Revolutionizing Anthraquinone Dye Intermediates: A Clean Catalytic Hydrogenation Route for Commercial Scale-Up
Introduction to Advanced Anthraquinone Intermediate Manufacturing
The global demand for high-performance anthraquinone dyes continues to surge, placing immense pressure on manufacturers to secure reliable sources of key intermediates like 1-aminoanthraquinone. Traditional production methods have long been plagued by severe environmental liabilities and inconsistent quality, creating a bottleneck for downstream dye synthesis. However, a groundbreaking technological shift is emerging, anchored by the innovations disclosed in patent CN102531925A. This patent details a sophisticated method for preparing high-purity 1-aminoanthraquinone through catalytic hydrogenation, representing a paradigm shift from polluting stoichiometric reductions to clean, catalytic processes. By leveraging specific crystallization mechanics and optimized solvent ratios, this technology achieves a total yield exceeding 98% and purity levels greater than 99%, effectively addressing the critical pain points of purity and environmental compliance that have historically constrained the industry.
For R&D directors and technical leaders, the significance of this patent lies not just in the final product specifications, but in the elegant simplicity of the reaction engineering. The method circumvents the formation of pulpous liquids—a common failure mode in low-solvent hydrogenation—by ingeniously utilizing the oxidation of trace over-hydrogenated byproducts to induce homogeneous crystallization. This self-purifying mechanism ensures that the resulting 1-aminoanthraquinone meets the stringent quality standards required for high-end textile and specialty dye applications. Furthermore, the process operates under relatively mild conditions, ranging from room temperature to 120°C, utilizing robust catalysts such as Pd/C or Raney Ni, which simplifies reactor design and enhances operational safety compared to high-pressure ammonolysis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1-aminoanthraquinone has relied heavily on two primary pathways: the sulfonation ammonolysis process and the sulfuration alkaline process. While these methods established the foundation of the anthraquinone dye industry, they are increasingly becoming untenable in the modern regulatory landscape. The sulfonation ammonolysis process, for instance, is notorious for generating mercury-containing wastewater, posing severe risks to operator health and requiring exorbitant costs for hazardous waste treatment and disposal. Similarly, the sulfuration alkaline process, though widely adopted domestically in certain regions, produces massive quantities of alkaline sulfur-bearing waste solutions that are notoriously difficult to treat and neutralize. These environmental burdens not only inflate the operational expenditure (OPEX) but also expose manufacturers to significant regulatory risks and potential production shutdowns due to non-compliance.
Beyond the environmental toll, these conventional methods suffer from inherent technical inefficiencies that compromise product quality and supply stability. The ammonolysis process often requires extremely high pressures, introducing substantial security risks and necessitating expensive, specialized high-pressure equipment that limits scalability. Moreover, the selectivity and yield of these older technologies are frequently suboptimal, leading to complex purification trains that erode profit margins. As global markets, particularly in North America and Europe, increasingly transfer their purchasing power to developing nations while simultaneously demanding higher quality and greener credentials, manufacturers relying on these legacy technologies find themselves unable to compete. The inability to consistently produce high-quality 1-aminoanthraquinone without generating toxic byproducts has created a supply gap that innovative catalytic technologies are now poised to fill.
The Novel Approach
In stark contrast to the dirty and hazardous legacy methods, the catalytic hydrogenation route described in patent CN102531925A offers a streamlined, environmentally benign alternative that redefines efficiency in fine chemical manufacturing. This novel approach replaces toxic reagents with molecular hydrogen and reusable heterogeneous catalysts, fundamentally altering the waste profile of the synthesis. The core innovation lies in the precise control of the reaction medium; by maintaining a solvent-to-1-nitroanthraquinone weight ratio between 1.0 and 10.0, the process avoids the formation of unmanageable pulpous states that typically hinder hydrogenation efficiency. Instead of viewing low solvent volumes as a limitation, the inventors have turned it into an advantage, coupling it with a unique post-reaction treatment where the reducing solution is exposed to an oxidant like air.
This exposure triggers the oxidation of a minute quantity of over-hydrogenated products, which acts as a nucleation driver to promote solution supersaturation and facilitate homogeneous crystallization. The result is a direct pathway to high-purity crystals without the need for energy-intensive recrystallization steps or complex chromatographic separations. The simplicity of the equipment requirements—standard atmospheric or low-pressure hydrogenation reactors—combined with the ability to recycle mother liquor and catalyst mechanically for multiple batches, drastically reduces both capital expenditure (CAPEX) and raw material consumption. For procurement and supply chain leaders, this translates to a more resilient supply chain with lower vulnerability to raw material price volatility and regulatory crackdowns on pollution.
