Advanced Synthesis of 5-Amino-1-Hydroxyethylpyrazole for Fourth-Generation Cephalosporins
Advanced Synthesis of 5-Amino-1-Hydroxyethylpyrazole for Fourth-Generation Cephalosporins
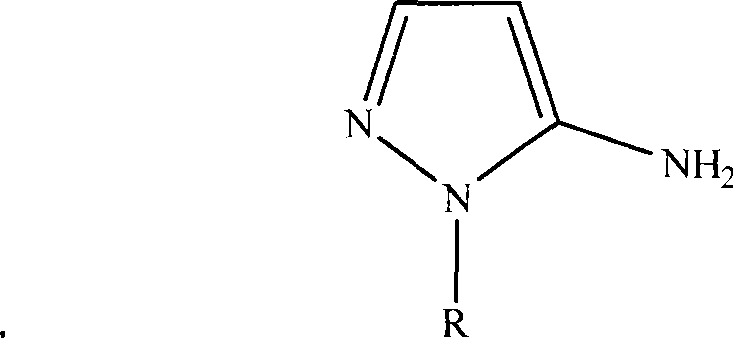
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates, particularly for fourth-generation cephalosporins like Cefoselis sulfate. Patent CN101367764A introduces a transformative synthesis process for 5-amino-1-hydroxyethylpyrazole and its analogs, which serve as the essential C-3 side chain for this potent antibiotic. This innovation addresses significant bottlenecks in the existing supply chain by replacing costly, specialized precursors with acrylonitrile, a ubiquitous bulk chemical. The strategic shift not only simplifies the operational workflow but also enhances the economic viability of producing high-purity pharmaceutical intermediates. By leveraging a sequence of substitution, cyclization, and elimination reactions, this method offers a reliable alternative for manufacturers aiming to optimize their production of beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-amino-1-hydroxyethylpyrazole has relied heavily on sophisticated starting materials such as 3-methoxyacrylonitrile, 3-methoxy-2-nitrile-acrylonitrile, or 3-methoxy-2-acetic acid oxymethyl-acrylonitrile. While these routes are chemically feasible, they present severe commercial disadvantages due to the high cost and limited availability of these specific nitrile derivatives. The dependency on such niche reagents creates a fragile supply chain, where price volatility and sourcing difficulties directly impact the final cost of the antibiotic Cefoselis sulfate. Furthermore, the nucleophilic substitution reactions required in these traditional pathways often demand stringent control to prevent side reactions, complicating the purification process and reducing overall process efficiency. These factors collectively hinder the widespread adoption and market expansion of this critical fourth-generation cephalosporin.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes acrylonitrile as the primary building block, fundamentally altering the economic landscape of this synthesis. Acrylonitrile is a globally produced commodity chemical, ensuring a stable supply and significantly lower procurement costs compared to methoxy-substituted alternatives. The new route employs a streamlined strategy where beta-hydroxyethylhydrazine reacts directly with acrylonitrile to form the pyrazoline core, bypassing the need for expensive pre-functionalized nitriles. This approach not only reduces the number of complex synthetic steps but also utilizes common solvents like toluene and standard reagents, making the process highly adaptable for large-scale industrial reactors. The result is a synthesis pathway that is both operationally convenient and economically superior, effectively lowering the barrier to entry for high-volume production.
Mechanistic Insights into Acrylonitrile-Based Cyclization and Elimination
The core of this innovative synthesis lies in a carefully orchestrated four-step mechanism that builds the pyrazole ring from simple precursors. Initially, sodium hydroxide and hydrazine hydrate are reacted to generate a reactive hydrazine species, which then undergoes nucleophilic substitution with a halogenated compound like chloroethanol at temperatures between 10°C and 55°C. This forms the critical beta-hydroxyethylhydrazine intermediate. Subsequently, this intermediate reacts with acrylonitrile in a Michael-type addition followed by intramolecular cyclization in toluene at elevated temperatures (60°C to 120°C). This step constructs the 5-amino-1-hydroxyethyl-5-pyrazoline skeleton, establishing the fundamental heterocyclic framework required for the final antibiotic side chain. The precise control of temperature and stoichiometry during this cyclization is vital to maximizing yield and minimizing oligomeric byproducts.
Following cyclization, the process employs a strategic tosylation step using p-toluenesulfonyl chloride in the presence of sodium bicarbonate and a phase transfer catalyst like sodium dodecylbenzenesulfonate. This transformation converts the hydroxyl group into a good leaving group (tosylate), activating the molecule for the final elimination reaction. The last stage involves treating the tosylated intermediate with sodium isopropoxide in isopropanol at 50°C to 100°C. This strong base facilitates an E2 elimination reaction, removing the tosyl group and aromatizing the pyrazoline ring to form the stable 5-amino-1-hydroxyethylpyrazole. This mechanistic pathway ensures high structural fidelity, with patent examples demonstrating intermediate purities of 98.5% and final product confirmation via NMR spectroscopy, guaranteeing the integrity of the pharmacophore.
