Advanced Synthetic Route for 9-Amino Camptothecin: Enhancing Purity and Commercial Scalability
Advanced Synthetic Route for 9-Amino Camptothecin: Enhancing Purity and Commercial Scalability
The development of efficient synthetic routes for potent anticancer agents remains a cornerstone of modern pharmaceutical manufacturing. Patent CN1222523A introduces a groundbreaking methodology for the preparation of 9-amino-20(S)-camptothecin, a critical intermediate in the production of topoisomerase I inhibitors used for treating leukemia and colorectal cancer. This technology addresses long-standing inefficiencies in the semi-synthetic production of camptothecin derivatives by transforming a historically problematic byproduct into a valuable precursor. By leveraging a unique sequence of reduction, diazotization, and selective nitration, this process offers a robust solution for producing high-purity API intermediates while significantly mitigating waste disposal challenges associated with traditional nitration methods.
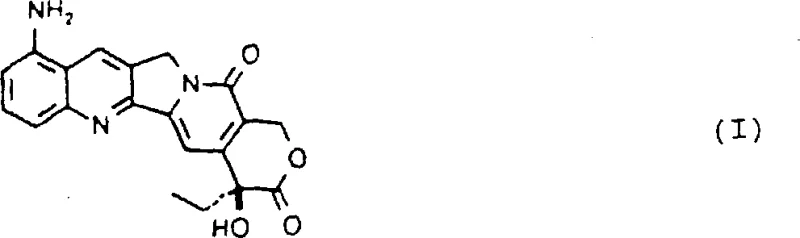
For R&D directors and process chemists, the structural integrity and stereochemical purity of the final product are paramount. The disclosed method ensures the retention of the critical 20(S) configuration throughout the synthetic sequence, utilizing mild reaction conditions that prevent racemization or degradation of the sensitive lactone ring system. This level of control is essential for meeting the stringent regulatory requirements of global health authorities, ensuring that the resulting 9-amino camptothecin is suitable for downstream conversion into clinical-grade therapeutics without compromising biological activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthetic production of 9-nitro and 9-amino camptothecin derivatives has been plagued by poor regioselectivity during the initial nitration of natural camptothecin. Conventional nitration protocols typically yield a mixture comprising approximately 70% of the unwanted 12-nitro isomer and only 30% of the desired 9-nitro isomer. This unfavorable ratio necessitates complex and costly separation processes to isolate the active component, while the bulk 12-nitro byproduct is often discarded as hazardous waste. For procurement managers, this represents a significant inefficiency, as the low overall yield drives up the cost of raw materials and increases the environmental burden of waste disposal, making large-scale operations economically and ecologically unsustainable.
The Novel Approach
The innovative strategy outlined in CN1222523A fundamentally reverses this paradigm by utilizing the 12-nitro isomer—the very waste product of conventional methods—as the primary starting material. This approach involves reducing the 12-nitro group to an amine, converting it into a removable halogen substituent via diazotization, and then selectively nitrating the 9-position. This clever manipulation of functional groups allows for the precise installation of the nitro group at the desired location without generating the troublesome isomeric mixtures seen in direct nitration. Consequently, this method transforms a liability into an asset, offering a streamlined pathway that enhances atom economy and simplifies the purification workflow for commercial scale-up of complex pharmaceutical intermediates.
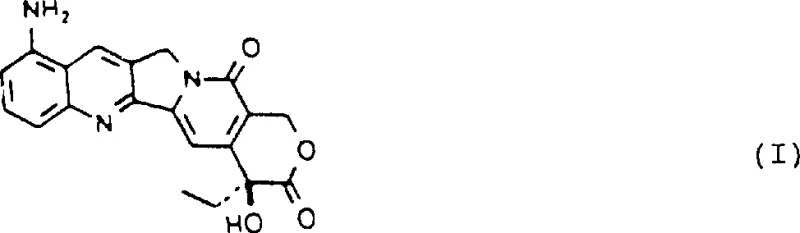
Mechanistic Insights into Selective Nitration and Reductive Dehalogenation
The core of this synthetic breakthrough lies in the strategic use of a temporary blocking group at the 12-position to direct subsequent electrophilic substitution. The process begins with the reduction of 12-nitro-20(S)-camptothecin to the corresponding 12-amino derivative using reagents such as tin(II) chloride in hydrochloric acid or catalytic hydrogenation over palladium on carbon. This amino group is then subjected to diazotization using sodium nitrite under acidic conditions, generating a highly reactive diazonium salt in situ. Immediate treatment with copper(I) halides facilitates a Sandmeyer-type reaction, replacing the diazonium moiety with a chlorine or bromine atom. This 12-halo intermediate serves as a crucial pivot point, as the halogen atom deactivates the 12-position against further electrophilic attack while directing the incoming nitronium ion exclusively to the 9-position during the subsequent nitration step.
