Revolutionizing Oligonucleotide Delivery: Scalable GalNAc Phosphoramidite Technology for Commercial Production
The landscape of oligonucleotide therapeutics is undergoing a transformative shift, driven by the critical need for efficient hepatocyte targeting delivery systems. Central to this advancement is the technology disclosed in patent CN108738321B, which introduces a novel class of GalNAc phosphoramidite derivatives designed to streamline the synthesis of GalNAc-cluster oligonucleotide conjugates. Unlike traditional approaches that often require complex post-synthetic modifications, these derivatives function as direct building blocks within solid-phase oligonucleotide synthesis, thereby integrating the targeting moiety directly into the genetic sequence during the assembly process. This strategic integration not only enhances the pharmacokinetic profile of the resulting therapeutics by leveraging the affinity of GalNAc for sialoglycoprotein receptors on hepatocytes but also fundamentally alters the manufacturing economics by reducing unit operations. For global pharmaceutical developers, this represents a pivotal opportunity to access high-purity pharmaceutical intermediates that align with rigorous regulatory standards while optimizing the overall cost structure of antisense and siRNA drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of GalNAc-conjugated oligonucleotides has been plagued by inefficiencies inherent in multi-step synthetic strategies that separate the oligonucleotide assembly from the conjugation of the targeting ligand. Conventional methodologies typically involve synthesizing the oligonucleotide chain first, followed by a distinct and often cumbersome chemical conjugation step to attach the GalNAc cluster, which introduces significant risks of yield loss and impurity generation. This disjointed approach necessitates additional purification stages to remove unreacted ligands and side products, drastically increasing the consumption of solvents and chromatography media, which are major cost drivers in fine chemical manufacturing. Furthermore, the separate conjugation step often requires specialized reagents and conditions that may not be fully compatible with the delicate phosphodiester or phosphorothioate backbone of the oligonucleotide, potentially leading to degradation or incomplete reactions that compromise the therapeutic efficacy of the final drug substance.
The Novel Approach
In stark contrast, the innovative methodology outlined in the patent data proposes a unified synthesis strategy where the GalNAc phosphoramidite derivative serves as an integral monomer during the solid-phase synthesis cycle. By incorporating the GalNAc moiety as a phosphoramidite building block, the need for a separate conjugation reaction is entirely eliminated, allowing for the seamless construction of the GalNAc-cluster oligonucleotide conjugate in a single continuous flow. This consolidation of steps not only accelerates the production timeline but also significantly enhances the overall process mass intensity by reducing the number of isolation and purification events required. The structural design of these derivatives, featuring robust protecting groups and optimized linkers, ensures compatibility with standard automated synthesizers, thereby facilitating the reliable scale-up of complex pharmaceutical intermediates without the need for bespoke equipment or hazardous reaction conditions that typically hinder commercial adoption.
