Advanced Selenium/Sulfur Compounds for High-Efficiency Asymmetric Catalysis and Commercial Manufacturing
The landscape of asymmetric catalysis is continuously evolving, driven by the urgent industrial demand for more efficient and cost-effective chiral ligands. Patent CN115197271B introduces a groundbreaking class of selenium and sulfur compounds based on a 1,2-diaryl-1,2-ethylene glycol (amine) skeleton, addressing critical gaps in current catalytic technologies. Unlike traditional catalysts that rely on expensive and structurally rigid binaphthol or TADDOL backbones, this innovation leverages the flexible and synthetically accessible 1,2-diphenyl framework. The patent details two distinct synthetic pathways: one utilizing 1,2-diphenyl-1,2-ethylenediamine analogues and another employing chiral 1,2-diphenyl-1,2-ethylene glycol. Both routes culminate in the formation of robust P-Se or P-S bonds, which are pivotal for enhancing catalytic activity in the synthesis of high-value axial chiral sulfur-containing derivatives. This technological advancement represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediates supplier partnerships that prioritize both performance and economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral ligands has been dominated by scaffolds such as binaphthol, spiro phenols, and TADDOL, which, while effective, suffer from inherent limitations in terms of cost and synthetic complexity. The preparation of these traditional ligands often involves multi-step sequences requiring harsh reaction conditions and expensive precursors, which drastically inflates the final cost of the catalyst. Furthermore, the rigid structural motifs of these conventional ligands can sometimes limit their substrate scope, making them less versatile for diverse asymmetric transformations. In the specific context of selenium and sulfur-based catalysis, prior art has largely focused on oxide derivatives or metal-organic frameworks grafted with diamines, leaving the potential of direct 1,2-diphenyl-based selenium/sulfur compounds largely unexplored. This gap has resulted in a market reliance on suboptimal catalysts that fail to deliver the necessary balance of high enantioselectivity and process efficiency required for modern fine chemical manufacturing.
The Novel Approach
The methodology disclosed in CN115197271B revolutionizes this domain by introducing a streamlined, one-pot synthesis strategy that functionalizes hydrogen on the 1,2-diphenyl substrate directly. This approach bypasses the need for complex pre-functionalization steps, utilizing cheap and easily purchased starting materials like 1,2-diphenyl alcohol or amine substrates. The core innovation lies in the sequential treatment of these substrates with lithiating agents or phosphorus halides, followed by a controlled oxidation with elemental selenium or sulfur powder. This process not only simplifies the synthetic route but also allows for precise tuning of the steric and electronic environment around the phosphorus center by varying the N-substituents (R and R1 groups). The resulting compounds exhibit exceptional catalytic activity, particularly in the asymmetric direct aldol condensation of p-nitrobenzaldehyde and acetone, demonstrating yields and enantiomeric excess values that surpass many existing commercial alternatives.
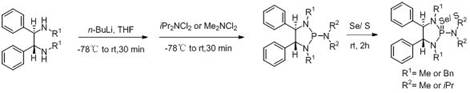
Mechanistically, the success of this novel approach hinges on the precise control of the phosphorylation and oxidation stages. In the amine-based pathway, the initial deprotonation with n-butyllithium at cryogenic temperatures (-78°C) generates a highly reactive nucleophilic species that attacks the phosphorus center of dichloro-N,N-diisopropylphosphoramidite. This step is critical for establishing the P-N bond framework without inducing racemization or decomposition of the chiral backbone. Subsequently, the introduction of selenium or sulfur powder acts as a soft oxidant, converting the trivalent phosphorus intermediate into the corresponding selenide or sulfide. The formation of the P=Se or P=S double bond significantly alters the Lewis basicity of the nitrogen atoms and the Lewis acidity of the phosphorus, creating a cooperative catalytic site. This dual activation mode is essential for stabilizing the transition state during the synthesis of axial chiral sulfur-containing diphenyl derivatives, thereby ensuring high stereocontrol. The ability to modulate these electronic properties through simple substituent changes offers R&D teams unprecedented flexibility in optimizing catalyst performance for specific reaction profiles.
Impurity control is another paramount aspect of this synthesis, particularly given the sensitivity of organoselenium and organosulfur compounds to oxidation and hydrolysis. The patent specifies rigorous anhydrous conditions and the use of dry solvents such as tetrahydrofuran (THF) or dichloromethane (DCM) to prevent the formation of phosphine oxides or other hydrolysis byproducts. The low-temperature protocols employed during the addition of reactive reagents like n-BuLi and PCl3 minimize side reactions such as over-lithiation or elimination, which could otherwise degrade the chiral integrity of the 1,2-diphenyl scaffold. Furthermore, the final purification via column chromatography ensures the removal of unreacted selenium or sulfur powder and any inorganic salts generated during the triethylamine neutralization steps. This meticulous attention to reaction parameters guarantees a high-purity final product, which is indispensable for applications in the pharmaceutical sector where trace metal or heteroatom impurities can compromise downstream drug safety and efficacy.
