Advanced Titanium-Silicon Catalysts for Efficient Diphenyl Carbonate Production
The chemical industry is currently witnessing a paradigm shift in the synthesis of critical carbonate intermediates, driven by the urgent need for greener, more efficient manufacturing pathways. Patent CN110894204A introduces a groundbreaking advancement in this sector by disclosing a novel class of titanium-silicon organometallic catalysts designed specifically for the preparation of diphenyl carbonate compounds. Unlike traditional methods that rely on toxic phosgene or expensive oxidative carbonylation routes, this innovation leverages a transesterification mechanism that is both environmentally benign and economically superior. The core breakthrough lies in the unique structural integration of silicon and titanium centers, which creates a catalytic species capable of withstanding atmospheric conditions while delivering exceptional conversion rates exceeding 60%. For R&D directors and process engineers, this represents a significant leap forward in catalyst design, offering a robust solution that bridges the gap between laboratory efficiency and industrial reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of diphenyl carbonate has been plagued by significant technical and safety hurdles. The traditional phosgene method, while effective, involves the use of highly toxic reagents that pose severe environmental and occupational health risks, leading to increasingly stringent regulatory pressures. Alternatively, the oxidative carbonylation route suffers from the use of prohibitively expensive catalysts and notoriously low reaction selectivity, making it commercially unviable for many applications. Even within the greener transesterification domain, existing homogeneous catalysts like simple organic titanates exhibit poor stability; they are hypersensitive to moisture in the air, necessitating rigorous drying of all raw materials and complex inert atmosphere storage protocols. Furthermore, prior art catalysts often struggle to achieve high phenol conversion rates, typically hovering around 23% to 45%, which results in substantial amounts of unreacted starting material that must be recovered and recycled, thereby inflating energy consumption and operational costs.
The Novel Approach
The novel approach detailed in the patent fundamentally addresses these legacy issues through the engineering of a hybrid titanium-silicon framework. By polymerizing specific silicon compounds, such as hydroxyl-terminated polydimethylsiloxane or triphenylsilanol, with titanium alkoxides or halides, the inventors have created a catalyst that retains high Lewis acidity for catalysis while gaining the steric and electronic stability conferred by the siloxane backbone. This structural modification renders the catalyst remarkably stable in air, eliminating the need for extreme moisture control during storage and handling. More importantly, this new class of catalysts drives the equilibrium of the transesterification reaction much further towards completion, achieving phenol conversion rates of over 60% under liquid phase conditions. This enhancement not only boosts throughput but also simplifies the downstream separation process, as the ratio of product to unreacted phenol is significantly improved, offering a clear pathway for cost reduction in diphenyl carbonate manufacturing.
Mechanistic Insights into Ti-Si Catalyzed Transesterification
The efficacy of this catalytic system stems from the synergistic interaction between the titanium active center and the silicon-based ligand environment. In the transesterification reaction between phenol and dimethyl carbonate, the titanium atom acts as a Lewis acid, coordinating with the carbonyl oxygen of the carbonate to activate the electrophile for nucleophilic attack by the phenol. The presence of the silicon-oxygen framework modulates the electron density around the titanium, preventing rapid deactivation via hydrolysis or oligomerization, which are common failure modes for simple titanates. This stabilization allows the catalyst to maintain its active geometry throughout the prolonged reaction times required for high conversion, typically ranging from 9 to 11 hours at temperatures between 130°C and 180°C. The result is a highly selective transformation where the formation of unwanted byproducts like anisole is minimized, ensuring that the reaction pathway favors the sequential formation of methyl phenyl carbonate and ultimately diphenyl carbonate.
Impurity control is another critical aspect where this mechanistic design excels. In conventional systems, the generation of anisole via the methylation of phenol is a persistent side reaction that complicates purification and lowers yield. The specific steric bulk provided by the R groups on the silicon and titanium centers in the novel catalyst appears to suppress this side pathway, directing selectivity towards the desired carbonate esters. The patent data indicates a total selectivity for carbonate products reaching up to 99.9%, which is a remarkable achievement for a homogeneous catalytic system. This high selectivity implies that the crude reaction mixture requires less intensive fractional distillation to isolate high-purity diphenyl carbonate, directly translating to lower utility costs and reduced thermal degradation of the product. Understanding this mechanism is vital for scaling the process, as it confirms that the catalyst's performance is intrinsic to its molecular architecture rather than transient reaction conditions.