Mechanistic Insights into GalNAc Phosphoramidite Coupling
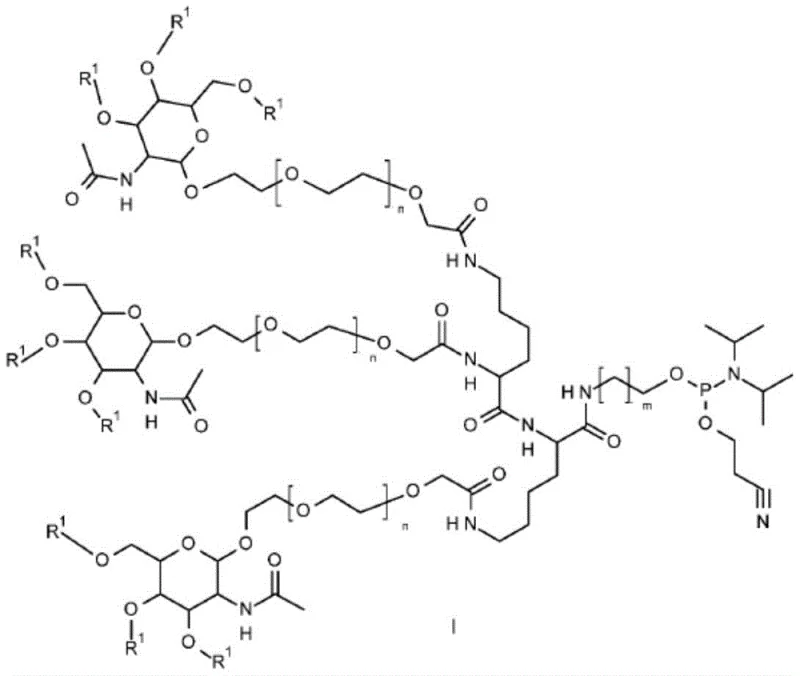
The chemical architecture of the GalNAc phosphoramidite derivatives described in Formula I is meticulously engineered to balance stability during synthesis with reactivity during the final coupling event. The core structure comprises a GalNAc sugar moiety linked via a polyethylene glycol or amino acid-based spacer to a phosphoramidite group, which is the active functionality for bond formation on the solid support. The presence of specific hydroxyl protecting groups, such as acetyl or benzoyl radicals, is critical for preventing unwanted side reactions at the sugar hydroxyls during the repetitive acidic and oxidative cycles of oligonucleotide synthesis. This precise protection strategy ensures that the GalNAc cluster remains intact and stereochemically pure throughout the elongation of the nucleotide chain, thereby guaranteeing the biological specificity of the final conjugate for liver cell uptake. The versatility of the linker region, defined by variables n and m, allows for fine-tuning of the spatial arrangement of the GalNAc clusters, which is a known determinant for binding affinity to the asialoglycoprotein receptor.
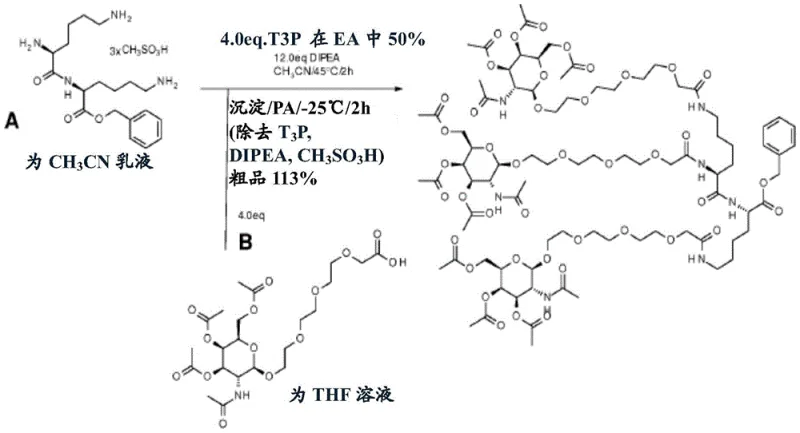
From a process chemistry perspective, the synthesis of these advanced intermediates relies on highly selective amide bond formation and phosphitylation reactions that are amenable to strict quality control. The preparation involves reacting a GalNAc acid derivative with a protected amine using coupling agents like N-propylphosphonic anhydride (T3P), which is favored for its ability to minimize racemization and facilitate easy workup by aqueous extraction. Subsequent deprotection steps, often involving catalytic hydrogenation with palladium on carbon, are employed to reveal the necessary hydroxyl groups for the final phosphitylation without affecting other sensitive functionalities within the molecule. This mechanistic pathway underscores the robustness of the chemistry, as it utilizes reagents and conditions that are well-understood in industrial organic synthesis, thereby reducing the technical risk associated with technology transfer from laboratory scale to commercial manufacturing facilities for high-purity oligonucleotide intermediates.
How to Synthesize GalNAc Phosphoramidite Efficiently
The practical implementation of this synthesis route requires adherence to precise stoichiometric controls and environmental conditions to maximize yield and purity. The process begins with the assembly of the GalNAc cluster backbone through peptide coupling, followed by the strategic removal of temporary protecting groups to expose the reactive hydroxyl handle. Detailed standardized synthesis steps see the guide below, which outlines the specific reagent grades, solvent drying protocols, and temperature profiles necessary to achieve the high diastereomeric purity required for therapeutic applications. Operators must ensure that all phosphitylation reactions are conducted under inert atmospheres to prevent hydrolysis of the sensitive phosphorus centers, and that final purification utilizes preparative chromatography techniques capable of resolving closely related phosphoramidite impurities.
- Coupling of GalNAc acid derivatives with protected amines using peptide coupling agents like T3P to form the amide backbone.
- Selective deprotection of hydroxyl groups via catalytic hydrogenation to expose reactive sites without damaging the GalNAc moiety.