Following the selective introduction of the nitro group at the 9-position, the final transformation involves a dual reduction strategy to yield the target 9-amino camptothecin. The patent discloses that the 9-nitro-12-halo intermediate can be reduced in a single step or via a two-step sequence to remove both the nitro group and the temporary halogen substituent. Remarkably, the newly formed 9-amino group acts as a weak non-nucleophilic base that can facilitate the reductive removal of the halogen without decomposing the sensitive camptothecin scaffold. This mechanistic nuance allows for the use of mild reducing agents like molecular hydrogen or formic acid derivatives, preserving the stereochemical integrity of the molecule while ensuring high conversion rates and minimal formation of side products.
How to Synthesize 9-Amino Camptothecin Efficiently
Implementing this synthesis requires careful control of reaction parameters to maximize yield and purity. The process starts with the reduction of the nitro group, followed by the critical diazotization and halogenation steps which must be monitored to prevent over-reaction. The subsequent nitration requires precise temperature control to ensure regioselectivity, and the final reduction must be optimized to cleave the carbon-halogen bond without affecting the lactone ring. Detailed operational procedures, including specific solvent systems, stoichiometric ratios, and workup protocols, are essential for reproducibility. For a comprehensive guide on executing these transformations in a GMP environment, please refer to the standardized synthesis steps provided below.
- Reduce 12-nitro-20(S)-camptothecin to 12-amino-20(S)-camptothecin using SnCl2 or catalytic hydrogenation.
- Convert the 12-amino derivative to a 12-halo compound via diazotization with NaNO2 and reaction with copper halides.
- Nitrate the 12-halo intermediate at the 9-position, followed by a final reduction step to remove both the nitro and halo groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented process offers substantial advantages by decoupling production yields from the limitations of natural product extraction ratios. By enabling the utilization of the 12-nitro isomer, manufacturers can effectively double the theoretical yield of valuable intermediates derived from natural camptothecin sources. This shift not only reduces the dependency on massive quantities of raw plant material but also stabilizes the supply chain against fluctuations in agricultural output. Furthermore, the elimination of complex isomer separation steps simplifies the manufacturing workflow, leading to shorter production cycles and reduced operational overheads for facilities aiming for cost reduction in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The ability to recycle the 12-nitro byproduct eliminates the need for expensive disposal procedures and converts waste into revenue-generating product. By avoiding the loss of 70% of the nitrated material typical in conventional methods, the overall cost of goods sold is drastically lowered. Additionally, the use of common industrial reagents such as copper halides and tin salts ensures that raw material costs remain predictable and manageable, avoiding the volatility associated with specialized catalysts or exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by providing a reliable alternative to direct nitration. Since the process relies on well-established chemical transformations like reduction and diazotization, it is less susceptible to the variability often encountered in biological extractions. This reliability allows procurement teams to secure long-term contracts with greater confidence, knowing that the production capacity is not bottlenecked by the low selectivity of upstream nitration processes.
- Scalability and Environmental Compliance: The reaction conditions described, ranging from ambient to moderately elevated temperatures and pressures, are highly amenable to large-scale reactor operations. The process avoids the use of extremely harsh conditions that would require specialized containment equipment, thereby facilitating easier technology transfer from pilot to commercial scale. Moreover, by minimizing waste generation through the recycling of the 12-nitro isomer, the process aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations regarding hazardous waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding the nuances of reagent selection and reaction monitoring is vital for successful technology adoption. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical stakeholders evaluating this methodology for their own production lines.
Q: How does this process solve the waste issue of traditional camptothecin nitration?
A: Traditional nitration produces a 70/30 mixture of unwanted 12-nitro and desired 9-nitro isomers. This patent utilizes the unwanted 12-nitro isomer as the starting material, effectively recycling waste into the valuable 9-amino product.
Q: What are the critical reaction conditions for the halogenation step?
A: The conversion of the 12-amino intermediate to the 12-halo derivative requires diazotization using sodium nitrite in acidic media (HCl or HBr) at 0-5°C, followed by reaction with copper(I) halides at elevated temperatures up to 80°C.
Q: Is the stereochemistry at the 20-position preserved during synthesis?
A: Yes, the mild reaction conditions employed throughout the sequence, particularly during the reduction steps, ensure that the 20(S) configuration is retained in the final 9-amino camptothecin product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Amino Camptothecin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 9-amino camptothecin meets the exacting standards required for API synthesis. Our commitment to quality assurance means that our clients can rely on consistent material performance, reducing the risk of batch failures in downstream processing.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your production costs and time-to-market for next-generation anticancer drugs.
