in the final product rather than in waste streams. By avoiding the use of transition metal catalysts or hazardous gases, the process simplifies downstream purification, thereby reducing the burden on wastewater treatment facilities. This approach aligns perfectly with modern green chemistry principles, offering a sustainable solution for the production of fluorinated aromatic organic compounds that meets stringent environmental compliance standards.
Mechanistic Insights into Acid-Catalyzed Difluoromethylation
The core of this innovative synthesis lies in the activation of the carbonyl group of the aromatic aldehyde by an acid or Lewis acid catalyst, facilitating nucleophilic attack by the fluorinating agent. In the presence of catalysts such as sulfuric acid, boron trifluoride, or methanesulfonic acid, the oxygen atom of the aldehyde group becomes protonated or coordinated, increasing the electrophilicity of the carbonyl carbon. This activation enables the 2,2-difluoro-1,3-benzodioxol to transfer its fluorine atoms effectively, resulting in the conversion of the aldehyde functionality into a difluoromethyl group. The reaction mechanism likely involves the formation of a transient intermediate where the benzodioxol ring opens or rearranges to deliver the fluorine atoms sequentially. This catalytic cycle is highly efficient, requiring only catalytic amounts of acid relative to the substrate, which minimizes corrosion issues and simplifies neutralization steps post-reaction. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction kinetics and maximize yields for specific substrate classes.
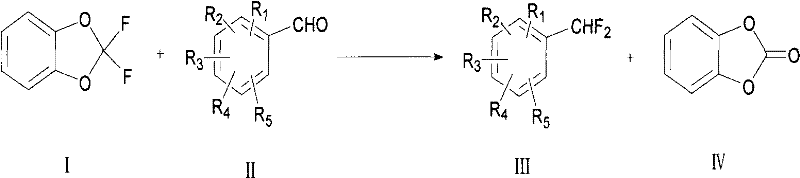
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional fluorination techniques. Because the reagent is a stable liquid and the reaction conditions are moderate, there is less likelihood of thermal decomposition or radical side reactions that often generate complex impurity profiles in gas-phase fluorinations. The selectivity of the acid-catalyzed process ensures that other functional groups on the aromatic ring, such as nitro or halo substituents, remain intact during the transformation. This chemoselectivity is paramount for pharmaceutical applications where strict purity specifications must be met to ensure patient safety and regulatory approval. Furthermore, the byproduct of the reaction, which typically involves the remaining portion of the benzodioxol scaffold, can often be separated easily due to differences in polarity and boiling point. This clean reaction profile reduces the need for extensive chromatographic purification, lowering solvent consumption and processing time, which directly contributes to cost reduction in manufacturing operations.
How to Synthesize Difluoromethyl Aromatic Compounds Efficiently
Executing this synthesis requires careful attention to stoichiometry and reaction conditions to ensure optimal conversion and yield. The general procedure involves charging the aromatic aldehyde substrate and 2,2-difluoro-1,3-benzodioxol into a reaction vessel, typically in a molar ratio ranging from 1:0.4 to 1:20, with a preferred range of 1:0.4 to 1:2 for economic efficiency. A catalyst is then introduced, which can be a strong mineral acid like sulfuric acid or a Lewis acid such as aluminum chloride, depending on the sensitivity of the substrate. The mixture is heated to a temperature between 20°C and 160°C, with 80°C often being sufficient for many substrates, and maintained for several hours to drive the reaction to completion. Solvents such as benzotrifluoride or dichloromethane may be used to facilitate mixing and heat transfer, although solvent-free conditions are also feasible for certain applications. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel by charging aromatic aldehyde substrate and 2,2-difluoro-1,3-benzodioxol fluorinating agent in a molar ratio ranging from 1: 0.4 to 1:20.
- Introduce an acid or Lewis acid catalyst such as sulfuric acid or boron trifluoride, optionally utilizing a solvent like benzotrifluoride or dichloromethane.