- Final phosphitylation using chlorophosphoramidite reagents to generate the active phosphoramidite derivative for oligonucleotide assembly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this integrated GalNAc phosphoramidite technology offers profound strategic benefits that extend beyond mere technical feasibility. By consolidating the oligonucleotide synthesis and conjugation steps, manufacturers can achieve substantial cost savings through the reduction of raw material consumption, particularly expensive nucleoside phosphoramidites and coupling reagents that are otherwise lost during intermediate purifications. The elimination of a dedicated conjugation step also translates to a significantly reduced manufacturing footprint and lower utility costs, as fewer reactors and less solvent handling capacity are required to produce the same batch size of active pharmaceutical ingredient. This streamlining of the process directly contributes to cost reduction in pharmaceutical intermediates manufacturing, enabling more competitive pricing models for downstream drug developers who are under constant pressure to manage R&D expenditures while bringing life-saving therapies to market.
- Cost Reduction in Manufacturing: The unified synthesis approach drastically simplifies the production workflow by removing the need for isolated conjugation reactions, which traditionally account for a significant portion of the total processing time and resource allocation. By avoiding the isolation of intermediate oligonucleotide species prior to conjugation, the process minimizes material hold-up and reduces the volume of hazardous waste generated, leading to lower disposal costs and a smaller environmental footprint. Furthermore, the use of commercially available and scalable reagents such as T3P and standard hydrogenation catalysts ensures that the supply chain for raw materials remains stable and cost-effective, shielding production schedules from the volatility often associated with specialty custom reagents.
- Enhanced Supply Chain Reliability: The reliance on robust, well-established chemical transformations enhances the predictability of production timelines, as the risks associated with scaling novel or unproven conjugation chemistries are effectively mitigated. Since the GalNAc phosphoramidite acts as a standard building block, it can be stocked and quality-controlled independently, allowing for greater flexibility in production planning and inventory management. This modularity ensures that disruptions in one part of the synthesis do not necessarily halt the entire operation, thereby improving the overall resilience of the supply chain for high-purity oligonucleotide intermediates and ensuring consistent availability for clinical and commercial demands.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferred from laboratory glassware to large-scale industrial reactors without significant re-optimization. The preference for catalytic hydrogenation over stoichiometric reducing agents and the ability to perform aqueous workups for byproduct removal align with green chemistry principles, reducing the load on wastewater treatment facilities. This commitment to environmentally compliant manufacturing processes not only satisfies increasingly stringent regulatory requirements but also appeals to stakeholders prioritizing sustainability, making the technology a viable long-term solution for the commercial scale-up of complex polymer additives and therapeutic conjugates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of GalNAc phosphoramidite technology in oligonucleotide drug development. These insights are derived directly from the patented methodologies and are intended to clarify the operational advantages and quality parameters associated with this advanced chemical platform. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these building blocks into their existing synthesis workflows to achieve superior therapeutic outcomes.
Q: How does this GalNAc phosphoramidite improve oligonucleotide synthesis efficiency?
A: By functioning as a direct building block, it eliminates the need for a separate post-synthetic conjugation step, significantly simplifying the manufacturing workflow and reducing potential purification losses.
Q: What are the key stability considerations for these phosphoramidite derivatives?
A: The derivatives utilize robust hydroxyl protecting groups such as acetyl or benzoyl, ensuring stability during storage and handling while allowing for clean deprotection under standard oligonucleotide cleavage conditions.
Q: Is this technology scalable for commercial API intermediate production?
A: Yes, the synthesis relies on standard peptide coupling and hydrogenation techniques using reagents like T3P and Pd/C, which are well-established for large-scale industrial manufacturing and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GalNAc Phosphoramidite Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate complex patent technologies into reliable commercial supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from preclinical research to late-stage clinical trials is supported by uninterrupted material flow. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch of GalNAc phosphoramidite derivatives we produce. Our commitment to excellence ensures that the pharmaceutical intermediates we supply meet the exacting standards required for global regulatory submissions, providing our clients with the confidence needed to advance their oligonucleotide programs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to evaluate the technical merits of our GalNAc phosphoramidite solutions against your internal benchmarks. Let us collaborate to accelerate the development of next-generation RNA therapeutics through superior chemical innovation and supply chain reliability.
