Advanced Synthesis of Diphenolic Acid Derivatives for Commercial Epoxy and Polycarbonate Applications
Advanced Synthesis of Diphenolic Acid Derivatives for Commercial Epoxy and Polycarbonate Applications
The chemical industry is witnessing a significant paradigm shift towards utilizing biomass-derived platform molecules for the synthesis of high-value fine chemicals, a trend exemplified by the technological breakthroughs detailed in patent CN103058831A. This pivotal intellectual property discloses a versatile and robust methodology for preparing a diverse array of diphenolic acid derivatives, ranging from bisphenol alcohols to complex epoxy resin precursors. By leveraging levulinic acid, a renewable resource whose cost has decreased due to production technology updates, manufacturers can now access a sustainable pathway to produce critical intermediates that were previously dependent on petrochemical feedstocks. The patent outlines a comprehensive suite of reactions that transform diphenolic acid esters into functional materials suitable for high-performance applications in the polymer and coating sectors. This development is particularly strategic for R&D teams seeking to diversify their supply chain with bio-based alternatives that do not compromise on thermal stability or mechanical performance. Furthermore, the simplicity of the reaction conditions described suggests a high degree of scalability, making these derivatives attractive candidates for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of specialized phenolic derivatives and epoxy resin precursors has been plagued by complex multi-step sequences that often require harsh reaction conditions and expensive catalysts. Conventional routes frequently suffer from low atom economy and generate substantial amounts of hazardous waste, posing significant challenges for environmental compliance and cost management in modern manufacturing facilities. Many existing processes rely on non-renewable feedstocks that are subject to volatile market pricing, creating supply chain instability for procurement managers who need predictable budgeting. Additionally, the purification of intermediates in older methodologies often involves tedious chromatographic separations or extensive recrystallization steps, which drastically reduce overall throughput and increase energy consumption. The lack of structural diversity in commercially available phenolic building blocks has also limited the ability of formulators to tune the properties of final polymer products, such as glass transition temperature and flexibility. These cumulative inefficiencies create a bottleneck for companies aiming to innovate in the fields of advanced coatings and engineering plastics.
The Novel Approach
In stark contrast, the methodology presented in patent CN103058831A introduces a streamlined and highly efficient synthetic strategy that utilizes diphenolic acid esters as a universal starting material for multiple derivative classes. This novel approach capitalizes on the inherent reactivity of the ester functionality to facilitate direct transformation into alcohols, amides, or epoxy networks through well-controlled reaction parameters. The process eliminates the need for exotic reagents, relying instead on standard reducing agents like lithium aluminum hydride or common amines, which are readily available in bulk quantities. By operating under relatively mild thermal conditions, typically between 100°C and 200°C depending on the specific transformation, the method minimizes thermal degradation of sensitive functional groups and ensures high product integrity. The versatility of this platform allows for the rapid generation of a library of compounds, enabling researchers to quickly screen for optimal performance characteristics in final applications. This flexibility represents a significant leap forward in process chemistry, offering a reliable diphenolic acid derivative supplier pathway that aligns with green chemistry principles.