Mechanistic Insights into Catalytic Hydrogenation and Oxidative Crystallization
To fully appreciate the technical sophistication of this process, one must delve into the mechanistic interplay between the hydrogenation step and the subsequent oxidative crystallization. The reaction begins with the reduction of the nitro group in 1-nitroanthraquinone (Formula II) to an amino group, forming 1-aminoanthraquinone (Formula I). This transformation is catalyzed by transition metals such as Palladium, Nickel, or Platinum, supported on carbon or in skeletal forms like Raney Nickel. The choice of solvent, typically N,N-Dimethylformamide (DMF) or a mixture of DMF and xylene, plays a critical role in solubilizing the anthraquinone substrate while maintaining a viscosity that allows for efficient mass transfer of hydrogen gas. The patent highlights that maintaining the solvent ratio within the specific 1.0 to 10.0 range is crucial; too much solvent dilutes the reaction and increases downstream energy costs for recovery, while too little traditionally leads to poor mixing and incomplete conversion.
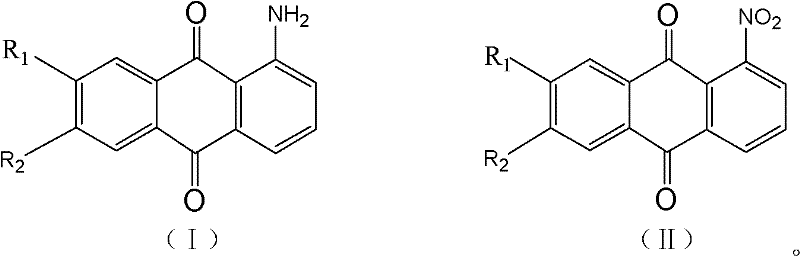
The true brilliance of the mechanism, however, is revealed in the workup phase. In standard hydrogenations, over-reduction can lead to impurities that are difficult to separate. Here, the process intentionally leverages the presence of these trace over-hydrogenated species. Upon filtration of the catalyst, the reducing solution is stirred or allowed to stand in the presence of an oxidant, preferably airborne oxygen. This controlled oxidation converts the unstable over-hydrogenated intermediates back into 1-aminoanthraquinone or related quinoid structures directly within the solution. This in-situ generation of product increases the local concentration beyond the saturation point, driving homogeneous crystallization. Because the crystals form uniformly throughout the solution rather than on foreign surfaces, they tend to exclude impurities more effectively, yielding a product with purity consistently above 99%. This mechanism effectively turns a potential impurity problem into a purification tool, showcasing a deep understanding of physical organic chemistry applied to process engineering.
How to Synthesize 1-Aminoanthraquinone Efficiently
Implementing this synthesis route requires careful attention to the balance between hydrogenation kinetics and crystallization thermodynamics. The process is designed to be robust enough for industrial scale-up while maintaining the precision needed for high-purity outputs. Operators must ensure that the catalyst loading is optimized—typically around 3-5% for Pd/C or slightly higher for Raney Ni—to achieve complete conversion within 3 to 4 hours. Following the reaction, the mechanical separation of the catalyst is vital, as the recovered catalyst can be directly reused in subsequent batches, further enhancing the economic viability of the process. The crystallization step, driven by oxidative supersaturation, eliminates the need for adding large volumes of anti-solvents initially, although water may be added to the final mother liquor to recover residual product. For a detailed, step-by-step breakdown of the standardized operating procedures and safety protocols required to execute this synthesis, please refer to the technical guide below.
- Conduct catalytic hydrogenation of 1-nitroanthraquinone in a solvent (DMF or DMF/Xylene mixture) using Pd/C or Raney Ni catalyst at temperatures ranging from room temperature to 120°C.
- Filter off the catalyst and stir or stand the reducing solution in an oxidant (air/oxygen) to oxidize trace over-hydrogenated products, inducing supersaturation.
