Optimizing Antidepressant Production: A Technical Analysis of the Novel Moclobemide Synthesis Route
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antidepressants like Moclobemide, a reversible inhibitor of monoamine oxidase A (RIMA). Patent CN1052977C introduces a transformative methodology that addresses long-standing inefficiencies in the commercial production of this active pharmaceutical ingredient. By shifting away from complex distillation requirements and hazardous reagents, this invention establishes a new benchmark for process reliability. The core innovation lies in the strategic utilization of 2-aminoethyl hydrogen sulfate as a starting material, which reacts with p-chlorobenzoyl chloride to form a stable, isolable solid intermediate. This approach not only streamlines the synthetic sequence but also fundamentally alters the economic landscape of manufacturing by reducing unit operations. For R&D directors and process chemists, this patent represents a pivotal shift towards greener, more manageable chemistry that maintains high purity standards without compromising on throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Moclobemide has been plagued by significant technical hurdles that inflate production costs and complicate supply chain logistics. Traditional routes often rely on N-(2-amino-ethyl) morpholine reacting with acid chlorides, a process that necessitates rigorous molecular distillation to purify the amine starting material. This distillation step is energy-intensive and requires specialized equipment, creating a bottleneck for large-scale operations. Furthermore, alternative pathways utilizing chlorobenzoyl nitrogen azoles introduce severe safety liabilities due to the carcinogenic nature of the reagents, posing unacceptable risks to labor protection and environmental compliance. Other methods involving chloro-N-(2-chloroethyl) benzamide suffer from inherently low yields, rendering them economically unviable for commercial adoption. These legacy processes collectively contribute to extended lead times and increased operational expenditures, forcing procurement teams to manage higher inventory buffers to mitigate production uncertainties.
The Novel Approach
The methodology disclosed in CN1052977C circumvents these historical bottlenecks by introducing a two-step sequence centered around a unique sulfate ester intermediate. Instead of struggling with difficult liquid amines, the process utilizes 2-aminoethyl hydrogen sulfate, a commercially accessible and stable reagent. The reaction proceeds in an alkaline aqueous solution, generating 2-p-chlorobenzamido ethyl sulfate sodium salt as a solid precipitate. This physical state change is critical; it allows for simple filtration to remove impurities, effectively replacing complex distillation or chromatographic purification steps. The subsequent reaction with morpholine occurs under reflux conditions, driving the nucleophilic substitution to completion with high efficiency. This novel approach transforms the production profile from a hazardous, low-yield operation into a streamlined, high-throughput process suitable for modern GMP facilities, directly addressing the need for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Aqueous Amidation and Nucleophilic Substitution
The first stage of this synthesis involves a Schotten-Baumann type amidation conducted in a biphasic or aqueous alkaline environment. The 2-aminoethyl hydrogen sulfate acts as the nucleophile, attacking the electrophilic carbonyl carbon of the p-chlorobenzoyl chloride. The presence of sodium hydroxide serves a dual purpose: it neutralizes the hydrochloric acid byproduct generated during the acylation and maintains the amine in its reactive, unprotonated form locally at the interface. The resulting product, 2-p-chlorobenzamido ethyl sulfate sodium salt, possesses limited solubility in the cold alkaline medium, prompting its precipitation. This precipitation is a key thermodynamic driver that pushes the equilibrium forward and simultaneously purifies the intermediate. By isolating this solid, the process effectively sequesters the reactive functionality, preventing side reactions such as hydrolysis of the acid chloride or polymerization, which are common pitfalls in liquid-phase syntheses.
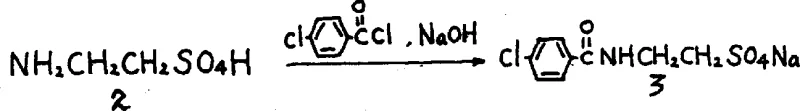
In the second mechanistic phase, the isolated sulfate ester intermediate undergoes a nucleophilic substitution reaction with morpholine. The sulfate group acts as an excellent leaving group, superior to halides in many contexts due to its stability and solubility characteristics in the reaction matrix. Under elevated temperatures ranging from 50°C to 140°C, the morpholine nitrogen attacks the ethyl chain, displacing the sulfate moiety. This step is kinetically favored by the excess of morpholine, which acts both as a reagent and a solvent base. The transition from the solid sulfate salt to the final Moclobemide structure is clean, with minimal formation of bis-alkylated byproducts. The robustness of this substitution ensures that the final crude product requires only a simple ethanol-water recrystallization to meet stringent pharmacopeial standards, demonstrating exceptional control over the impurity profile throughout the synthetic trajectory.
