Advanced Synthesis of Propynol Glycidyl Ether for High-Performance Industrial Applications
The chemical manufacturing landscape is constantly evolving, driven by the need for more efficient, environmentally compliant, and cost-effective synthesis routes for high-value intermediates. A pivotal development in this sector is documented in patent CN102936188A, which details a novel synthetic method for propynol glycidyl ether, a critical component in advanced electroplating and specialty chemical applications. This patent outlines a robust two-step process that fundamentally alters the traditional approach to ether synthesis, shifting away from corrosive acid catalysis towards a more controlled base-mediated reaction followed by specific hydrolysis. For R&D directors and procurement specialists, understanding the nuances of this technology is essential, as it represents a significant leap forward in process safety and yield optimization. The methodology described eliminates the need for volatile organic solvents, thereby streamlining the production workflow and reducing the environmental footprint associated with solvent recovery and disposal. Furthermore, the specific reaction conditions, operating between 30-60°C in the first stage and 40-70°C in the second, allow for precise thermal management that minimizes side reactions and polymerization risks. By adopting this technology, manufacturers can achieve conversion rates exceeding 90%, a metric that directly translates to reduced raw material waste and improved overall process economics. This report analyzes the technical merits and commercial implications of this synthesis route, providing a comprehensive overview for stakeholders looking to optimize their supply chain for high-purity specialty intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glycidyl ethers, including allyl and propynyl variants, has relied heavily on acid-catalyzed ring-opening reactions that pose significant challenges for large-scale manufacturing. As illustrated in the conventional reaction pathways, the process typically involves reacting an alcohol with epichlorohydrin under the influence of strong acids like sulfuric acid or Lewis acids such as tin chloride. 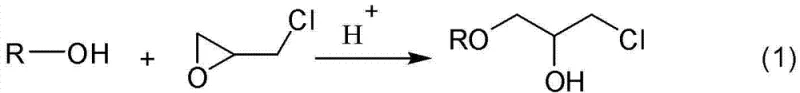
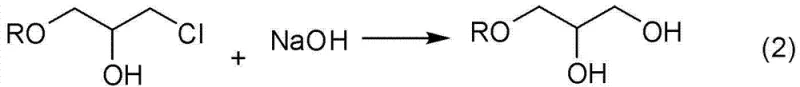 These acidic conditions are notoriously aggressive, leading to severe corrosion of production equipment which drastically shortens the service life of reactors and increases maintenance costs. Moreover, the presence of strong acids can induce unwanted polymerization of the propargyl group, resulting in reduced selectivity and the formation of difficult-to-remove byproducts that compromise the purity of the final intermediate. The subsequent cyclization step often requires precise stoichiometric control of sodium hydroxide, and any deviation can lead to incomplete conversion or the formation of chlorohydrin impurities. Additionally, many traditional methods necessitate the use of organic solvents to manage reaction exotherms and solubility, which introduces complex downstream separation processes and increases the volume of hazardous waste generated. The cumulative effect of these limitations is a process that is not only expensive to maintain due to equipment replacement but also difficult to scale without encountering significant safety and environmental regulatory hurdles. For supply chain managers, the reliance on corrosive catalysts also introduces variability in batch consistency, as equipment degradation over time can subtly alter reaction kinetics and heat transfer efficiency.
These acidic conditions are notoriously aggressive, leading to severe corrosion of production equipment which drastically shortens the service life of reactors and increases maintenance costs. Moreover, the presence of strong acids can induce unwanted polymerization of the propargyl group, resulting in reduced selectivity and the formation of difficult-to-remove byproducts that compromise the purity of the final intermediate. The subsequent cyclization step often requires precise stoichiometric control of sodium hydroxide, and any deviation can lead to incomplete conversion or the formation of chlorohydrin impurities. Additionally, many traditional methods necessitate the use of organic solvents to manage reaction exotherms and solubility, which introduces complex downstream separation processes and increases the volume of hazardous waste generated. The cumulative effect of these limitations is a process that is not only expensive to maintain due to equipment replacement but also difficult to scale without encountering significant safety and environmental regulatory hurdles. For supply chain managers, the reliance on corrosive catalysts also introduces variability in batch consistency, as equipment degradation over time can subtly alter reaction kinetics and heat transfer efficiency.