- Allow homogeneous crystallization to occur, filter the crystals, and recycle the mother liquor mechanically for subsequent batches to maximize yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology represents a strategic opportunity to de-risk the supply of critical dye intermediates while simultaneously driving down the total cost of ownership. The elimination of mercury and sulfur-based reagents removes the need for expensive hazardous waste disposal contracts and mitigates the risk of production halts due to environmental inspections. Furthermore, the ability to operate with significantly lower solvent loads reduces the volume of volatile organic compounds (VOCs) that must be managed, leading to substantial savings in solvent recovery and ventilation systems. The process's inherent safety profile, operating at near-atmospheric pressures compared to high-pressure ammonolysis, lowers insurance premiums and reduces the engineering controls required for facility maintenance.
- Cost Reduction in Manufacturing: The economic advantages of this method are derived from multiple vectors of efficiency. By eliminating the need for stoichiometric amounts of toxic reducing agents like sodium sulfide or mercury salts, the raw material bill of materials is significantly streamlined. Additionally, the catalyst recycling capability means that the expensive precious metals or nickel are not consumed in a single pass but are retained within the production loop, amortizing their cost over tons of product. The high yield (>98%) ensures that very little starting material is lost to side reactions or waste streams, maximizing the return on every kilogram of 1-nitroanthraquinone purchased. Consequently, the overall production cost is drastically simplified and reduced compared to legacy sulfonation routes.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory volatility surrounding dirty chemical processes. By switching to this clean hydrogenation method, manufacturers insulate their supply chains from sudden bans on mercury or sulfur discharge. The simplicity of the equipment—standard hydrogenation reactors and filtration units—means that maintenance downtime is minimized and spare parts are readily available globally. Moreover, the use of common solvents like DMF and xylene ensures that raw material sourcing remains stable and competitive, avoiding the bottlenecks associated with specialty reagents required for older technologies. This reliability makes the supplier a more dependable partner for long-term contracts.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen thermal and mixing challenges, but this hydrogenation route is inherently scalable due to its reliance on well-understood unit operations. The absence of highly exothermic sulfonation steps reduces the risk of thermal runaway during scale-up. From an environmental perspective, the process is nearly zero-discharge regarding heavy metals and sulfur, aligning perfectly with the increasingly stringent ESG (Environmental, Social, and Governance) criteria demanded by multinational corporations. This compliance facilitates smoother customs clearance and market access in regulated jurisdictions like the EU and North America, ensuring that the supply chain remains open and uninterrupted.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is essential for making informed sourcing decisions. The following questions address common concerns regarding the technical feasibility, quality assurance, and environmental impact of this catalytic method. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. We encourage technical teams to review these details to assess the fit for their specific application requirements.
Q: How does this catalytic hydrogenation method improve upon traditional sulfonation processes?
A: Unlike traditional sulfonation ammonolysis which generates hazardous mercury-containing wastewater, or sulfuration alkaline processes producing difficult-to-treat sulfur-bearing waste, this catalytic hydrogenation method is a clean process with virtually no three-waste emission, significantly lowering environmental compliance costs.
Q: What is the key mechanism ensuring high purity (>99%) in this synthesis?
A: The process utilizes a unique crystallization mechanism where a small amount of over-hydrogenated product is oxidized within the reducing solution. This oxidation promotes solution supersaturation, triggering homogeneous crystallization that inherently excludes impurities, resulting in product purity exceeding 99%.
Q: Can the solvent consumption be optimized in this industrial process?
A: Yes, the patent specifies a critical solvent-to-substrate weight ratio of 1.0 to 10.0:1. By operating at these lower ratios compared to conventional methods, and utilizing mother liquor recycling techniques, the process drastically reduces solvent consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aminoanthraquinone Supplier
As the global chemical landscape shifts towards sustainability and high efficiency, partnering with a manufacturer who masters these advanced catalytic technologies is crucial. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep expertise in catalytic hydrogenation and crystallization engineering to deliver superior intermediates. Our facilities are equipped to handle complex synthetic pathways, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key; therefore, our rigorous QC labs enforce stringent purity specifications to ensure that every batch of 1-aminoanthraquinone meets the exacting standards required for high-performance dye synthesis. Our commitment to green chemistry aligns with the patent's principles, ensuring that your supply chain is not only cost-effective but also environmentally responsible.
We invite you to explore how our optimized manufacturing capabilities can enhance your product portfolio and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of catalytic hydrogenation can become a competitive advantage for your business, securing your supply of high-purity 1-aminoanthraquinone for the future.
