Advanced Synthesis of 1,4,6,10-Tetradouble-Bond Pentadecyl Phosphonate for Scalable Lycopene Manufacturing
The global demand for high-purity carotenoids, particularly lycopene, continues to surge driven by its proven efficacy in scavenging free radicals, anti-aging applications, and potential in oncology and cardiovascular health. However, the industrial synthesis of this vital nutrient has historically been constrained by complex multi-step pathways involving hazardous reagents and difficult purification protocols. Patent CN102140117B introduces a transformative approach to this challenge by detailing the preparation of a novel key intermediate, 1,4,6,10-tetradouble-bond pentadecyl phosphonate, and its subsequent conversion into lycopene. This technology represents a significant leap forward for any reliable lycopene intermediate supplier seeking to optimize their manufacturing footprint. By shifting away from traditional triphenylphosphine-based Wittig reactions, this method offers a cleaner, more direct route that aligns perfectly with modern green chemistry principles while maintaining the rigorous stereochemical control required for pharmaceutical and nutraceutical grade products.
Historically, the industrial production of lycopene has been dominated by a few major players utilizing routes characterized by the use of expensive and toxic triphenylphosphine, as seen in early developments by Roche and BASF. While effective, these conventional methods generate substantial phosphine oxide waste, creating significant environmental disposal burdens and inflating operational costs. Alternative approaches, such as those described in WO 0031086, attempted to utilize allenyl phosphonates but introduced new bottlenecks, specifically the technically arduous selective reduction of an allenyl group to a specific double bond configuration. This reduction step is notoriously difficult to control on a large scale, often leading to inconsistent isomer ratios and reduced overall yields. The novel approach presented in the patent data circumvents these legacy issues entirely by employing a Wittig-Horner strategy that utilizes a stable phosphonate ester, thereby eliminating the need for difficult reduction steps and toxic phosphines.
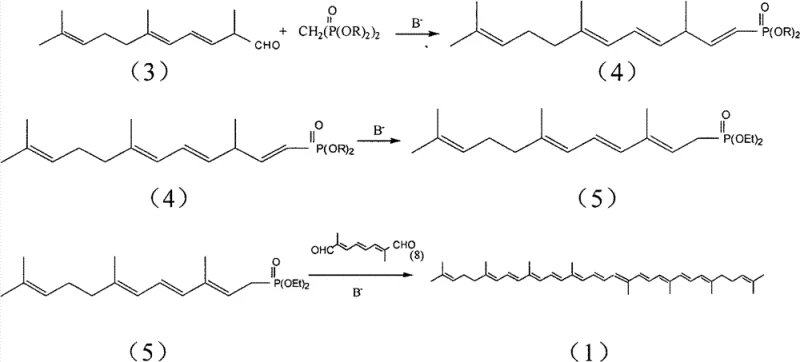
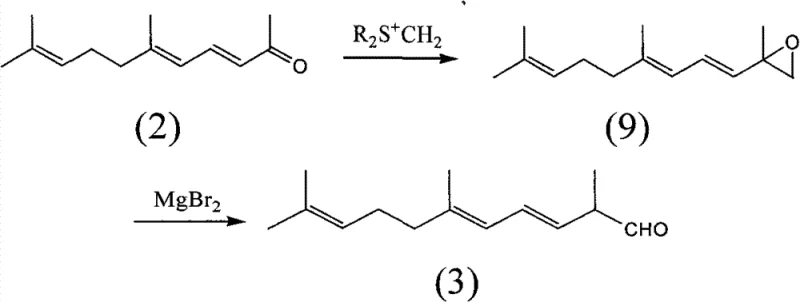
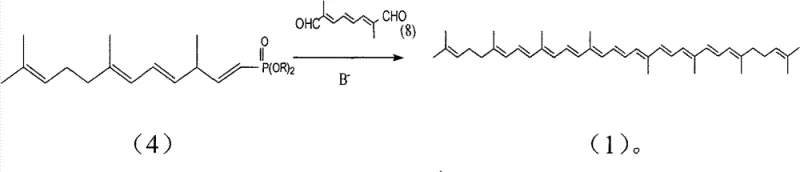
The mechanistic elegance of this new pathway lies in its strategic use of a C-14 aldehyde precursor derived directly from pseudoionone, a readily available bulk chemical. The process initiates with the reaction of pseudoionone with a sulfonium salt to generate an epoxide intermediate, which is subsequently rearranged using magnesium bromide to yield the critical C-14 aldehyde. This aldehyde then undergoes a Wittig-Horner condensation with tetraethyl methylene diphosphonate under mild basic conditions to form the target 1,4,6,10-tetradouble-bond pentadecyl phosphonate. This specific intermediate is pivotal because it possesses the correct carbon skeleton and double bond positioning to couple directly with a C-10 dialdehyde in the final step. The final coupling involves a base-catalyzed rearrangement and condensation that efficiently constructs the full conjugated polyene system of lycopene with high stereoselectivity for the all-trans isomer.
