Revolutionizing OLED Material Production: A One-Step Copper-Catalyzed Route to Tetra-aryl Ethylenes
Revolutionizing OLED Material Production: A One-Step Copper-Catalyzed Route to Tetra-aryl Ethylenes
The rapid evolution of the organic electronics sector demands increasingly efficient and cost-effective synthetic routes for high-performance materials. Patent CN111732532B introduces a groundbreaking methodology for the synthesis of tetra-aryl substituted ethylene derivatives bearing electron-deficient groups, a class of compounds critical for the fabrication of advanced Organic Light Emitting Diodes (OLEDs) and solar cell materials. This innovation addresses long-standing challenges in the field by replacing complex, multi-step sequences with a direct, one-step oxidative coupling reaction mediated by inexpensive copper reagents. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and economically viable manufacturing processes for high-value electronic chemicals. The ability to generate these star-shaped molecular architectures directly from readily available diarylmethane precursors not only streamlines the supply chain but also significantly reduces the environmental footprint associated with traditional halogenated or titanium-mediated pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetra-aryl ethylene core has been a formidable synthetic challenge, often necessitating harsh reaction conditions and expensive catalytic systems. Traditional approaches frequently rely on the McMurry coupling reaction, which utilizes low-valent titanium species generated in situ from TiCl4 and reducing agents like zinc or lithium aluminum hydride. While effective, this method suffers from severe drawbacks including the generation of massive amounts of titanium waste, strict requirement for anhydrous and oxygen-free environments, and difficulties in controlling stereochemistry and byproduct formation. Furthermore, alternative routes involving the dehydration of tetraaryl ethanol intermediates or the dehydrogenation of tetraarylethanes often involve multiple synthetic steps, leading to cumulative yield losses and increased operational costs. These legacy methods create significant bottlenecks for commercial scale-up, particularly when targeting the high purity standards required for electronic grade materials, where trace metal contaminants can devastate device performance.
The Novel Approach
In stark contrast, the methodology disclosed in CN111732532B offers a remarkably elegant solution by leveraging the oxidative power of simple copper salts to drive the direct coupling of diarylmethanes. This novel approach bypasses the need for pre-functionalized intermediates or hazardous reducing agents, instead utilizing molecular oxygen or the copper oxidant itself to facilitate the dehydrogenative dimerization. As illustrated in the general reaction scheme below, the transformation proceeds smoothly in polar aprotic solvents under a nitrogen atmosphere, tolerating a wide array of electron-withdrawing substituents.
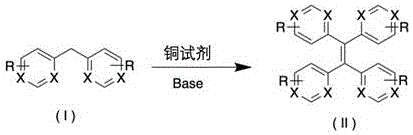
The simplicity of this protocol cannot be overstated; it combines the substrate, a copper source such as copper acetate, and a base in a single pot, heating the mixture to moderate temperatures between 80°C and 150°C. This drastic reduction in procedural complexity translates directly into lower capital expenditure for reactor infrastructure and reduced labor costs for process execution. For a reliable OLED material supplier, adopting this route means faster time-to-market for new emissive or charge-transporting materials, providing a distinct competitive advantage in the fast-paced display technology market.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The success of this transformation hinges on the unique ability of copper species to mediate single-electron transfer processes that activate the benzylic C-H bonds of the diarylmethane substrates. Mechanistically, the reaction likely initiates with the coordination of the copper(II) species to the electron-deficient aromatic rings or the basic nitrogen atoms in heterocyclic variants, facilitating the abstraction of a benzylic proton by the added base. This generates a resonance-stabilized benzylic radical or carbanion intermediate, which subsequently undergoes oxidative dimerization to form the central carbon-carbon double bond. The presence of electron-deficient groups such as cyano, nitro, or acetyl moieties plays a crucial role in stabilizing these intermediates, thereby lowering the activation energy barrier for the coupling event. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters for specific substrates, ensuring maximum conversion and minimal formation of over-oxidized byproducts.
Furthermore, the choice of base and solvent system is instrumental in controlling the impurity profile of the final product. The patent data indicates that bases like potassium carbonate or potassium tert-butoxide, often supplemented with phase transfer catalysts like 18-crown-6, effectively promote the reaction while maintaining a clean product distribution. For instance, the synthesis of tetrakis(2-pyridyl)ethylene demonstrates the robustness of this system even with coordinating nitrogen heterocycles, achieving a respectable 75% yield with high purity after simple chromatographic purification.
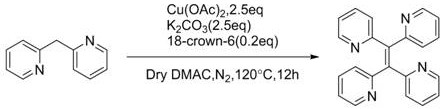
This high level of selectivity is paramount for electronic applications, where trace impurities can act as charge traps or quenching sites, severely degrading the efficiency and lifespan of OLED devices. The ability of this copper-catalyzed system to deliver products with such structural fidelity underscores its potential as a premier method for cost reduction in electronic chemical manufacturing, allowing producers to meet stringent quality specifications without resorting to exhaustive recrystallization or sublimation processes.