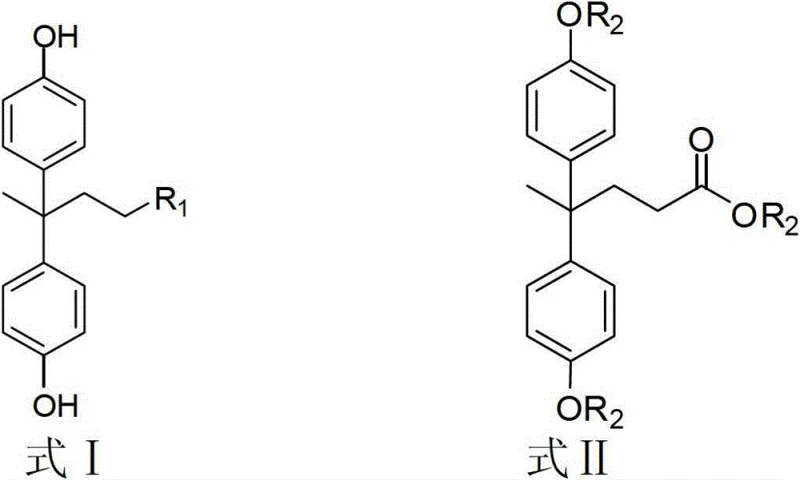
Mechanistic Insights into Diphenolic Acid Derivative Transformations
The core of this technological advancement lies in the precise control of nucleophilic substitution and reduction mechanisms acting upon the diphenolic acid ester backbone. For the synthesis of bisphenol alcohols, the mechanism involves the nucleophilic attack of hydride ions from reducing agents such as sodium borohydride or lithium aluminum hydride on the carbonyl carbon of the ester group. This reduction proceeds through a tetrahedral intermediate which subsequently collapses to release the alkoxide, eventually yielding the primary alcohol functionality after aqueous workup. The patent specifies reaction temperatures ranging from 0°C to 100°C, allowing for fine-tuning of the reaction kinetics to prevent over-reduction or side reactions on the aromatic rings. In the case of amidation, the mechanism shifts to a thermal nucleophilic acyl substitution where the amine nitrogen attacks the ester carbonyl, displacing the alkoxy leaving group to form the stable amide bond. This thermal process is particularly effective for long-chain aliphatic amines, where the hydrophobic interactions can be leveraged to drive the equilibrium towards product formation.
Regarding the formation of epoxy resins, the mechanism involves the ring-opening of epichlorohydrin or diglycidyl ethers by the phenolic hydroxyl groups of the diphenolic acid core. This etherification reaction is catalyzed by phase transfer catalysts like tetrabutyl ammonium bromide, which facilitate the transport of reactive anionic species into the organic phase. The resulting glycidyl ether structures possess high reactivity due to the strained three-membered oxirane ring, making them ideal for cross-linking applications. The patent highlights the importance of stoichiometric control, recommending a molar ratio of epoxy functional groups to diphenolic acid of approximately 6:1 to ensure complete conversion and optimal molecular weight distribution. This precise control over the polymerization precursor structure is critical for achieving consistent curing behavior in the final resin formulation. The ability to introduce various spacer lengths via different diglycidyl ethers allows for the customization of the resin's flexibility and toughness.
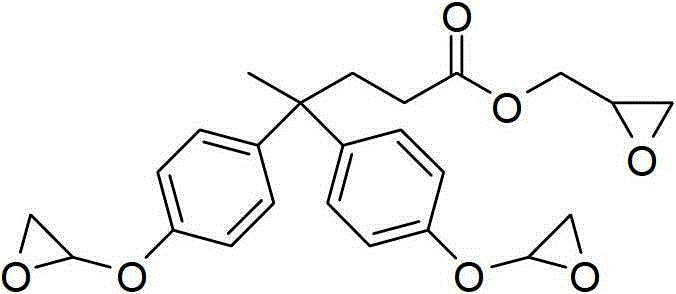
How to Synthesize Diphenolic Acid Derivatives Efficiently
The synthesis protocols outlined in the patent provide a clear roadmap for producing these high-value intermediates with minimal operational complexity. The procedures are designed to be robust, tolerating slight variations in reagent quality while maintaining high yields and purity profiles. For R&D teams looking to replicate these results, the key lies in strict adherence to the specified molar ratios and temperature profiles, particularly during the exothermic reduction steps. The workup procedures are notably straightforward, often requiring only filtration to remove inorganic salts followed by rotary evaporation to isolate the crude product, which can often be used directly in subsequent polymerization steps. This operational simplicity reduces the barrier to entry for pilot-scale production and accelerates the timeline from laboratory discovery to commercial validation. Detailed standardized synthesis steps see the guide below.
- For bisphenol alcohol synthesis, dissolve a reducing agent such as lithium aluminum hydride in an organic solvent like tetrahydrofuran, then add diphenolic acid esters and react at 0-100°C for 5-10 hours followed by filtration and solvent removal.
