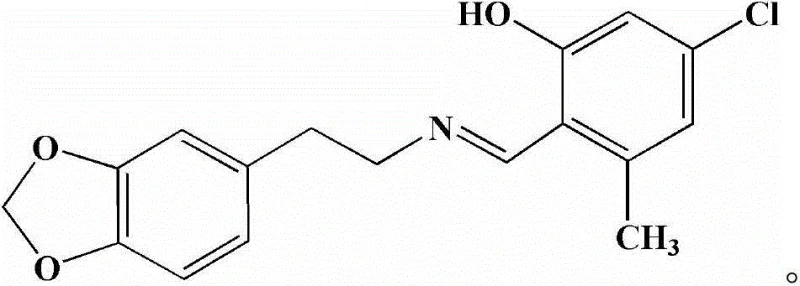
Advanced Synthesis of Homopiperonylamine Derivatives for High-Purity Antitumor Intermediates
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved therapeutic indices, particularly in the challenging field of oncology. Patent CN102924425B introduces a significant advancement in this domain by disclosing a novel Schiff base compound, specifically homopiperonylamine 3-methyl-5-chlorosalicylide, along with a robust and scalable synthesis method. This compound represents a strategic intersection of accessible raw materials and sophisticated molecular design, targeting high-selectivity antitumor activity. The structural integrity of this molecule, characterized by a stable imine linkage between a substituted salicylaldehyde and a homopiperonylamine moiety, provides a unique pharmacophore that has demonstrated remarkable efficacy in preliminary biological screenings. For R&D teams and procurement specialists alike, understanding the synthetic accessibility and biological potential of this intermediate is crucial for future drug development pipelines. The following analysis dissects the technical merits of this patent, highlighting its potential as a reliable pharmaceutical intermediate supplier solution for next-generation anticancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of Schiff bases involving sensitive aldehydes and amines has been plagued by equilibrium limitations and hydrolytic instability. Conventional methods often rely on acid catalysis or high-temperature azeotropic distillation to remove water, which can lead to the degradation of thermally sensitive functional groups or the formation of unwanted polymeric byproducts. Furthermore, many existing protocols utilize stoichiometric amounts of harsh dehydrating agents that complicate downstream purification and increase waste disposal costs. In the context of complex heterocyclic amines like homopiperonylamine, controlling the regioselectivity and preventing side reactions at the amine nitrogen can be exceptionally difficult without precise condition control. These inefficiencies result in lower overall yields, typically ranging significantly below optimal levels, and necessitate extensive chromatographic purification, which is economically unviable for large-scale commercial production. The reliance on such cumbersome processes creates bottlenecks in the supply chain, delaying the availability of high-purity intermediates required for preclinical and clinical studies.
The Novel Approach
The methodology outlined in patent CN102924425B offers a transformative solution by employing a mild, dehydration-driven condensation strategy that circumvents the pitfalls of traditional acid catalysis. By utilizing activated 4A molecular sieves directly within the reaction medium, the process continuously scavenges the water byproduct, effectively driving the equilibrium towards the desired imine product without the need for extreme temperatures or corrosive catalysts. This approach allows the reaction to proceed smoothly within a moderate temperature range of 45°C to the solvent's boiling point, preserving the integrity of the chloro and methyl substituents on the aromatic ring. The simplicity of the workup procedure, which involves merely cooling the reaction mixture to induce crystallization, eliminates the need for complex extraction or column chromatography steps. This streamlined workflow not only enhances the operational safety profile by avoiding hazardous reagents but also drastically reduces the processing time and solvent consumption. Consequently, this novel approach establishes a new benchmark for efficiency in Schiff base manufacturing, aligning perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Molecular Sieve-Assisted Condensation
The core of this synthesis lies in the nucleophilic attack of the primary amine group of homopiperonylamine on the carbonyl carbon of 3-methyl-5-chlorosalicylaldehyde. This initial addition forms a hemiaminal intermediate, which is inherently unstable and prone to reverting to starting materials in the presence of water. The innovation here is the strategic integration of 4A molecular sieves, which possess a pore size specifically tuned to adsorb water molecules while excluding the larger organic reactants and products. By sequestering the water generated during the elimination step of the hemiaminal, the molecular sieves shift the reaction equilibrium irreversibly towards the formation of the C=N double bond. This physical removal of water is far superior to chemical dehydration methods as it avoids the introduction of acidic or basic impurities that could catalyze the hydrolysis of the newly formed Schiff base. The presence of the electron-withdrawing chlorine atom and the electron-donating methyl group on the salicylaldehyde ring further modulates the electrophilicity of the carbonyl carbon, optimizing the reaction kinetics for high conversion rates under mild thermal conditions.
Impurity control in this process is inherently managed through the crystallization mechanism driven by solvent polarity and temperature gradients. As the reaction proceeds to completion, indicated by TLC monitoring, the concentration of the target Schiff base increases until it exceeds its solubility limit upon cooling. The slow cooling process from reflux temperature down to 10-25°C encourages the formation of well-defined yellow needle-like crystals, which naturally exclude soluble impurities and unreacted starting materials from the crystal lattice. This phenomenon, known as fractional crystallization, serves as a powerful purification step that often renders further chromatographic purification unnecessary. The patent data indicates that recrystallization from the same reaction solvent can further enhance purity to greater than 95%, ensuring that the final material meets the stringent specifications required for biological testing. This robust control over the solid-state properties of the product ensures batch-to-batch consistency, a critical factor for regulatory compliance and supply chain reliability in the pharmaceutical sector.
