Advanced Synthesis of Ethylene Glycol Diaryl Ether: Mechanism, Scalability, and Commercial Viability
Advanced Synthesis of Ethylene Glycol Diaryl Ether: Mechanism, Scalability, and Commercial Viability
The global demand for high-performance functional materials, particularly in the thermal recording and polymer sectors, relies heavily on the consistent supply of specialized organic intermediates. Patent CN1237040C introduces a groundbreaking preparation method for ethylene glycol diaryl ether, a critical structural motif found in sensitizers for thermal recording materials and various flame retardants. This technology represents a significant departure from conventional synthetic routes by leveraging the principles of neighboring group participation to overcome historical yield limitations. For R&D directors and procurement specialists seeking a reliable ethylene glycol diaryl ether supplier, understanding the mechanistic underpinnings of this patent is essential. The process achieves yields exceeding 80% and purity levels greater than 99% through a streamlined operation that utilizes inexpensive raw materials like dichloroethane and potassium hydroxide. By shifting the paradigm from equilibrium-driven excess phenol methods to kinetic control via cyclic intermediates, this invention offers a robust pathway for the commercial scale-up of complex organic ethers.
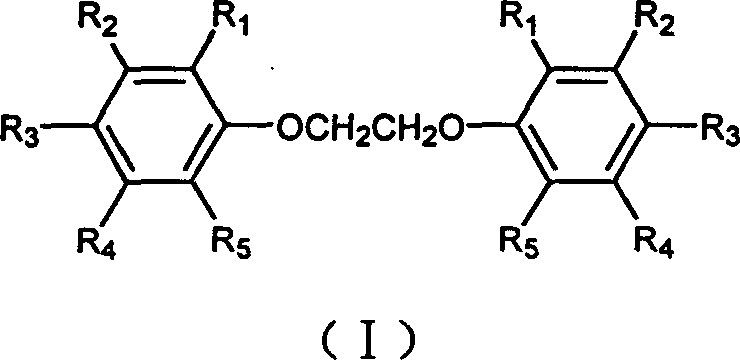
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethylene glycol diaryl ethers has been plagued by thermodynamic and kinetic challenges that severely impact industrial viability. Conventional wisdom dictated that to drive the nucleophilic substitution of dichloroethane by phenoxide ions to completion, a large excess of phenol was required to shift the equilibrium towards the product. However, this approach created significant downstream bottlenecks; recovering the unreacted, often expensive phenol required energy-intensive distillation and complex separation protocols, which drastically increased operational expenditures. Furthermore, alternative methods involving stepwise addition of alkali attempted to control free base concentration to minimize side reactions such as the elimination of dichloroethane or hydroxyl substitution. While these methods marginally improved yields to around 72%, they introduced excessive operational complexity, requiring precise, long-term monitoring of base concentrations that is difficult to maintain in large-scale reactors. Consequently, the industry has long suffered from inconsistent batch quality and suboptimal resource utilization in the manufacturing of these vital fine chemical intermediates.
The Novel Approach
The methodology described in CN1237040C fundamentally reimagines the reaction stoichiometry and mechanism to bypass these traditional pitfalls. Instead of relying on excess phenol, this novel approach employs an excess of dichloroethane, a significantly cheaper and more volatile reagent that is easier to manage and recover. The core innovation lies in the recognition that the reaction does not proceed via a simple bimolecular nucleophilic substitution but rather through a highly reactive cyclic oxonium intermediate. By utilizing a phase transfer catalyst, specifically polyethylene glycol, the process accelerates the rate-determining first substitution step, ensuring that the dichloroethane is efficiently converted before it can undergo degradation via elimination pathways. This strategic shift allows for a simplified workup procedure where inorganic salts are easily washed away with water, leaving a high-purity residue that requires only recrystallization. For procurement managers focused on cost reduction in fine chemical intermediates manufacturing, this transition from complex recovery systems to simple filtration and crystallization represents a substantial opportunity for margin improvement.
Mechanistic Insights into Neighboring Group Participation
To fully appreciate the technical superiority of this synthesis, one must delve into the specific reaction kinetics governed by the neighboring group participation mechanism. As illustrated in the reaction scheme, the process begins with the deprotonation of the substituted phenol by a strong base like potassium hydroxide to form the phenoxide anion. This anion attacks one end of the dichloroethane molecule in a slow, rate-determining step to form a monosubstituted intermediate. Crucially, unlike traditional models where this intermediate might accumulate or revert, the lone pair electrons on the ether oxygen atom immediately participate in an intramolecular nucleophilic attack on the adjacent carbon bearing the remaining chlorine atom. This internal displacement generates a strained, three-membered cyclic oxonium ion, which possesses exceptionally high electrophilic character. This high-energy intermediate is instantly attacked by a second molecule of phenoxide anion, ring-opening the structure to yield the final ethylene glycol diaryl ether product. This cascade ensures that the monosubstituted species never accumulates to detectable levels, effectively driving the reaction to completion even with non-stoichiometric ratios of phenol.