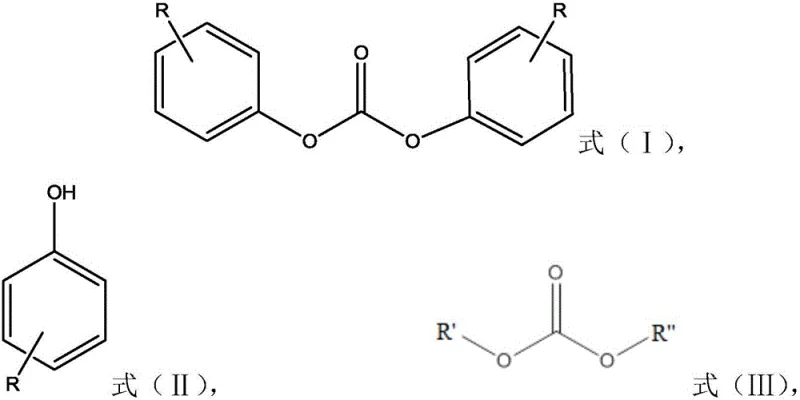
How to Synthesize Diphenyl Carbonate Efficiently
The synthesis protocol outlined in the patent provides a versatile framework adaptable to both solution and bulk polymerization techniques, offering flexibility for different production scales. The process begins with the precise preparation of the catalyst, where a titanium compound such as tetrabutyl titanate is contacted with a silicon compound like diphenylsilanediol. This polymerization step can be conducted in solvents like toluene or benzene at mild temperatures around 30°C, or alternatively as a bulk melt reaction, providing options to optimize solvent recovery or eliminate solvent use entirely. Once the catalyst is formed and purified via vacuum distillation to remove volatile alcohols, it is introduced to the phenol feedstock. The subsequent transesterification with dimethyl carbonate is managed through a controlled addition strategy, often involving the continuous removal of methanol byproduct to drive the equilibrium forward. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst by contacting a silicon compound (e.g., hydroxyl-terminated PDMS) with a titanium compound (e.g., tetrabutyl titanate) via solution or bulk polymerization.
- Mix phenol with the prepared catalyst and heat to the reaction temperature (130-180°C) under nitrogen protection.
- Add dimethyl carbonate dropwise while continuously removing methanol byproduct via distillation to drive equilibrium towards diphenyl carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this titanium-silicon catalyst technology offers profound strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain logistics associated with catalyst management. Because the catalyst exhibits exceptional stability in air, it removes the requirement for specialized, moisture-free packaging and climate-controlled warehousing that is mandatory for traditional titanate catalysts. This robustness significantly reduces the risk of catalyst degradation during transit and storage, ensuring that the material received at the manufacturing site performs exactly as specified without the need for pre-treatment or quality reassurance testing. Furthermore, the ability to recover and reuse the catalyst after the reaction cycle introduces a circular economy element to the production process, effectively lowering the recurring raw material expenditure per batch and insulating the operation from volatility in titanium precursor pricing.
- Cost Reduction in Manufacturing: The implementation of this catalyst leads to substantial cost savings through multiple mechanisms, primarily driven by the reduction in catalyst loading and the elimination of expensive co-catalysts. Traditional processes often require high loadings of tin or titanium catalysts combined with ionic liquids or amines to achieve acceptable conversion, whereas this novel system achieves superior performance with significantly lower titanium content. Additionally, the high selectivity towards diphenyl carbonate minimizes the formation of difficult-to-separate byproducts, reducing the energy intensity of the purification stage. The removal of the need for rigorous raw material drying further cuts down on utility costs associated with desiccation and inert gas purging, creating a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly enhanced by the air stability and ease of handling of the new catalyst. Unlike moisture-sensitive reagents that can spoil upon brief exposure to humid air, causing batch failures and production delays, this robust catalyst ensures consistent performance regardless of minor environmental fluctuations in the plant. This reliability reduces the frequency of production stoppages and quality deviations, allowing for more predictable scheduling and inventory management. Moreover, the raw materials required for the catalyst synthesis, such as common titanates and siloxanes, are widely available commodity chemicals, mitigating the risk of supply bottlenecks that often plague specialized or exotic catalytic systems.
- Scalability and Environmental Compliance: From a scalability perspective, the process is highly amenable to commercial scale-up of complex carbonate intermediates due to its operation in standard liquid-phase reactors without the need for high-pressure equipment typical of carbonylation routes. The transesterification conditions are moderate, utilizing standard heating media like oil baths, which simplifies reactor design and maintenance. Environmentally, the process aligns with green chemistry principles by avoiding toxic phosgene and generating methanol as the primary byproduct, which can be easily recovered and valorized. The reduced waste generation and lower energy footprint contribute to a smaller environmental impact, facilitating compliance with increasingly strict global environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a factual basis for evaluating the technology's fit for your specific production needs. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: How does the new Ti-Si catalyst improve upon traditional titanate catalysts?
A: Traditional titanate catalysts are highly sensitive to moisture, requiring strict drying of raw materials and inert storage. The novel Ti-Si catalyst described in CN110894204A exhibits exceptional air stability, maintaining catalytic activity even after 6 months of exposure, which drastically simplifies handling and storage logistics.
Q: What represent the key selectivity advantages of this transesterification process?
A: The process achieves a total selectivity of up to 99.9% for carbonate products (DPC and MPC). Crucially, it minimizes the formation of anisole, a common byproduct in phenol methylation, ensuring a cleaner product profile that reduces downstream purification costs.
Q: Is the catalyst reusable in industrial scale-up scenarios?
A: Yes, the patent explicitly details a recovery process where the catalyst can be separated from the reaction mixture and reused. This recyclability is a critical factor for reducing the overall cost of goods sold (COGS) in large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Carbonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced titanium-silicon catalyst technology in modernizing the production of high-value carbonate intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of diphenyl carbonate meets the exacting standards required for pharmaceutical and polymer applications. We are committed to leveraging this innovative catalytic chemistry to deliver products with superior quality profiles and consistent supply reliability.
We invite you to collaborate with us to explore how this technology can optimize your specific supply chain requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current production metrics, demonstrating the tangible economic benefits of switching to this greener, more efficient process. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market for fine chemical intermediates.
