Advanced Organotin-Catalyzed Synthesis of Dimethoxy Bisphenol A Diester for High-Performance Polycarbonates
The global demand for high-performance engineering plastics continues to drive innovation in polymer intermediate synthesis, specifically seeking alternatives to hazardous traditional methods. Patent CN102442911B introduces a groundbreaking preparation method for dimethoxy bisphenol A diester (DmC(1)), a critical precursor for non-phosgene polycarbonate production. This technology leverages an organotin-catalyzed transesterification between bisphenol A (BPA) and dimethyl carbonate (DMC), operating effectively at temperatures between 140-250°C. By shifting away from the historically dominant but dangerous phosgene process, this invention addresses critical safety and environmental concerns while maintaining exceptional product quality. The resulting intermediate serves as a robust foundation for synthesizing polycarbonates with superior thermal stability and mechanical strength, positioning it as a vital component for advanced material supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of polycarbonate has relied heavily on the interfacial polycondensation method using phosgene, which presents severe logistical and safety challenges. This traditional route necessitates the use of dichloromethane as a solvent and generates substantial quantities of corrosive hydrogen chloride gas as a byproduct, requiring complex scrubbing systems and posing significant risks to worker health. Alternatively, the melt transesterification process using diphenyl carbonate (DPC) avoids phosgene but suffers from economic and thermodynamic drawbacks, including the high cost of DPC raw materials and the difficulty in removing the high-boiling phenol byproduct. These conventional pathways often struggle with equilibrium limitations that hinder the formation of high molecular weight polymers without extreme vacuum conditions and excessive energy input, creating bottlenecks in efficient commercial scale-up of complex polymer additives.
The Novel Approach
The patented methodology revolutionizes this landscape by utilizing dimethyl carbonate (DMC) as a green carbonyl source, which is not only more cost-effective than DPC but also generates methanol as a low-boiling byproduct that is easily separated. This novel approach employs specific organotin catalysts, such as diaryltin oxides or halides, to drive the double alkoxy-carbonylation of bisphenol A with high selectivity. By optimizing the molar ratio of DMC to BPA between 2 and 20, the process effectively suppresses oligomerization side reactions, ensuring the formation of the discrete diester intermediate rather than premature polymerization. This strategic shift enables a streamlined workflow that drastically simplifies downstream purification and significantly reduces the overall environmental footprint of polycarbonate manufacturing.
Mechanistic Insights into Organotin-Catalyzed Transesterification
The core of this technological advancement lies in the precise activation of the carbonate bond by the organotin catalyst, which facilitates a nucleophilic attack by the phenolic hydroxyl groups of bisphenol A. The reaction proceeds through a stepwise mechanism where the catalyst coordinates with the carbonyl oxygen of DMC, increasing its electrophilicity and allowing the first hydroxyl group of BPA to form a mono-methyl carbonate intermediate (MmC). Subsequently, the second hydroxyl group undergoes a similar transesterification event to yield the final dimethoxy bisphenol A diester (DmC(1)). This catalytic cycle is highly efficient, achieving bisphenol A conversion rates exceeding 99% and selectivity for the target diester greater than 95%, thereby minimizing the formation of alkylated impurities that could degrade the final polymer's optical clarity.
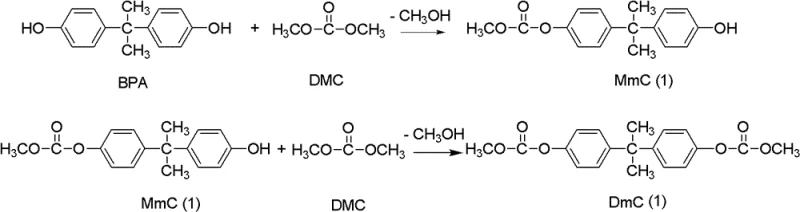
Impurity control is rigorously managed through the specific choice of organotin species and the implementation of a unique recrystallization protocol. The patent specifies that crude products can be purified using an alcohol-water mixed solvent system with a volume ratio controlled between 1 and 3. This solvent system is engineered to exploit the differential solubility of the target diester versus unreacted starting materials and mono-substituted intermediates. By dissolving the crude solid at elevated temperatures (e.g., 65-70°C) and cooling slowly, the process ensures the exclusion of trace organic impurities and catalyst residues. This results in a refined intermediate with purity levels surpassing 99.8%, which is essential for preventing discoloration and maintaining the high impact resistance required in premium polycarbonate applications.