How to Synthesize 5-Amino-1-Hydroxyethylpyrazole Efficiently
The synthesis of this critical cephalosporin side chain requires precise adherence to the four-step protocol outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the hydrazine intermediate, followed by the crucial cyclization with acrylonitrile which forms the heterocyclic core. Detailed operational parameters regarding temperature gradients, solvent ratios, and workup procedures are essential for reproducibility. For R&D teams looking to implement this route, understanding the specific nuances of the tosylation and elimination steps is key to avoiding common pitfalls such as incomplete conversion or impurity carryover. The standardized synthetic steps见下方的指南 provide a comprehensive roadmap for translating this laboratory-scale success into a robust manufacturing protocol.
- React sodium hydroxide and hydrazine hydrate, then add a halogenated compound (e.g., chloroethanol) at 10-55°C to form beta-hydroxyethylhydrazine.
- Cyclize the hydrazine intermediate with acrylonitrile in toluene at 60-120°C to form the pyrazoline ring structure.
- Perform tosylation using p-toluenesulfonyl chloride followed by elimination with sodium isopropoxide to yield the final aromatic pyrazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this acrylonitrile-based synthesis represents a significant opportunity to optimize the cost structure of antibiotic production. By eliminating the reliance on scarce and expensive methoxy-acrylonitrile derivatives, manufacturers can decouple their production costs from the volatility of niche chemical markets. The use of bulk commodities like acrylonitrile, toluene, and chloroethanol ensures that raw material availability is never a bottleneck, thereby enhancing supply chain resilience. Furthermore, the simplified reaction sequence reduces the consumption of solvents and energy per kilogram of product, contributing to substantial cost savings in utility and waste management. This economic efficiency allows for more competitive pricing of the final Cefoselis sulfate API, expanding market access.
- Cost Reduction in Manufacturing: The primary driver for cost reduction is the substitution of high-value specialty chemicals with low-cost bulk reagents. Acrylonitrile is produced on a massive global scale, offering a price advantage that is orders of magnitude lower than the traditional 3-methoxyacrylonitrile precursors. Additionally, the process avoids the use of expensive transition metal catalysts or complex protecting group strategies, which simplifies the downstream purification requirements. This reduction in material intensity and processing complexity translates directly into a lower cost of goods sold (COGS), enabling manufacturers to maintain healthy margins even in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for pharmaceutical supply chains, and this new route excels by utilizing universally available feedstocks. Unlike specialized nitriles that may have single-source suppliers or long lead times, acrylonitrile and chloroethanol are stocked by numerous chemical distributors worldwide. This diversity of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the chemical steps means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply of the intermediate. This reliability is essential for meeting the rigorous delivery schedules demanded by major pharmaceutical companies.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing standard unit operations such as reflux, filtration, and distillation that are easily replicated in multi-purpose chemical plants. The absence of hazardous heavy metals or extremely harsh reaction conditions simplifies the environmental compliance profile, reducing the burden on wastewater treatment facilities. The solid byproducts, primarily sodium salts, are easier to handle and dispose of compared to complex organic waste streams. This environmental friendliness, combined with the ease of scale-up from pilot batches to multi-ton production, makes this technology an attractive asset for contract development and manufacturing organizations (CDMOs) looking to expand their cephalosporin capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the technical specifications and experimental data provided in patent CN101367764A. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for commercial manufacturing. The insights provided here cover aspects ranging from raw material selection to final product quality control, ensuring a comprehensive understanding of the technology's value proposition.
Q: What are the primary cost advantages of the acrylonitrile route compared to traditional methods?
A: Traditional methods rely on expensive and hard-to-source precursors like 3-methoxyacrylonitrile. The new process utilizes acrylonitrile, a widely available bulk commodity chemical, which drastically reduces raw material costs and supply chain risks.
Q: How does this synthesis route ensure high purity for antibiotic applications?
A: The process incorporates a tosylation step using p-toluenesulfonyl chloride which acts as a purification checkpoint. Experimental data from the patent indicates that intermediates can achieve purities exceeding 96% before the final elimination step, ensuring a high-quality final API side chain.
Q: Is this process scalable for industrial production of Cefoselis sulfate side chains?
A: Yes, the reaction conditions utilize standard unit operations such as reflux, filtration, and distillation without requiring exotic catalysts or extreme pressures. This simplicity facilitates straightforward scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-1-Hydroxyethylpyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the acrylonitrile-based route described in CN101367764A and possesses the expertise to execute this chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 5-amino-1-hydroxyethylpyrazole meets the exacting standards required for fourth-generation cephalosporin synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective technology for their API production. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your antibiotic portfolio.
