Advanced Synthesis of Ladder-Structured Dibutyltin Piperonate for Oncology Applications
The pharmaceutical industry is constantly seeking novel therapeutic agents that can overcome the limitations of existing treatments, particularly in the field of oncology where resistance and toxicity remain significant hurdles. Patent CN103275115A introduces a groundbreaking development in this arena with the disclosure of a dibutyltin piperonate featuring a unique ladder structure. This specific organotin compound represents a significant departure from traditional platinum-based anticancer drugs, offering a new chemical scaffold that demonstrates potent inhibitory activity against a broad spectrum of human cancer cell lines including liver, nasopharyngeal, breast, lung, and colon cancers. The technical breakthrough lies not only in the biological efficacy but also in the structural stability provided by the tetranuclear tin-oxygen cluster, which forms a robust ladder-like configuration. For R&D directors and technical decision-makers, this patent data suggests a viable pathway for developing next-generation anticancer intermediates that balance high bioactivity with manageable synthesis protocols. The ability to synthesize such complex coordination compounds using relatively straightforward chemical methods opens up new avenues for drug discovery pipelines that require diverse chemical libraries with proven biological potential.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anticancer therapies, particularly those based on platinum coordination complexes, have long been the cornerstone of chemotherapy regimens but suffer from well-documented drawbacks that limit their long-term efficacy and patient quality of life. The primary concern with platinum-based drugs is the development of multidrug resistance, where cancer cells evolve mechanisms to efflux the drug or repair DNA damage more efficiently, rendering the treatment ineffective over time. Furthermore, the toxicity profile of heavy metal-based therapeutics often leads to severe side effects such as nephrotoxicity and neurotoxicity, necessitating complex dosage management and supportive care that increases the overall burden on healthcare systems. From a manufacturing perspective, the synthesis of platinum complexes often involves expensive precious metal precursors and requires stringent purification steps to remove trace metal impurities that could be harmful to patients. These factors contribute to high production costs and supply chain vulnerabilities, as the availability of high-purity platinum group metals can be subject to geopolitical and market fluctuations. Consequently, there is an urgent demand in the pharmaceutical sector for alternative chemical entities that can bypass these resistance mechanisms while offering a more favorable safety and cost profile.
The Novel Approach
The novel approach detailed in the patent data utilizes a ladder-structured dibutyltin piperonate that fundamentally changes the interaction between the metal center and the biological target. By employing a tetranuclear tin-oxygen cluster core, the compound achieves a level of structural rigidity and electronic distribution that enhances its ability to interact with cellular components differently than monomeric organotins or platinum drugs. This ladder structure is formed through the condensation of three four-membered rings using Sn-O bond atoms as bridgehead atoms, creating a stable heterospiro ring structure at the ends where piperic acid ligands coordinate. This specific architecture not only improves the stability of the compound during storage and handling but also appears to correlate with the observed high anticancer activity across multiple cell lines. For procurement and supply chain managers, this new approach is particularly attractive because it relies on organotin chemistry which, while requiring careful handling, utilizes reagents that are generally more accessible and cost-effective than precious metal catalysts. The synthesis method avoids the need for exotic ligands or extreme reaction conditions, suggesting a more robust and scalable manufacturing process that can be integrated into existing fine chemical production facilities with minimal retrofitting.
Mechanistic Insights into Organotin Coordination and Ladder Formation
The formation of the ladder-structured dibutyltin piperonate is a fascinating example of self-assembly in coordination chemistry, driven by the specific affinity between the tin atoms and the oxygen donors from both the methoxy groups and the carboxylate ligands. The core of the molecule features a Sn2O2 planar four-membered ring, which acts as the foundational unit for the larger tetranuclear cluster. Crystallographic analysis reveals that the compound crystallizes in the monoclinic system with space group P21/n, exhibiting precise lattice parameters that confirm the integrity of the ladder structure. The symmetry center of the molecule is located at the center of the middle ring, with two oxygen atoms bridging the three tin atoms within the ladder and two methoxyl oxygen atoms bridging the ladder tin atoms. This intricate arrangement ensures that the butyl groups are positioned to provide steric protection to the metal center, potentially reducing premature degradation in physiological environments. Understanding this mechanism is crucial for R&D teams aiming to replicate or modify the synthesis, as deviations in the molar ratios of reactants or the choice of solvent could disrupt the delicate equilibrium required to form the tetranuclear cluster rather than simpler monomeric or dimeric species.