From a process chemistry perspective, the control of reaction conditions is paramount to ensuring high purity and yield. The patent specifies that the Wittig-Horner condensation to form the phosphonate intermediate should be conducted at temperatures ranging from 0°C to 30°C, preferably between 10°C and 20°C, using bases such as sodium hydride, potassium tert-butoxide, or n-butyllithium. Solvent selection is equally critical, with polar aprotic solvents like DMSO or DMF, or ethers like THF, providing the optimal environment for carbanion generation and stability. The final coupling step to produce lycopene requires precise temperature management, typically between -40°C and 30°C, to facilitate the necessary rearrangement of the phosphonate prior to condensation. This level of control ensures that the final product exhibits the characteristic 13 peaks in the 120-140 ppm range of the 13C NMR spectrum, confirming the desired all-trans geometry essential for biological activity.
How to Synthesize 1,4,6,10-Tetradouble-Bond Pentadecyl Phosphonate Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and scalability, making it an ideal candidate for commercial adoption. The process begins with the preparation of the C-14 aldehyde from pseudoionone, followed by the key phosphonate formation step. Detailed standard operating procedures for each stage, including specific molar ratios, solvent volumes, and workup protocols, are essential for reproducibility. For laboratories and pilot plants aiming to implement this technology, adhering to the specified inert atmosphere conditions and temperature profiles is crucial to prevent side reactions and ensure maximum conversion. The standardized synthesis steps outlined below provide a roadmap for achieving the high purity levels demanded by the fine chemical industry.
- React pseudoionone with a sulfonium salt to form an epoxide intermediate, followed by ring-opening with magnesium bromide to yield the C-14 aldehyde.
- Perform a Wittig-Horner condensation between the C-14 aldehyde and tetraethyl methylene diphosphonate in the presence of a base to generate the 1,4,6,10-tetradouble-bond pentadecyl phosphonate.
- Execute a base-catalyzed rearrangement and subsequent condensation with a C-10 dialdehyde to finalize the synthesis of all-trans lycopene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver for cost reduction in carotenoid manufacturing with this technology is the substitution of expensive, hazardous reagents with commodity chemicals. By eliminating triphenylphosphine, manufacturers avoid not only the high purchase price of the reagent but also the significant downstream costs associated with the removal and disposal of triphenylphosphine oxide byproducts. This simplification of the waste stream translates directly into lower environmental compliance costs and reduced processing time, enhancing the overall throughput of the facility without requiring capital-intensive new equipment.
- Cost Reduction in Manufacturing: The reliance on pseudoionone and tetraethyl methylene diphosphonate as starting materials leverages existing global supply chains for bulk industrial chemicals, ensuring price stability and availability. Unlike specialized reagents that may be subject to supply volatility, these inputs are produced at massive scales for other industries, providing a buffer against market fluctuations. Furthermore, the elimination of the difficult selective reduction step found in prior art reduces the consumption of reducing agents and catalysts, while simultaneously lowering energy requirements by shortening the overall reaction sequence. This streamlined process flow results in substantial cost savings per kilogram of final product, improving margin potential for high-volume producers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain continuity. The tolerance of the Wittig-Horner reaction to slight variations in temperature and the use of common solvents like THF and toluene mean that production is less susceptible to stoppages caused by strict environmental controls or specialized utility requirements. Additionally, the high yields reported in the patent examples, often exceeding 80-90% for the intermediate steps, minimize the need for extensive recycling of unreacted materials. This efficiency ensures a predictable output rate, allowing supply chain planners to commit to tighter delivery schedules with greater confidence and reducing the safety stock levels required to buffer against production variability.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of highly exothermic or hazardous steps that typically complicate scale-up. The reaction exotherms are manageable, and the workup procedures involve standard aqueous washes and crystallizations that are easily adapted to large-scale reactors. From an environmental standpoint, the reduction in phosphorus-containing waste aligns with increasingly stringent global regulations on chemical discharge. This proactive compliance reduces the risk of regulatory fines and enhances the corporate sustainability profile, which is becoming a critical factor for multinational customers when selecting a reliable lycopene intermediate supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams establishing specification limits for the new intermediate.
Q: What are the primary technical advantages of this new phosphonate intermediate over conventional lycopene synthesis routes?
A: The novel 1,4,6,10-tetradouble-bond pentadecyl phosphonate intermediate eliminates the need for toxic and expensive triphenylphosphine used in traditional Wittig reactions. Furthermore, it bypasses the technically challenging selective reduction of allenyl phosphonates required in previous methods, resulting in a more robust and higher-yielding process suitable for industrial scale-up.
Q: Are the raw materials for this synthesis readily available for commercial production?
A: Yes, the synthesis relies on pseudoionone and tetraethyl methylene diphosphonate, both of which are established bulk industrial chemicals. This ensures a stable supply chain and significantly lowers the barrier to entry for cost reduction in carotenoid manufacturing compared to routes requiring specialized or hazardous reagents.
Q: What level of stereochemical control can be achieved in the final lycopene product?
A: The process is designed to favor the formation of the all-trans isomer, which is the biologically active and commercially desired form of lycopene. Analytical data from the patent indicates high purity with distinct NMR signals confirming the all-trans structure, minimizing the need for complex downstream isomer separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,6,10-Tetradouble-Bond Pentadecyl Phosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation, including HPLC, GC-MS, and NMR, to guarantee that every batch of 1,4,6,10-tetradouble-bond pentadecyl phosphonate meets the exacting standards required for downstream lycopene synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic impact of switching to this phosphonate-based methodology for your operations. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your production volume requirements, ensuring a seamless transition to a more efficient and sustainable manufacturing model.