How to Synthesize Dimethoxy Bisphenol A Diester Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and thermal management to maximize yield while ensuring operational safety. The process is versatile, accommodating both batch autoclave operations under nitrogen pressure and continuous atmospheric reactors with distillation setups for methanol removal. Detailed standard operating procedures regarding specific temperature ramps, stirring velocities, and solvent ratios are critical for reproducibility. For a comprehensive breakdown of the exact experimental parameters and step-by-step execution guide, please refer to the standardized synthesis protocol below.
- Mix Bisphenol A (BPA) and Dimethyl Carbonate (DMC) with an organotin catalyst (e.g., diphenyltin oxide) at a molar ratio of 2-20: 1.
- Heat the mixture to 140-250°C under normal or high pressure for 0.5 to 48 hours to facilitate transesterification.
- Purify the crude product via recrystallization using an alcohol-water mixed solvent (volume ratio 1-3) to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, this patented process offers a compelling value proposition centered on raw material optimization and risk mitigation. By substituting expensive diphenyl carbonate with widely available dimethyl carbonate, manufacturers can achieve substantial cost savings in polymer intermediate manufacturing without compromising on quality. The elimination of toxic phosgene removes the need for specialized containment infrastructure and hazardous material transport permits, thereby simplifying logistics and reducing insurance liabilities. Furthermore, the ease of separating methanol byproducts translates to lower energy consumption during the devolatilization stage, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The utilization of DMC as a feedstock provides a distinct economic advantage over DPC due to lower market pricing and higher availability. Additionally, the solvent-free nature of the reaction (or use of minimal solvent) combined with the easy removal of methanol reduces the capital expenditure associated with solvent recovery units. This streamlined process flow eliminates the need for complex wastewater treatment systems required for phosgene-based routes, leading to significant operational expenditure reductions over the lifecycle of the plant.
- Enhanced Supply Chain Reliability: Sourcing DMC and organotin catalysts is generally more stable and less regulated than sourcing phosgene or managing chlorine supply chains. The robustness of the reaction conditions, which tolerate a range of pressures and temperatures, ensures consistent output even with minor fluctuations in utility supplies. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major automotive and electronics clients who depend on steady polycarbonate supplies.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial reactors, as demonstrated by the patent's examples ranging from gram-scale to potential industrial application. The absence of chlorinated solvents and corrosive gases aligns perfectly with increasingly stringent global environmental regulations, such as REACH and TSCA. This compliance future-proofs the manufacturing asset against regulatory tightening, ensuring long-term operational continuity and enhancing the brand reputation of the end-product as an eco-friendly material.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organotin-catalyzed synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their existing polymer production lines.
Q: What are the advantages of using DMC over DPC for polycarbonate synthesis?
A: Using Dimethyl Carbonate (DMC) instead of Diphenyl Carbonate (DPC) significantly lowers raw material costs. Furthermore, the byproduct methanol has a much lower boiling point than phenol, making it easier to remove via devolatilization, which simplifies the process and reduces energy consumption.
Q: What purity levels can be achieved with this organotin-catalyzed method?
A: The patented process utilizes a specific alcohol-water recrystallization technique that allows the refined intermediate DmC(1) to achieve a purity exceeding 99.8%, which is critical for producing high-molecular-weight polycarbonates with excellent optical and mechanical properties.
Q: Is this synthesis method environmentally friendly compared to traditional phosgene routes?
A: Yes, this method is a non-phosgene green synthesis route. It eliminates the use of highly toxic phosgene and corrosive dichloromethane solvents, thereby removing the generation of hazardous hydrogen chloride gas and significantly reducing environmental and safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethoxy Bisphenol A Diester Supplier
As the industry transitions towards greener polymer synthesis methodologies, having a partner with deep technical expertise in intermediate scale-up is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot data to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >99.8% purity benchmarks required for high-performance polycarbonate precursors. We understand the critical nature of supply continuity and maintain robust inventory management systems to support your long-term production goals.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your specific material requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our high-purity intermediates can enhance your final product performance while reducing your overall manufacturing costs.