How to Synthesize Tetra-aryl Substituted Ethylene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and atmospheric control, although the conditions are far less demanding than traditional titanium-mediated couplings. The process begins with the precise weighing of the diarylmethane precursor and the copper oxidant, typically using a molar ratio of copper to substrate between 1:1 and 5:1, with 2.5 equivalents often proving optimal for balancing reaction rate and cost. The reaction is conducted in dry, high-boiling polar solvents such as N,N-dimethylacetamide (DMAC) or hexamethylphosphoric triamide (HMPA), which solubilize both the organic substrates and the inorganic bases effectively. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for ensuring batch-to-batch consistency and safety.
- Combine diaryl substituted methane, anhydrous copper acetate (2.5 eq), and a base such as potassium carbonate or potassium tert-butoxide in a dry reaction vessel under nitrogen atmosphere.
- Add a phase transfer catalyst like 18-crown-6 and a polar aprotic solvent such as DMAC or HMPA, then heat the mixture to 80-150°C for 12 hours.
- Upon completion, cool the mixture, perform aqueous workup with acid or base depending on substituents, extract with organic solvents, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this copper-catalyzed methodology offers profound benefits that extend well beyond the laboratory bench. The primary driver for cost optimization lies in the replacement of exotic and hazardous reagents with commodity chemicals that are available in bulk quantities globally. Copper acetate and simple inorganic bases represent a fraction of the cost of titanium tetrachloride or palladium catalysts, and their handling does not require specialized containment systems or extensive safety training. This shift in raw material profile fundamentally alters the cost structure of producing tetra-aryl ethylene derivatives, enabling significant margin improvement for manufacturers of OLED intermediates and allowing for more aggressive pricing strategies in a competitive market.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences and the use of inexpensive copper catalysts drastically reduce the overall cost of goods sold. By avoiding the need for cryogenic cooling, inert gas lines for sensitive reagents, and complex waste neutralization streams associated with titanium sludge, the operational expenditure is significantly lowered. Furthermore, the high atom economy of the direct coupling reaction minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable saleable product, thereby enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Sourcing risks are markedly diminished as the key reagents—diarylmethanes and copper salts—are produced by a vast network of chemical suppliers worldwide, ensuring continuity of supply even during market fluctuations. Unlike specialized catalysts that may have long lead times or single-source dependencies, the commoditized nature of the inputs for this process guarantees that production schedules can be maintained without interruption. This reliability is critical for downstream clients in the display and photovoltaic industries, who depend on just-in-time delivery of high-purity materials to keep their own fabrication lines running smoothly.
- Scalability and Environmental Compliance: The robustness of the reaction conditions allows for seamless scale-up from benchtop to commercial tonnage without the need for re-engineering the core process. The use of standard solvents and the absence of highly toxic byproducts simplify the environmental compliance landscape, reducing the burden on wastewater treatment facilities and lowering the costs associated with hazardous waste disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, a factor of increasing importance to global enterprise customers evaluating their supply chain partners.
To illustrate the commercial viability of this approach, consider the synthesis of tetrakis(4-cyanophenyl)ethylene, a valuable intermediate for n-type semiconductor materials. The patent reports an exceptional 96% isolated yield for this transformation, a figure that is rarely achieved in traditional multi-step syntheses.
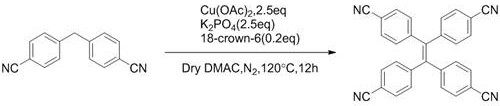
Such high efficiency directly correlates to reduced solvent consumption and lower energy usage per kilogram of product, further reinforcing the economic and environmental arguments for adopting this technology. For supply chain heads, this means a more predictable and lean inventory model, as less safety stock is required to buffer against process inefficiencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what potential partners can expect regarding process performance and product quality. Understanding these details is essential for making informed decisions about integrating this technology into existing manufacturing portfolios.
Q: What are the primary advantages of this copper-catalyzed method over traditional TiCl4 mediated synthesis?
A: The copper-catalyzed method described in CN111732532B eliminates the need for hazardous and moisture-sensitive reagents like TiCl4, operates under milder thermal conditions, and utilizes inexpensive, commercially available copper salts, drastically simplifying the operational complexity and waste treatment protocols.
Q: Can this synthesis method accommodate diverse electron-deficient groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully synthesizing derivatives with cyano, nitro, acetyl, and pyridyl groups with high yields ranging from 68% to 96%, making it highly versatile for tuning the electronic properties of OLED materials.
Q: Is this process suitable for large-scale commercial production of OLED intermediates?
A: Absolutely. The reaction uses robust reagents, standard polar aprotic solvents like DMAC, and straightforward workup procedures involving extraction and chromatography, which are easily adaptable to kilogram-scale manufacturing without requiring specialized cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-aryl Ethylene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis described in CN111732532B for the next generation of electronic materials. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust, GMP-compliant commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to guarantee that every batch of tetra-aryl ethylene derivative meets the exacting standards required for high-performance OLED and solar cell applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific product needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current supply chain constraints. We encourage you to reach out today to discuss your project requirements,索取 specific COA data for our reference standards, and obtain comprehensive route feasibility assessments that will demonstrate how we can drive value and innovation in your electronic materials portfolio.