The Novel Approach
In stark contrast to the traditional acidic routes, the method disclosed in CN102936188A introduces a streamlined two-step process that prioritizes equipment longevity and reaction selectivity. 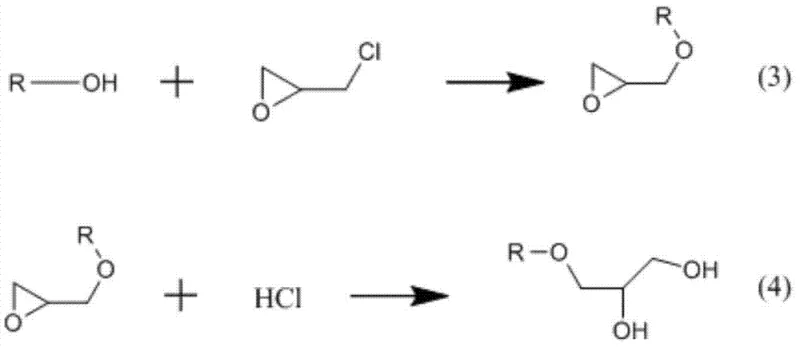 The first stage involves the direct etherification of propynol with epichlorohydrin using a sodium hydroxide aqueous solution as the catalyst, operating within a mild temperature range of 30-60°C. This base-catalyzed approach effectively bypasses the need for corrosive strong acids during the critical bond-forming step, thereby preserving the integrity of the stainless steel reactors and reducing the frequency of costly equipment replacements. The second stage utilizes a controlled hydrolysis with concentrated hydrochloric acid, but crucially, this is performed on the pre-formed intermediate rather than the raw starting materials, allowing for better management of the reaction exotherm and selectivity. By avoiding the addition of external organic solvents, the process simplifies the workup procedure, as the product can often be separated via phase separation or simple washing, significantly reducing the energy consumption associated with solvent distillation. The patent data indicates that this method achieves a conversion rate higher than 90%, demonstrating superior efficiency compared to the roughly 80% yields often seen in older acid-catalyzed literature. This novel approach not only enhances the purity profile of the propynol glycidyl ether but also aligns with modern green chemistry principles by minimizing waste generation and hazardous reagent usage. For technical teams, this represents a more robust and scalable platform that can be adapted for continuous flow processing or large-batch production with minimal modification to existing infrastructure.
The first stage involves the direct etherification of propynol with epichlorohydrin using a sodium hydroxide aqueous solution as the catalyst, operating within a mild temperature range of 30-60°C. This base-catalyzed approach effectively bypasses the need for corrosive strong acids during the critical bond-forming step, thereby preserving the integrity of the stainless steel reactors and reducing the frequency of costly equipment replacements. The second stage utilizes a controlled hydrolysis with concentrated hydrochloric acid, but crucially, this is performed on the pre-formed intermediate rather than the raw starting materials, allowing for better management of the reaction exotherm and selectivity. By avoiding the addition of external organic solvents, the process simplifies the workup procedure, as the product can often be separated via phase separation or simple washing, significantly reducing the energy consumption associated with solvent distillation. The patent data indicates that this method achieves a conversion rate higher than 90%, demonstrating superior efficiency compared to the roughly 80% yields often seen in older acid-catalyzed literature. This novel approach not only enhances the purity profile of the propynol glycidyl ether but also aligns with modern green chemistry principles by minimizing waste generation and hazardous reagent usage. For technical teams, this represents a more robust and scalable platform that can be adapted for continuous flow processing or large-batch production with minimal modification to existing infrastructure.