How to Synthesize 1,2-Diphenylamines Compound Efficiently
The synthesis of these high-performance catalysts is designed to be operationally simple yet chemically robust, making it highly suitable for translation from benchtop to pilot plant. The process begins with the dissolution of the chiral diamine or glycol substrate in an appropriate solvent, followed by careful temperature management to ensure reaction fidelity. Detailed standardized operating procedures for the lithiation, phosphitylation, and oxidation steps are critical for reproducibility, especially when scaling up to meet commercial demand. The following guide outlines the fundamental workflow derived from the patent's preferred embodiments, providing a clear roadmap for technical teams aiming to implement this technology.
- Prepare the substrate by dissolving 1,2-diphenylamine analogues or chiral 1,2-diphenyl-1,2-ethylene glycol in anhydrous solvents like THF or DCM under inert atmosphere.
- Execute the phosphorylation step using n-BuLi and dichloro-N,N-diisopropylphosphoramidite for amines, or PCl3 and triethylamine for glycols, maintaining strict temperature control between -78°C and 0°C.
- Complete the synthesis by oxidizing the intermediate phosphorus species with elemental selenium or sulfur powder at room temperature, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible strategic benefits centered around cost optimization and supply security. The primary driver for cost reduction is the substitution of expensive, specialized chiral ligands with commoditized 1,2-diphenyl building blocks that are widely available in the global chemical market. By eliminating the need for complex multi-step syntheses associated with traditional binaphthol derivatives, manufacturers can significantly reduce raw material consumption and processing time. Moreover, the one-pot nature of the reaction minimizes solvent usage and waste generation, aligning with green chemistry principles that are increasingly becoming a prerequisite for supplier qualification in the pharmaceutical industry. These efficiencies translate directly into a more competitive pricing structure for the final catalyst, allowing downstream users to achieve substantial cost savings in their own manufacturing processes without compromising on quality.
Supply chain reliability is further enhanced by the use of standard, non-proprietary reagents such as phosphorus trichloride, triethylamine, and elemental selenium. Unlike proprietary ligands that may be subject to single-source bottlenecks or long lead times, the inputs for this synthesis are produced by multiple global suppliers, ensuring a resilient and continuous supply stream. The scalability of the process is another critical advantage; the reaction conditions, ranging from -78°C to room temperature, are compatible with standard stainless steel reactors found in most fine chemical facilities. This compatibility reduces the barrier to entry for commercial scale-up, enabling rapid deployment of production capacity to meet fluctuating market demands. Consequently, partners can secure a stable source of high-purity chiral catalysts that supports uninterrupted production schedules for critical active pharmaceutical ingredients.
Environmental compliance and waste management are also significantly improved through this synthetic route. The avoidance of transition metal catalysts, which are often required in traditional cross-coupling or hydrogenation reactions to generate chiral centers, eliminates the costly and technically challenging step of heavy metal removal from the final product. This not only reduces the environmental footprint of the manufacturing process but also simplifies the regulatory approval pathway for pharmaceutical intermediates. Additionally, the high atom economy of the phosphorylation and oxidation steps ensures that the majority of the starting materials are incorporated into the final product, minimizing the volume of hazardous waste requiring disposal. For supply chain heads focused on sustainability metrics, this technology offers a clear path to reducing the overall carbon intensity of the chemical supply chain while maintaining the rigorous quality standards demanded by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these novel selenium/sulfur compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, scalability, and comparative performance. Understanding these details is essential for R&D directors and procurement specialists evaluating the feasibility of integrating this technology into their existing manufacturing workflows.
Q: What are the primary advantages of 1,2-diphenyl-based selenium catalysts over traditional binaphthol ligands?
A: The 1,2-diphenyl backbone offers superior flexibility and controllability compared to rigid binaphthol structures. The synthesis utilizes cheaper, readily available starting materials and avoids complex multi-step routes, significantly reducing production costs while maintaining excellent catalytic activity in asymmetric reactions.
Q: How does the oxidation step with selenium or sulfur powder impact the catalyst's performance?
A: The introduction of selenium or sulfur atoms creates P=Se or P=S bonds which modify the electronic properties of the phosphorus center. This enhancement improves the Lewis acidity or basicity of the catalyst, leading to higher enantioselectivity and yield in the synthesis of axial chiral sulfur-containing diphenyl derivatives.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method employs standard industrial reagents such as phosphorus trichloride and n-butyllithium in common solvents like THF and DCM. The one-pot nature of the reaction and mild temperature requirements (-78°C to room temperature) facilitate straightforward scale-up from laboratory to commercial tonnage without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selenium/Sulfur Compounds Supplier
As the global demand for advanced chiral catalysts continues to surge, NINGBO INNO PHARMCHEM stands ready to support your organization with world-class manufacturing capabilities and technical expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of selenium/sulfur compound meets the highest international standards. By leveraging our deep understanding of organophosphorus and organoselenium chemistry, we can assist you in optimizing reaction parameters to maximize yield and enantioselectivity for your specific applications.
We invite you to engage with our technical procurement team to discuss how these innovative catalysts can transform your production efficiency. Whether you require a Customized Cost-Saving Analysis for your current ligand portfolio or need specific COA data to validate our quality systems, we are prepared to provide comprehensive support. Contact us today to request route feasibility assessments and discover how our partnership can drive value and innovation in your fine chemical manufacturing operations.