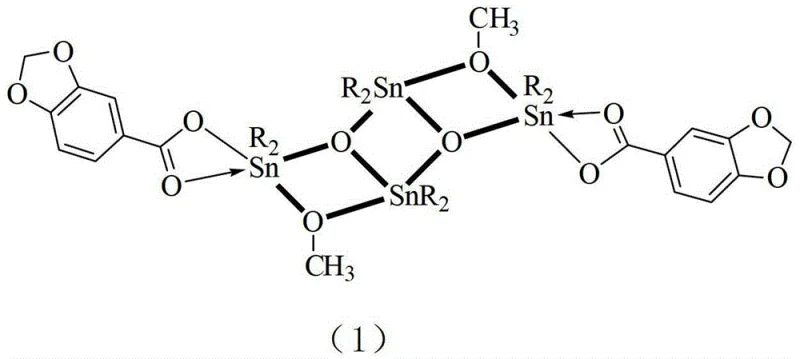
Impurity control in the synthesis of such organotin compounds is paramount, as residual starting materials or incomplete coordination products could compromise the biological safety profile of the final intermediate. The patent data indicates that the reaction proceeds through a reflux mechanism in anhydrous methanol, which serves both as a solvent and a reactant source for the methoxy bridges observed in the final structure. The use of sodium methoxide as a catalyst or base in certain embodiments helps to deprotonate the piperic acid, facilitating the nucleophilic attack on the tin center and promoting the elimination of chloride ions if dibutyltin dichloride is used. This reaction pathway minimizes the formation of hydrolyzed tin oxides which are common impurities in organotin chemistry, provided that moisture is strictly excluded from the reaction environment. The subsequent recrystallization step using a dichloromethane-methanol mixed solvent system is critical for purifying the crude product, as it selectively dissolves unreacted organic acids and simple tin salts while allowing the high-molecular-weight ladder complex to precipitate as colorless crystals. This level of control over the crystallization process ensures that the final product meets the stringent purity specifications required for pharmaceutical intermediates, reducing the risk of batch-to-batch variability in downstream biological testing.
How to Synthesize Dibutyltin Piperonate Efficiently
The synthesis of this high-value anticancer intermediate is designed to be operationally simple while maintaining high standards of chemical purity and structural integrity. The process begins with the precise weighing of piperic acid and the organotin precursor, either dibutyltin oxide or dibutyltin dichloride, which are then suspended in anhydrous methanol. The reaction mixture is subjected to heating under reflux conditions for a period ranging from 8 to 12 hours, ensuring that the thermal energy is sufficient to overcome the activation barrier for the coordination bond formation without degrading the sensitive organic ligands. Following the reaction, the mixture is cooled and filtered to remove any insoluble particulate matter, and the filtrate is concentrated under reduced pressure at controlled temperatures to prevent thermal decomposition of the product. The resulting white solid is then subjected to recrystallization, a critical purification step that leverages the differential solubility of the ladder complex in mixed organic solvents to yield high-purity colorless crystals suitable for further pharmaceutical development.
- Combine piperic acid and dibutyltin dichloride or oxide with anhydrous methanol in a reaction vessel.
- Heat the mixture under stirring reflux for 8 to 12 hours to ensure complete coordination.