Mechanistic Insights into Base-Catalyzed Etherification and Hydrolysis
The core of this technological advancement lies in the mechanistic shift from electrophilic activation of the epoxide to nucleophilic activation of the alcohol. In the first step, the sodium hydroxide deprotonates the propynol to form a propynolate anion, which is a potent nucleophile capable of attacking the less hindered carbon of the epichlorohydrin epoxide ring. This SN2-type attack opens the epoxide ring to form a chlorohydrin ether intermediate, a reaction that is highly sensitive to temperature and base concentration. Maintaining the temperature between 30-60°C is critical; temperatures that are too low may result in sluggish kinetics and incomplete conversion, while temperatures exceeding this range could promote the polymerization of the acetylenic bond or the homopolymerization of epichlorohydrin. The molar ratio of sodium hydroxide to propynol, specified between 0.9 and 1.2 parts, ensures that there is sufficient base to drive the deprotonation without leaving excessive alkalinity that could trigger side reactions in the subsequent steps. The absence of a strong acid catalyst in this initial phase prevents the protonation of the epoxide oxygen, which would otherwise lead to a mixture of regioisomers and potential polymeric byproducts. This precise control over the nucleophilic substitution mechanism is what allows the process to achieve such high selectivity and conversion rates, ensuring that the majority of the starting material is funneled into the desired intermediate rather than waste.
The second mechanistic phase involves the cyclization or modification of the intermediate using dilute to concentrated hydrochloric acid, depending on the specific embodiment. In this stage, the chlorohydrin intermediate undergoes an intramolecular substitution or hydrolysis to finalize the glycidyl ether structure. The addition of water and hydrochloric acid at 40-70°C facilitates the removal of the chlorine atom and the formation of the final ether linkage, or in some interpretations of glycidyl synthesis, the closure of the epoxide ring if it was opened in a specific manner. The patent specifies using 0.1 to 0.4 parts of concentrated hydrochloric acid, a amount that is sufficient to catalyze the reaction without creating the highly corrosive environment seen in traditional one-pot acid methods. This step is crucial for impurity control, as the mild acidic conditions help to neutralize any residual base from the first step and prevent the degradation of the sensitive propynyl group. The cooling to room temperature post-reaction allows for the phase separation of the organic product from the aqueous salt layer, a physical separation that is far more efficient than the complex distillations required in solvent-heavy processes. By understanding these mechanistic details, R&D teams can better troubleshoot potential scale-up issues, such as heat removal during the exothermic neutralization or the optimization of stirring rates to ensure proper mass transfer between the aqueous and organic phases.
How to Synthesize Propynol Glycidyl Ether Efficiently
Implementing this synthesis route requires strict adherence to the molar ratios and temperature profiles outlined in the patent to ensure reproducibility and safety. The process begins with the precise metering of propynol and epichlorohydrin into a reactor equipped with efficient cooling capabilities to manage the exotherm upon base addition. Operators must monitor the dripping rate of the sodium hydroxide solution carefully to maintain the reaction temperature within the 30-60°C window, as deviations can impact the purity of the intermediate. Following the initial reaction, a thorough washing step is essential to remove inorganic salts before proceeding to the acid treatment phase, ensuring that the final product meets stringent purity specifications. The detailed standardized synthesis steps, including specific equipment setups and safety protocols, are provided in the technical guide below for qualified manufacturing partners.
- Mix propynol and epichlorohydrin (1: 0.8-1.2 molar ratio) and heat to 30-60°C while dripping sodium hydroxide solution.
- Wash the resulting propynol epoxypropane ether to remove impurities before the second reaction stage.
