Advanced Organotin Coordination Chemistry for Next-Generation Anticancer Drug Manufacturing
Advanced Organotin Coordination Chemistry for Next-Generation Anticancer Drug Manufacturing
The landscape of oncology therapeutics is undergoing a significant paradigm shift, moving beyond the limitations of traditional platinum-based chemotherapy towards more targeted and lipophilic organometallic candidates. A pivotal development in this arena is documented in Chinese Patent CN103044482A, which discloses a novel dibutyltin coordination compound exhibiting potent antitumor properties. This specific chemical entity, characterized by its unique coordination geometry involving tin-oxygen bonds, represents a breakthrough in designing metal-based drugs that can overcome cellular resistance mechanisms often encountered with cisplatin derivatives. For pharmaceutical developers and procurement strategists, understanding the synthesis and commercial viability of this compound is critical, as it offers a pathway to high-efficacy treatments with a markedly simplified manufacturing footprint compared to legacy anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of metal-based anticancer drugs has been dominated by platinum complexes, which, while effective, suffer from severe drawbacks including high systemic toxicity, poor solubility profiles, and the rapid development of tumor resistance. The synthesis of these conventional agents often requires harsh reaction conditions, expensive noble metal precursors, and complex purification steps to remove toxic metal residues, leading to inflated production costs and supply chain vulnerabilities. Furthermore, the hydrophilic nature of many platinum drugs limits their ability to penetrate lipid bilayers effectively, reducing bioavailability at the tumor site. These factors collectively create a bottleneck in the cost reduction in anticancer drug manufacturing, forcing manufacturers to rely on high-volume production to maintain margins, which is not always feasible for specialized oncology intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined coordination strategy utilizing abundant tin precursors rather than scarce platinum group metals. The novel approach leverages the inherent reactivity of dibutyltin dichloride with 2-phthalimidopropionic acid to form a stable, lipophilic complex under mild conditions. This shift not only drastically reduces the raw material costs associated with noble metals but also simplifies the downstream processing requirements. The resulting organotin compound demonstrates superior fat solubility, which is a critical pharmacokinetic parameter for enhancing cellular uptake and therapeutic index. By adopting this synthetic route, manufacturers can achieve a reliable pharmaceutical intermediate supplier status through a process that is both economically sustainable and chemically robust, addressing the dual needs of efficacy and manufacturability.
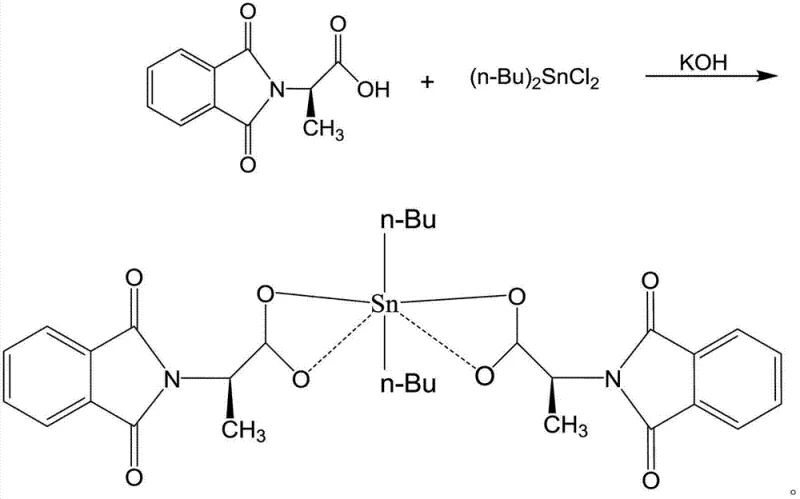
Mechanistic Insights into Dibutyltin Coordination Chemistry
The core of this innovation lies in the specific coordination environment surrounding the tin center, which dictates the biological activity and stability of the final molecule. As illustrated in the structural analysis, the tin atom adopts a distorted octahedral geometry, coordinated by two n-butyl groups and four oxygen atoms derived from the carboxylate ligands of the phthalimido-propionic acid moieties. This specific arrangement creates a rigid yet accessible structure that facilitates interaction with biological targets, potentially through DNA binding or enzyme inhibition mechanisms distinct from platinum drugs. The presence of the phthalimide group further enhances the lipophilic character of the molecule, allowing it to traverse cell membranes more efficiently than charged platinum species. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize the high-purity organotin compounds for specific therapeutic applications, as slight variations in the coordination sphere can significantly alter the toxicity profile.
