Advanced Synthesis of High-Purity Tetraphenol Ethane Epoxy Resin for Electronic Packaging
The rapid evolution of the microelectronics industry demands materials that can withstand increasingly rigorous performance standards, particularly in the realms of high integration, thermal management, and mechanical stability. As devices become thinner and more powerful, the encapsulation materials protecting them must offer exceptional electrical insulation, heat dissipation, and resistance to environmental stressors without compromising structural integrity. Traditional epoxy resins, such as bisphenol-A types, often suffer from brittleness and insufficient thermal properties, necessitating complex modifications that can introduce new weaknesses. Addressing these critical challenges, patent CN101045781A introduces a sophisticated preparation method for high-purity electronic grade 1,1,2,2-tetrakis(p-hydroxyphenyl)ethane tetraglycidyl ether epoxy resin. This innovation represents a significant leap forward in electronic chemical manufacturing, utilizing a specialized phase transfer catalysis system to achieve unprecedented purity levels. By decoupling the etherification and epoxidation stages and employing precise temperature controls, this technology delivers a resin skeleton inherently possessing high toughness and thermal stability, eliminating the need for rubber modifiers that typically degrade mechanical strength.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing multifunctional epoxy resins, such as those documented in patents JP2005220302 and US5098964, relied heavily on a one-pot strategy where ring-opening addition etherification and ring-closing epoxidation dechlorination occurred simultaneously under strong alkaline conditions. While operationally simple, this conventional methodology suffers from severe chemical inefficiencies that compromise the quality of the final product. Under such aggressive basic environments, the raw material epichlorohydrin is highly susceptible to hydrolysis side reactions, leading to the formation of undesirable by-products and a significant reduction in the overall epoxy value of the resin. Furthermore, the intermediate products are prone to uncontrolled polymerization with the starting tetraphenol ethane, resulting in a broad molecular weight distribution and elevated levels of hydrolyzable chlorine. These impurities are detrimental in semiconductor packaging applications, where even trace amounts of ionic contaminants can cause catastrophic failure of integrated circuits through electrochemical migration or corrosion of fine metal lines.
The Novel Approach
The methodology outlined in CN101045781A fundamentally restructures the synthesis pathway to overcome these inherent defects by introducing a distinct separation of reaction phases and utilizing phase transfer catalysts (PTC). Instead of a single harsh alkaline environment, the process employs quaternary ammonium or phosphonium salts to facilitate a gentle yet efficient ring-opening addition etherification at controlled temperatures between 60°C and 110°C. This selectivity ensures that the primary product is the desired chlorohydroxyl ether intermediate small molecule, effectively suppressing premature polymerization. Subsequently, the process implements a two-stage ring-closing epoxidation strategy: an initial mild dechlorination at 60-80°C followed by a secondary, more vigorous reaction at 70-110°C using methyl isobutyl ketone as a co-solvent. This staged approach guarantees the thorough conversion of intermediates while minimizing the hydrolysis of epichlorohydrin, thereby drastically lowering the hydrolyzable chlorine content to ≤200ppm and maximizing the epoxy value to a range of 0.502 to 0.513.
Mechanistic Insights into Phase Transfer Catalyzed Epoxidation
The core chemical innovation driving this process lies in the mechanistic role of the phase transfer catalyst, which acts as a molecular shuttle to transport hydroxide ions from the aqueous phase into the organic phase where the reaction occurs. In traditional systems, the reliance on direct contact between strong aqueous alkali and organic reactants creates a heterogeneous environment prone to interfacial side reactions. However, the quaternary ammonium or phosphonium cations in this novel method form lipophilic ion pairs with hydroxide anions, allowing them to dissolve readily in the organic solvent containing the chlorohydroxyl ether intermediates. This mechanism facilitates a nucleophilic attack on the carbon-chlorine bond under much milder conditions than previously possible, promoting the intramolecular ring closure to form the glycidyl ether groups without degrading the sensitive epoxy rings. The result is a highly controlled catalytic cycle that preserves the integrity of the tetraphenol ethane backbone, ensuring a narrow molecular weight distribution between 528 and 591 Daltons.
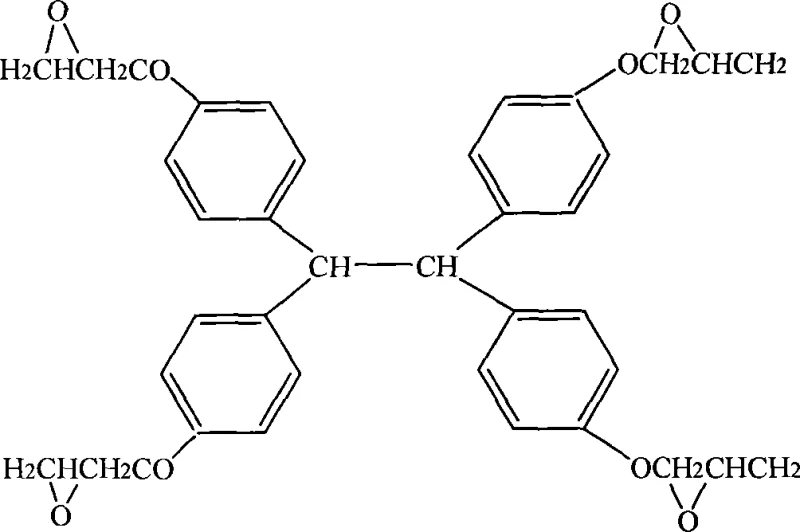
Beyond the primary catalytic cycle, the impurity control mechanism is equally critical for achieving high-purity electronic chemical standards. The specific selection of solvents, such as isopropanol for the initial etherification and methyl isobutyl ketone for the secondary epoxidation, plays a pivotal role in solubilizing the intermediates while keeping inorganic salts precipitated or easily separable. The two-stage addition of alkali solution, with specific mass ratios and concentrations (30-50% for the first stage, 5-20% for the second), prevents local overheating and concentration spikes that could trigger exothermic runaway reactions or oligomerization. By separating the removal of excess epichlorohydrin and solvent between the two epoxidation stages, the process ensures that the second reaction proceeds in a cleaner environment, further driving the equilibrium towards the fully epoxidized product. This meticulous control over reaction kinetics and thermodynamics is what allows the final resin to exhibit a softening point of 80-85°C and a white crystalline appearance, indicative of its high structural regularity and lack of colored degradation products.