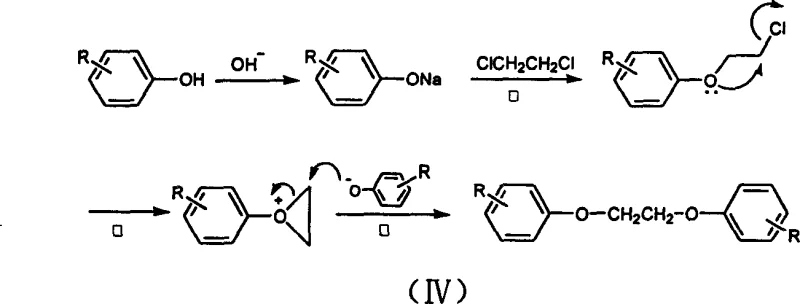
The implications of this mechanistic pathway for impurity control are profound and directly address the concerns of quality assurance teams. In conventional syntheses, the presence of free hydroxide ions often leads to the hydrolysis of dichloroethane into ethylene glycol or its elimination into vinyl chloride, creating difficult-to-separate byproducts that contaminate the final API or intermediate. However, in this patented process, the rapid conversion of the monosubstituted intermediate into the cyclic oxonium ion outcompetes these degradation pathways. Furthermore, the use of potassium hydroxide instead of sodium hydroxide is a deliberate choice; the resulting potassium chloride byproduct exhibits superior solubility in the aqueous wash steps compared to sodium chloride, ensuring that residual ash content in the final organic product is minimized. This meticulous attention to ionic solubility and reaction kinetics results in a crude product with purity already exceeding 99% prior to final recrystallization, thereby reducing the burden on purification units and ensuring a consistent impurity profile suitable for sensitive applications like thermal paper coatings.
How to Synthesize Ethylene Glycol Diaryl Ether Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing multipurpose chemical reactors without requiring exotic equipment. The process begins by charging the reactor with the substituted phenol and an aqueous alkali solution, typically potassium hydroxide, followed by the addition of the phase transfer catalyst and the excess dichloroethane. The mixture is then heated to reflux temperatures between 70°C and 100°C, where the phase transfer catalyst facilitates the transport of phenoxide ions into the organic phase to react with the dichloroethane. Following the initial reaction period, the system is subjected to a concentration step at elevated temperatures (95-130°C) to remove water and drive the equilibrium towards the final product by increasing the concentration of the reactive species.
- React substituted phenol with alkali solution (preferably KOH) to form phenoxide, then add excess dichloroethane and a phase transfer catalyst like PEG.
- Reflux the mixture at 70-100°C for 15-35 hours to allow the neighboring group participation mechanism to drive the formation of the cyclic oxonium intermediate.
- Concentrate the reaction at 95-130°C to remove water and complete the reaction, followed by aqueous workup and recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need to recover large quantities of unreacted phenol, manufacturers can significantly reduce energy consumption associated with distillation and lower the labor costs linked to complex separation operations. This streamlining of the production process translates directly into a more competitive cost structure, allowing suppliers to offer high-purity ethylene glycol diaryl ether at more attractive price points while maintaining healthy margins. Additionally, the reliance on commodity chemicals like dichloroethane and potassium hydroxide, rather than specialized or scarce reagents, enhances supply chain resilience against raw material volatility. This stability is crucial for long-term contracting and ensures that production schedules remain uninterrupted even during periods of market fluctuation.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of expensive, hard-to-recover phenol with inexpensive, volatile dichloroethane. Since dichloroethane is used in excess but is cheap and easily removed, the overall material cost per kilogram of product is significantly lowered. Furthermore, the high selectivity of the neighboring group participation mechanism minimizes the formation of waste byproducts, reducing the costs associated with waste treatment and disposal. The elimination of complex phenol recovery columns also lowers capital expenditure requirements for new production lines, making it a highly efficient choice for capacity expansion.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this reaction conditions ensures consistent batch-to-batch reproducibility. The tolerance for slight variations in reagent ratios, afforded by the excess dichloroethane strategy, reduces the risk of batch failures due to minor weighing errors or feed inconsistencies. This reliability is paramount for customers in the thermal recording and polymer industries who require just-in-time delivery of materials with strict specification adherence. The simplified workup also shortens the overall cycle time per batch, effectively increasing the throughput of existing reactor assets and reducing lead times for order fulfillment.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than traditional methods. The use of potassium hydroxide generates potassium chloride as a byproduct, which, unlike some organic wastes, can be purified and potentially utilized in other industries such as agriculture, aligning with green chemistry principles. The absence of heavy metal catalysts or toxic solvents simplifies regulatory compliance and reduces the burden on environmental health and safety teams. Moreover, the straightforward nature of the reaction makes it highly scalable from pilot plant quantities to multi-ton commercial production without the need for extensive re-optimization, facilitating rapid market entry for new derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ethylene glycol diaryl ethers based on the patented technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding sourcing and formulation strategies.
Q: Why does this method use excess dichloroethane instead of excess phenol?
A: Traditional methods use excess phenol to drive equilibrium, but this complicates purification. This patent utilizes a neighboring group participation mechanism where the monosubstituted intermediate rapidly forms a reactive cyclic oxonium ion. Using excess dichloroethane compensates for side reactions without generating hard-to-remove phenolic impurities, significantly simplifying downstream processing.
Q: What is the role of the phase transfer catalyst in this synthesis?
A: The phase transfer catalyst, specifically polyethylene glycol (PEG), is critical because the initial nucleophilic substitution between the phenoxide and dichloroethane has a high activation energy and is the rate-determining step. The catalyst facilitates the interaction between the phases, accelerating this slow step and preventing the consumption of dichloroethane via elimination side reactions.
Q: How does this process improve supply chain reliability for thermal recording materials?
A: By achieving yields greater than 80% with purity exceeding 99% through a simple aqueous workup, this method eliminates the need for complex distillation columns required to recover excess phenol. This reduces production lead time and equipment dependency, ensuring a more consistent and reliable supply of high-quality sensitizers for the thermal paper industry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethylene Glycol Diaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications promised by CN1237040C are consistently met in our facilities. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest international standards. Our commitment to quality assurance means that every kilogram of ethylene glycol diaryl ether we supply is backed by comprehensive data packages, giving your R&D and production teams the confidence to integrate our materials into your critical formulations without delay.
We invite you to leverage our technical expertise to optimize your supply chain for thermal recording materials and polymer additives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your growth and innovation goals in the global fine chemicals market.