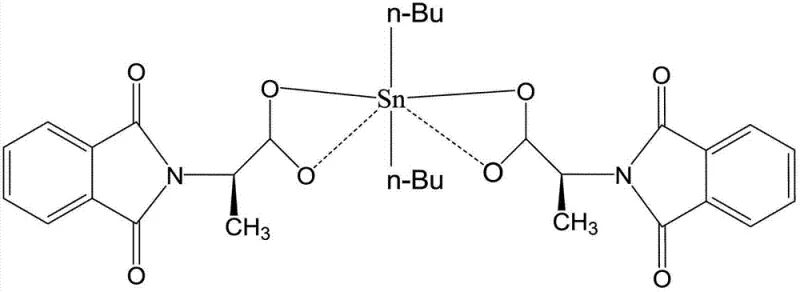
From an impurity control perspective, the reaction mechanism favors the formation of a discrete monomeric or oligomeric species depending on the stoichiometry, but the patented conditions specifically drive the equilibrium towards the desired neutral complex. The use of potassium hydroxide acts as a deprotonating agent, facilitating the nucleophilic attack of the carboxylate oxygen on the tin center while eliminating chloride ions as potassium salts. This clean displacement reaction minimizes the formation of hydrolytic byproducts or polymeric tin oxides that often plague organotin syntheses. Consequently, the impurity profile is significantly cleaner, reducing the burden on purification teams and ensuring that the final API intermediate meets stringent regulatory standards for heavy metal content and organic impurities, a key consideration for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Dibutyltin Coordination Compound Efficiently
The synthesis protocol described in the patent is remarkably straightforward, designed to maximize yield while minimizing operational complexity. The process involves a direct condensation reaction in a polar protic solvent, specifically methanol, which serves both as a reaction medium and a solubilizing agent for the ionic intermediates. The reaction proceeds at ambient temperatures, eliminating the need for energy-intensive heating or cooling cycles, which is a significant advantage for large-scale production facilities aiming to reduce their carbon footprint and utility costs. The simplicity of the workup, involving merely rotary evaporation followed by a standard recrystallization, ensures that the reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising on quality. Below is the detailed procedural framework derived from the patent data.
- Combine 2-phthalimidopropionic acid, potassium hydroxide, and dibutyltin dichloride in methanol solvent.
- Stir the reaction mixture at room temperature for 5 to 6 hours to ensure complete coordination.
- Perform rotary evaporation followed by recrystallization using a dichloromethane-petroleum ether system to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organotin-based scaffold offers compelling economic and logistical benefits that extend beyond simple raw material substitution. The elimination of platinum, a volatile and geopolitically sensitive commodity, stabilizes the cost structure of the manufacturing process, shielding the organization from sudden market spikes in precious metal prices. Additionally, the use of common laboratory solvents like methanol and petroleum ether, along with widely available starting materials such as dibutyltin dichloride, ensures a resilient supply chain that is less prone to disruption. The operational simplicity of the process, which does not require specialized high-pressure reactors or inert atmosphere gloveboxes for the main reaction step, allows for flexible manufacturing across multiple sites, thereby enhancing overall supply security.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the replacement of expensive platinum precursors with cost-effective tin reagents. Tin is orders of magnitude cheaper than platinum, and the stoichiometry of the reaction is efficient, typically requiring a near 1:1 molar ratio of reactants. Furthermore, the mild reaction conditions (room temperature stirring) significantly lower energy consumption compared to processes requiring prolonged reflux or cryogenic conditions. The high yields reported, ranging from 76% to 86%, indicate excellent atom economy, meaning less waste is generated per unit of product, which directly translates to lower disposal costs and higher overall process efficiency.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, including 2-phthalimidopropionic acid and dibutyltin dichloride, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction, which tolerates minor variations in stoichiometry (e.g., KOH ranging from 1.0 to 1.5 mmol), provides a wide operating window that ensures consistent batch-to-batch quality even with slight fluctuations in raw material quality. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of potassium chloride salts and solvent streams that can be readily recovered and recycled. The absence of heavy metal catalysts that require complex scavenging steps simplifies the environmental compliance burden. The recrystallization step uses a binary solvent system of dichloromethane and petroleum ether, which allows for precise control over crystal morphology and purity, facilitating easy filtration and drying on an industrial scale. This scalability ensures that the transition from gram-scale R&D to multi-ton commercial production can be executed smoothly without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this dibutyltin coordination compound. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios.
Q: What are the primary advantages of this organotin compound over platinum-based drugs?
A: According to patent CN103044482A, this dibutyltin coordination compound exhibits higher antitumor activity and superior liposolubility compared to traditional platinum agents, alongside a significantly simpler and lower-cost preparation method.
Q: What is the expected yield and purity profile for this synthesis?
A: The patented process demonstrates robust yields ranging from 76% to 86% under optimized conditions, producing light yellow transparent crystals with verified structural integrity via IR and NMR spectroscopy.
Q: Is this compound scalable for industrial pharmaceutical production?
A: Yes, the synthesis utilizes readily available precursors like dibutyltin dichloride and operates at room temperature without requiring extreme pressure or exotic catalysts, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibutyltin Coordination Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organotin technology in the field of oncology. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready therapy is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of our organotin intermediates meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent supply of high-performance materials essential for your drug development pipeline.
We invite you to collaborate with us to optimize your supply chain and accelerate your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals. Let us be your partner in bringing next-generation anticancer therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