How to Synthesize Homopiperonylamine 3-methyl-5-chlorosalicylide Efficiently
The synthesis protocol described in the patent is designed for operational simplicity and high reproducibility, making it ideal for both laboratory-scale optimization and industrial scale-up. The process begins with the dissolution of equimolar quantities of the aldehyde and amine precursors in a suitable organic solvent, with ethanol being a preferred choice due to its low cost and environmental profile. The addition of pre-activated molecular sieves is the critical step that dictates the success of the reaction, requiring careful attention to the dryness of the reagents and the activation state of the sieves. Once the mixture is heated to reflux, the reaction progress is monitored via thin-layer chromatography to ensure complete consumption of the starting materials before initiating the crystallization phase. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and precise temperature profiles for crystallization, are provided in the technical guide below to ensure successful replication of the high yields reported in the patent literature.
- Dissolve equimolar amounts of 3-methyl-5-chlorosalicylaldehyde and homopiperonylamine in an organic solvent such as ethanol or dichloromethane.
- Add activated 4A molecular sieves to the reaction mixture to remove water and drive the condensation equilibrium forward.
- Heat the mixture to reflux (45°C to boiling point) for 1-12 hours, then cool and crystallize the product for isolation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive transition metal catalysts and harsh dehydrating reagents translates directly into a simplified bill of materials, reducing the dependency on volatile commodity markets for specialized chemicals. Furthermore, the use of common solvents like ethanol and methanol, which are readily available in bulk quantities globally, mitigates the risk of supply disruptions that often plague projects relying on exotic or regulated solvents. The ability to isolate the product through simple cooling and filtration, rather than energy-intensive distillation or chromatography, significantly lowers the utility costs associated with production. These factors combine to create a manufacturing process that is not only cost-effective but also resilient to market fluctuations, ensuring a stable supply of this critical intermediate for downstream drug development programs.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive catalysts and complex purification infrastructure. By relying on physical dehydration via molecular sieves, the method avoids the procurement and disposal costs associated with chemical dehydrating agents. Additionally, the high crude purity achieved through crystallization minimizes the loss of material during purification, maximizing the effective yield per batch. This efficiency allows for a more competitive pricing structure for the final intermediate, providing a clear economic advantage in cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 3-methyl-5-chlorosalicylaldehyde and homopiperonylamine ensures that raw material sourcing is straightforward and secure. These precursors are established industrial chemicals with multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a range of solvents and moderate temperatures, further enhances process reliability, minimizing the likelihood of batch failures due to minor parameter deviations. This stability is essential for reducing lead time for high-purity pharmaceutical intermediates and maintaining consistent production schedules.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as the use of molecular sieves and simple crystallization scales linearly without the engineering challenges associated with azeotropic distillation on a large scale. The reduced solvent usage and the ability to recycle mother liquors contribute to a lower environmental footprint, aligning with modern green chemistry principles. This facilitates easier regulatory approval for manufacturing sites and supports the commercial scale-up of complex pharmaceutical intermediates without significant capital investment in waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel Schiff base derivative. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for integrating this intermediate into your specific research or production workflows. The responses cover aspects ranging from reaction optimization to biological activity profiles, providing a comprehensive overview of the technology's capabilities.
Q: What is the primary advantage of using molecular sieves in this Schiff base synthesis?
A: The use of 4A molecular sieves effectively removes water generated during the condensation reaction. This shifts the chemical equilibrium towards product formation according to Le Chatelier's principle, significantly improving yields without requiring harsh dehydrating agents or azeotropic distillation.
Q: Does this compound show activity against drug-resistant cancer cell lines?
A: Yes, experimental data indicates that this homopiperonylamine derivative exhibits significant inhibitory activity against the cisplatin-resistant ovarian cancer cell line SK-OV-3/DDP. Its mechanism appears distinct from cisplatin, suggesting potential for overcoming multidrug resistance in chemotherapy applications.
Q: What solvents are compatible with this synthesis protocol?
A: The patent specifies flexibility in solvent choice, including ethanol, methanol, dichloromethane, and chloroform. This versatility allows manufacturers to select solvents based on cost, availability, and downstream processing requirements, facilitating easier scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homopiperonylamine 3-methyl-5-chlorosalicylide Supplier
The technical potential of homopiperonylamine 3-methyl-5-chlorosalicylide as a selective antitumor agent underscores the importance of securing a manufacturing partner with deep expertise in complex organic synthesis. NINGBO INNO PHARMCHEM stands ready to support your development needs, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, guaranteeing the consistency required for preclinical and clinical trials. We understand the critical nature of timeline and quality in drug development, and our team is dedicated to delivering high-quality intermediates that accelerate your path to market.
We invite you to engage with our technical procurement team to discuss your specific requirements for this compound or related derivatives. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized manufacturing processes can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to evaluate the suitability of our materials for your unique applications with confidence. Let us collaborate to bring this promising antitumor candidate from the laboratory to the clinic.