- React the intermediate with water and concentrated hydrochloric acid (0.1-0.4 molar ratio) at 40-70°C to finalize the ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits that extend beyond simple chemical yield, impacting the total cost of ownership and operational reliability. The elimination of corrosive acid catalysts in the primary reaction step significantly extends the lifespan of production vessels and piping, leading to substantial capital expenditure savings over the long term. By removing the need for organic solvents, the process reduces the volume of hazardous waste requiring disposal, thereby lowering environmental compliance costs and simplifying the regulatory reporting burden. The high conversion rate reported in the patent implies a more efficient use of raw materials, which stabilizes the cost of goods sold and reduces the volatility associated with raw material price fluctuations. Furthermore, the simplified workup procedure, which relies on phase separation rather than complex distillation, shortens the overall batch cycle time, allowing for increased production throughput without additional capital investment. These factors combine to create a more resilient supply chain capable of meeting demanding delivery schedules while maintaining competitive pricing structures.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the significant extension of equipment service life due to the avoidance of strong corrosive acids in the initial synthesis stage. Traditional methods often require frequent replacement of reactors and agitators due to acid corrosion, representing a hidden but substantial operational cost that is effectively eliminated here. Additionally, the solvent-free nature of the reaction removes the costs associated with purchasing, recovering, and disposing of large volumes of organic solvents, which are often subject to volatile market pricing and strict environmental taxes. The high conversion efficiency ensures that raw material utilization is maximized, meaning less feedstock is wasted as byproducts, further driving down the variable cost per kilogram of the final product. These cumulative savings allow for a more competitive pricing model without sacrificing margin, providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the robustness of the reaction conditions, which are less sensitive to minor fluctuations in temperature or reagent quality compared to highly acidic alternatives. The use of common, readily available reagents like sodium hydroxide and hydrochloric acid ensures that production is not bottlenecked by the supply of exotic or specialized catalysts that may have long lead times. The simplified purification process reduces the risk of batch failures due to complex separation issues, ensuring a more consistent output of saleable product that can be reliably promised to customers. This stability is crucial for maintaining long-term contracts with downstream users who require uninterrupted supply for their own manufacturing lines, such as electroplating facilities or specialty chemical formulators. By minimizing unplanned downtime for equipment maintenance or batch reworks, the overall availability of the product is significantly enhanced.
- Scalability and Environmental Compliance: Scalability is inherently supported by the exotherm management profile of the reaction, which operates at moderate temperatures (30-70°C) that are easily achievable in large-scale industrial reactors without requiring cryogenic cooling or high-pressure steam. The absence of solvents simplifies the scale-up equation, as there is no need to engineer complex solvent recovery loops or manage large volumes of flammable vapors, reducing the safety risk profile of the plant. From an environmental compliance perspective, the reduction in hazardous waste generation and the elimination of solvent emissions align with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory changes. This green chemistry approach not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable supplier, a factor that is becoming increasingly important in vendor selection criteria for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of propynol glycidyl ether, based on the data provided in the patent literature. These answers are designed to clarify the operational advantages and chemical properties of this specific manufacturing route for potential partners and technical evaluators. Understanding these details is critical for assessing the feasibility of integrating this intermediate into existing production workflows or new product formulations. The information provided reflects the specific embodiments and claims of the referenced intellectual property, ensuring accuracy and relevance for industrial applications.
Q: How does this synthesis method reduce equipment corrosion?
A: By avoiding strong acidic catalysts in the initial ring-opening step and utilizing a base-catalyzed etherification process, the method significantly reduces corrosive wear on production reactors.
Q: What is the expected conversion rate for this process?
A: According to patent data, the conversion rate for this synthesis method is consistently higher than 90%, ensuring high material efficiency.
Q: Is solvent required for this reaction?
A: No, the process is designed to be solvent-free, which simplifies the workup procedure and enhances environmental compliance by reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propynol Glycidyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthesis routes like the one described in CN102936188A for delivering high-performance specialty chemicals to the global market. Our CDMO capabilities are specifically engineered to handle complex organic syntheses, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from laboratory patent data to industrial reality requires rigorous process optimization, which is why our stringent purity specifications and rigorous QC labs are dedicated to ensuring every batch meets the exacting standards required for electroplating and fine chemical applications. Our team of expert chemists can adapt this base-catalyzed methodology to fit your specific volume requirements, ensuring that the benefits of reduced corrosion and high conversion are realized at scale.
We invite you to collaborate with us to optimize your supply chain for propynol glycidyl ether and related intermediates. By leveraging our technical expertise, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this more efficient synthesis route for your operations. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a stable, cost-effective, and high-quality supply of this critical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