- Filter the solution, evaporate the filtrate under reduced pressure, and recrystallize using dichloromethane-methanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ladder-structured dibutyltin piperonate synthesis route offers substantial advantages for procurement managers and supply chain heads looking to optimize their manufacturing costs and reliability. The primary driver for cost reduction lies in the substitution of expensive precious metal catalysts with organotin reagents, which are produced on a much larger industrial scale and are subject to less price volatility than platinum or palladium. Furthermore, the synthesis protocol eliminates the need for complex ligand synthesis, as piperic acid is a naturally derived or readily synthesized aromatic carboxylic acid that is available from multiple global suppliers. This diversification of the raw material base significantly reduces supply chain risk, ensuring that production schedules are not held hostage by the availability of a single specialized reagent. The simplicity of the reaction conditions, which utilize standard reflux equipment and common solvents like methanol and dichloromethane, means that the process can be executed in existing multipurpose chemical plants without requiring capital-intensive specialized reactors. This flexibility allows for rapid scale-up from pilot batches to commercial tonnage, enabling manufacturers to respond quickly to market demand fluctuations for anticancer intermediates.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of commodity chemicals for the reaction medium drastically lower the raw material costs associated with producing this anticancer intermediate. By avoiding the expensive purification steps typically required to remove trace heavy metals from pharmaceutical products, the overall processing cost is significantly reduced, allowing for more competitive pricing in the global market. The high yield and selectivity of the reaction minimize waste generation, which further contributes to cost savings by reducing the burden on waste treatment facilities and lowering the consumption of raw materials per unit of product. Additionally, the energy requirements for the reflux process are moderate compared to high-pressure or cryogenic reactions, resulting in lower utility costs over the lifecycle of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as dibutyltin oxide and piperic acid ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. Methanol and dichloromethane are bulk solvents produced by major chemical companies worldwide, guaranteeing consistent quality and availability even during periods of high market demand. This robustness in raw material sourcing translates directly into improved delivery reliability for downstream pharmaceutical clients, who depend on timely shipments to maintain their own drug development timelines. The ability to source materials from multiple geographic regions also provides procurement teams with greater negotiating leverage, further enhancing the economic viability of the project.
- Scalability and Environmental Compliance: The synthesis route is inherently scalable, as the reaction kinetics and heat transfer characteristics are well-suited for large-scale batch reactors commonly found in fine chemical manufacturing. The use of standard solvents simplifies the solvent recovery and recycling process, allowing manufacturers to implement closed-loop systems that minimize environmental impact and comply with increasingly stringent regulatory standards. The solid product is easily isolated by filtration and crystallization, avoiding complex extraction or chromatography steps that are difficult to scale and generate large volumes of hazardous waste. This streamlined downstream processing not only improves operational efficiency but also reduces the environmental footprint of the manufacturing process, aligning with the sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ladder-structured dibutyltin piperonate, based on the detailed data provided in the patent documentation. These insights are intended to clarify the feasibility of integrating this intermediate into existing drug discovery workflows and to highlight the specific advantages it offers over conventional alternatives. Understanding these details is essential for technical teams evaluating the compound for preclinical studies and for commercial teams assessing its potential market value.
Q: How does the ladder structure affect anticancer activity?
A: The tetranuclear tin-oxygen cluster ladder structure enhances stability and biological interaction compared to monomeric organotins, leading to superior inhibitory activity against various cancer cell lines.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes common solvents like methanol and standard reflux conditions, avoiding complex catalytic systems, which facilitates easy scale-up from laboratory to commercial manufacturing.
Q: What are the purity specifications for this intermediate?
A: The process yields colorless crystals with verified elemental analysis and crystallographic data, ensuring high purity suitable for downstream pharmaceutical development and rigorous QC testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibutyltin Piperonate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, offering specialized expertise in the scale-up and production of complex organometallic intermediates like the ladder-structured dibutyltin piperonate. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We understand the critical importance of stringent purity specifications in the pharmaceutical sector and have invested heavily in rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and purity of every batch. Our commitment to quality assurance means that clients can rely on us to deliver intermediates that meet the exacting standards required for regulatory filings and clinical trials, minimizing the risk of delays in their drug development programs.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for anticancer drug development. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis that identifies specific opportunities to reduce manufacturing expenses without compromising on quality or safety. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Whether you need small quantities for initial screening or large volumes for commercial production, our flexible manufacturing platform is designed to support your needs at every stage of the product lifecycle.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