- Heat the mixture to a temperature between 20°C and 160°C for several hours, then purify the resulting difluoromethyl product via distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this fluorination technology presents compelling economic and logistical benefits that extend beyond simple chemical yield. The shift from hazardous gaseous reagents to a stable liquid fluorinating agent fundamentally alters the risk profile of the manufacturing process, reducing insurance premiums and safety compliance costs. The elimination of specialized gas handling infrastructure lowers capital expenditure requirements for new production lines, making it easier to scale operations in response to market demand. Additionally, the improved atom economy means that less raw material is wasted, leading to substantial cost savings over the lifecycle of the product. These factors combine to create a more resilient supply chain capable of withstanding regulatory changes and raw material fluctuations. By partnering with a supplier who utilizes this advanced chemistry, companies can secure a more reliable source of critical intermediates.
- Cost Reduction in Manufacturing: The use of 2,2-difluoro-1,3-benzodioxol eliminates the need for expensive and unstable reagents like DAST or hazardous gases like SF4, which require costly containment and disposal protocols. The ability to operate under solvent-free conditions or with common organic solvents further reduces material costs and waste treatment expenses. Since the reagent is a stable liquid, storage and handling do not require cryogenic facilities, significantly lowering utility and infrastructure overheads. The higher fluorine utilization rate means less reagent is needed per kilogram of product, directly improving the gross margin of the manufacturing process. These cumulative efficiencies result in a more competitive pricing structure for the final fluorinated aromatic organic compounds without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing stable liquid reagents is inherently less risky than managing supply chains for toxic gases or thermally unstable solids that may degrade during transit. The widespread availability of the precursors for 2,2-difluoro-1,3-benzodioxol ensures that production is not bottlenecked by scarce raw materials. The robustness of the reaction conditions allows for manufacturing in a broader range of facilities, diversifying the geographic footprint of production and mitigating regional disruption risks. This stability translates to consistent lead times and on-time delivery performance, which is crucial for just-in-time manufacturing models in the pharmaceutical industry. Companies can rely on a steady flow of high-purity intermediates to maintain their own production schedules without unexpected delays.
- Scalability and Environmental Compliance: The non-explosive nature of the reagent and the moderate reaction temperatures make this process highly scalable from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, minimizing the risk of fines or shutdowns due to compliance issues. The simplified workup procedure reduces the volume of organic solvents required for purification, lowering the facility's overall carbon footprint and VOC emissions. This environmental stewardship enhances the corporate social responsibility profile of the supply chain, appealing to end customers who prioritize sustainable sourcing. The ease of scale-up ensures that supply can grow in tandem with market demand, supporting long-term business growth strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology in industrial settings. These answers are derived directly from the experimental data and claims presented in patent CN102153430A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios. The information covers safety, efficiency, and applicability to various chemical structures.
Q: What are the safety advantages of using 2,2-difluoro-1,3-benzodioxol over SF4?
A: Unlike sulfur tetrafluoride (SF4), which is a toxic and difficult-to-control gas with a boiling point of -40.4°C, 2,2-difluoro-1,3-benzodioxol is a stable liquid with a boiling point of 129°C, significantly reducing handling risks and equipment complexity.
Q: How does this method improve fluorine utilization compared to amine salts?
A: Traditional amine salt reagents often utilize only one of two fluorine atoms per molecule, resulting in 50% atom economy. This novel process utilizes the benzodioxol structure to achieve higher fluorine incorporation efficiency into the target difluoromethyl group.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reagent is non-explosive and thermally stable, allowing for operation in standard reactors without specialized high-pressure gas containment, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethyl Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemical market. Our technical team has extensively evaluated the fluorination route described in CN102153430A and confirmed its potential for delivering high-quality fluorinated aromatic organic compounds. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory discovery to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required by the pharmaceutical and agrochemical industries. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to complex synthesis challenges.
We invite you to discuss how this innovative fluorination technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific product requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By collaborating with us, you gain access to a wealth of chemical expertise and a robust production infrastructure designed to support your long-term growth. Let us help you engineer the next generation of fluorinated intermediates with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