- To prepare aliphatic or aromatic amides, mix diphenolic acid esters with the corresponding amine (aliphatic or aniline) and heat the mixture to 150-200°C under nitrogen protection for 5-20 hours to achieve amidation.
- For epoxy resin production, react diphenolic acid with epichlorohydrin or a bis-epoxy functional compound in the presence of a phase transfer catalyst at 90-120°C, followed by neutralization and distillation to isolate the final resin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors focused on cost optimization and risk mitigation. The reliance on levulinic acid, a biomass-derived platform molecule, insulates the supply chain from the volatility associated with fossil-fuel-based feedstocks, ensuring greater long-term price stability. Furthermore, the high productivity and ease of separation reported in the patent translate directly into reduced manufacturing overheads, as less time and energy are spent on purification and waste treatment. The elimination of complex catalytic systems and the use of common solvents like tetrahydrofuran or ether simplify the logistics of raw material sourcing, allowing for consolidation of suppliers. This streamlining of the production process enhances the overall agility of the supply chain, enabling faster response times to market demands for specialized epoxy and polycarbonate materials.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by utilizing inexpensive and readily available starting materials such as diphenolic acid esters and common amines, avoiding the need for precious metal catalysts. The high reaction yields and simple isolation methods, often involving just filtration and distillation, drastically reduce the consumption of solvents and energy required for downstream processing. By minimizing the number of unit operations and avoiding complex purification techniques like column chromatography, the overall cost of goods sold is substantially lowered. This economic efficiency makes the derivatives highly competitive against traditional petrochemical alternatives in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing raw materials from a biomass-based value chain diversifies the supply base and reduces dependency on single-source petrochemical providers. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by minor fluctuations in utility supplies or reagent quality. Additionally, the versatility of the diphenolic acid ester intermediate allows manufacturers to switch between producing alcohols, amides, or resins based on real-time market demand without retooling entire production lines. This flexibility ensures a continuous supply of critical intermediates even during periods of market disruption.
- Scalability and Environmental Compliance: The synthesis methods are inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant changes in reaction dynamics. The use of nitrogen or argon atmospheres for amidation reactions prevents oxidation and ensures product consistency, while the absence of heavy metal catalysts simplifies wastewater treatment and regulatory compliance. The resulting low-toxicity epoxy resins and polycarbonates align with increasingly stringent global regulations regarding consumer safety and environmental impact, particularly for applications in food contact materials like infant feeding bottles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of these diphenolic acid derivatives in industrial settings. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating these materials into existing formulations.
Q: What are the primary applications of the diphenolic acid derivatives described in this patent?
A: The derivatives, including bisphenol alcohols and various amides, are primarily utilized as intermediates for synthesizing low-toxicity epoxy resins and polycarbonates, serving as safer alternatives to traditional bisphenol A-based materials in coatings and plastics.
Q: How does the amidation process for aromatic amines differ from aliphatic amines in this synthesis?
A: The amidation with aromatic amines like aniline typically requires higher reaction temperatures ranging from 150°C to 250°C and longer reaction times of 10 to 20 hours compared to aliphatic amines, which react efficiently at 160°C to 200°C within 5 to 10 hours.
Q: What advantages does this preparation method offer regarding product isolation?
A: The method is characterized by high productivity and ease of separation, often involving simple filtration and rotary evaporation steps to remove solvents and unreacted reagents, which significantly streamlines the downstream purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenolic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic pathways described in patent CN103058831A for the next generation of sustainable polymers. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diphenolic acid derivatives or epoxy resin precursors meets the highest industry standards. We are committed to supporting your R&D efforts with custom synthesis services that leverage these advanced chemistries to deliver superior performance materials.
We invite you to collaborate with our technical procurement team to explore how these innovative derivatives can enhance your product portfolio and reduce your overall manufacturing costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about integrating these bio-based intermediates into your supply chain. Let us be your partner in driving innovation and sustainability in the fine chemical and polymer industries.