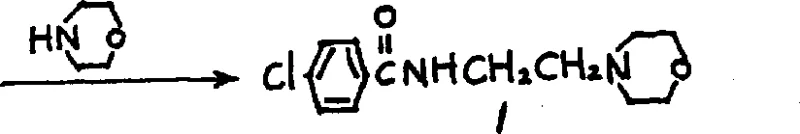
How to Synthesize Moclobemide Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the yield of the solid intermediate and ensure complete conversion in the second step. The process begins with the careful addition of p-chlorobenzoyl chloride to a cooled mixture of the amino sulfate and base, maintaining the temperature between 0°C and 25°C to prevent thermal degradation. Following the isolation of the intermediate, the subsequent reaction with morpholine is conducted under reflux to provide the necessary activation energy for the displacement. Detailed standardized operating procedures regarding specific mixing rates, quenching protocols, and crystallization parameters are essential for reproducibility. For a comprehensive guide on the exact molar ratios and workup procedures validated by the patent data, please refer to the technical instructions below.
- React 2-aminoethyl hydrogen sulfate with p-chlorobenzoyl chloride in an alkaline aqueous solution at 0-25°C to form the solid sodium salt intermediate.
- Filter the crude solid intermediate and react it with excess morpholine at elevated temperatures (50-140°C) to effect nucleophilic substitution.
- Precipitate the final product in water, filter, and recrystallize from ethanol-water to obtain high-purity Moclobemide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for supply chain resilience and cost management. The elimination of molecular distillation equipment drastically reduces capital expenditure (CapEx) requirements for manufacturing facilities, allowing for faster deployment of production lines. Furthermore, the reliance on aqueous media rather than expensive anhydrous organic solvents significantly lowers the variable costs associated with solvent purchase, recovery, and disposal. The simplicity of the workup—primarily filtration and recrystallization—reduces the manpower and time required per batch, thereby increasing overall facility throughput. These factors combine to create a highly competitive cost structure that can withstand market fluctuations in raw material pricing, ensuring a stable supply of high-purity pharmaceutical intermediates for downstream API manufacturers.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the removal of energy-intensive purification steps. By generating a solid intermediate that can be purified via filtration, the process eliminates the need for high-vacuum distillation columns and the associated energy consumption. Additionally, the use of water as the primary reaction medium for the first step removes the cost burden of drying and recycling large volumes of organic solvents. This shift to aqueous chemistry also mitigates the risk of solvent-related safety incidents, potentially lowering insurance premiums and compliance costs. Consequently, the overall cost of goods sold (COGS) is substantially reduced, providing a distinct margin advantage in the competitive antidepressant market.
- Enhanced Supply Chain Reliability: The raw materials utilized in this pathway, specifically 2-aminoethyl hydrogen sulfate and p-chlorobenzoyl chloride, are commodity chemicals with robust global supply chains. Unlike specialized reagents that may face sourcing bottlenecks, these inputs are readily available from multiple vendors, reducing the risk of supply disruption. The high yield per step, consistently reported above 80%, ensures that material throughput is predictable and efficient. This predictability allows supply chain managers to optimize inventory levels and reduce safety stock requirements. The ability to produce the intermediate as a stable solid also offers logistical flexibility, as it can potentially be stored or transported if multi-site manufacturing strategies are employed, further de-risking the supply network.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges, but this aqueous-based method is inherently scalable due to the high heat capacity of water. The exothermic nature of the amidation step is easily managed in large reactors, preventing thermal runaways that could compromise safety or quality. Moreover, the process aligns with green chemistry principles by minimizing the use of volatile organic compounds (VOCs) and avoiding carcinogenic reagents. This environmental profile simplifies the permitting process for new facilities and ensures long-term compliance with increasingly stringent environmental regulations. The waste stream, primarily consisting of inorganic salts and water, is easier to treat than complex organic solvent mixtures, reducing the operational burden on wastewater treatment plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Moclobemide synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering clarity on why this method is superior to legacy technologies. Understanding these nuances is critical for technical teams evaluating process transfer feasibility and for procurement officers assessing supplier capabilities. The answers highlight the specific operational improvements that translate into tangible business value.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: Unlike conventional methods requiring difficult molecular distillation or utilizing carcinogenic reagents, this route employs readily available raw materials and generates a solid intermediate that is easily purified by filtration, significantly simplifying the operational workflow.
Q: How does this method improve safety and environmental compliance?
A: The process avoids the use of toxic mustard-structure intermediates and carcinogenic azole derivatives found in prior art, while utilizing an aqueous reaction medium that reduces the reliance on volatile organic solvents, thereby enhancing labor protection and environmental safety.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is specifically designed for scalability, featuring high yields per step (greater than 80%) and robust reaction conditions that allow for straightforward temperature control and product isolation, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moclobemide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the pharmaceutical sector. Our technical team has extensively analyzed the pathway described in CN1052977C and possesses the expertise to implement this aqueous-based strategy at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling exothermic aqueous reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Moclobemide meets the highest international standards.
We invite potential partners to engage with us to explore how this optimized synthesis can enhance your supply chain efficiency. By leveraging our technical capabilities, you can achieve significant operational improvements and secure a stable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate our commitment to quality and reliability as your trusted partner in fine chemical manufacturing.