How to Synthesize Tetraphenol Ethane Tetraglycidyl Ether Efficiently
The synthesis of this advanced epoxy resin requires precise adherence to the three-stage protocol defined in the patent to ensure reproducibility and electronic-grade quality. The process begins with the preparation of the chlorohydroxyl ether intermediate under inert gas protection, followed by the critical dual-temperature epoxidation sequence, and concludes with a rigorous purification regimen involving crystallization and washing. Each step is optimized to maximize yield while minimizing the formation of hydrolyzable chlorine, which is the primary metric for electronic grade suitability. For detailed operational parameters, including specific molar ratios of epichlorohydrin to tetraphenol ethane (8-30:1) and catalyst loading rates, operators must refer to the standardized technical documentation provided below.
- Conduct ring-opening addition etherification of tetraphenol ethane and epichlorohydrin using a quaternary ammonium or phosphonium phase transfer catalyst at 60-110°C.
- Perform a two-stage ring-closing epoxidation dechlorination: first at 60-80°C with concentrated alkali, then at 70-110°C with methyl isobutyl ketone and dilute alkali.
- Refine the crude resin by dissolving in methyl isobutyl ketone, cooling crystallization, washing with n-hexane, and vacuum drying to obtain the final white crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase transfer catalyzed synthesis route offers substantial strategic advantages over legacy manufacturing processes. The primary value driver is the significant enhancement in product reliability, which directly translates to reduced failure rates in downstream semiconductor assembly and lower warranty costs for end-users. By eliminating the need for post-synthesis rubber modification to improve toughness, the supply chain is simplified, reducing the number of raw materials that need to be sourced, qualified, and inventory-managed. Furthermore, the ability to produce a resin with inherently low chlorine content removes the necessity for expensive and time-consuming ion-exchange purification steps that are often required in conventional epoxy production. This streamlining of the purification process not only accelerates the overall production cycle but also reduces the consumption of auxiliary chemicals and water, aligning with modern sustainability goals and regulatory compliance standards.
- Cost Reduction in Manufacturing: The implementation of phase transfer catalysts allows for milder reaction conditions and higher selectivity, which significantly reduces the formation of by-products that would otherwise require costly disposal or reprocessing. By avoiding the hydrolysis of expensive epichlorohydrin raw materials, the atom economy of the process is improved, leading to a more efficient utilization of inputs. Additionally, the simplified purification workflow, which relies on crystallization rather than complex chromatographic or ion-exchange techniques, lowers the capital expenditure required for processing equipment and reduces the operational costs associated with energy and solvent recovery. These efficiencies collectively contribute to a more competitive cost structure for electronic chemical manufacturing without compromising on the stringent quality specifications required by the semiconductor industry.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process, including tetraphenol ethane, epichlorohydrin, and common phase transfer catalysts like tetraethylammonium bromide, are widely available commodities with stable global supply chains. Unlike specialized modifiers or rare metal catalysts that might be subject to geopolitical supply risks or price volatility, these inputs ensure a consistent and predictable production schedule. The robustness of the synthesis method, characterized by its tolerance to slight variations in reaction conditions due to the buffering effect of the phase transfer system, further enhances manufacturing reliability. This stability allows suppliers to maintain high service levels and reducing lead time for high-purity epoxy resins, ensuring that downstream electronics manufacturers can meet their own aggressive production targets without interruption.
- Scalability and Environmental Compliance: The process design is inherently scalable, moving seamlessly from laboratory bench scales to multi-ton commercial production facilities without the need for fundamental process re-engineering. The use of recoverable solvents like isopropanol and methyl isobutyl ketone, combined with the efficient separation of salt by-products, minimizes the generation of hazardous liquid waste. The low chlorine content of the final product also reduces the environmental burden associated with the disposal of corrosive waste streams. This alignment with green chemistry principles facilitates easier permitting and regulatory approval in jurisdictions with strict environmental controls, securing the long-term viability of the manufacturing site and ensuring uninterrupted supply for customers in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized epoxy resin. The answers are derived directly from the experimental data and process descriptions found in patent CN101045781A, providing accurate insights for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this material into high-performance electronic packaging solutions.
Q: Why is low hydrolyzable chlorine content critical for electronic grade epoxy resins?
A: High chlorine content leads to corrosion of metal interconnects in integrated circuits and reduces the long-term reliability of semiconductor packages. The patented process achieves ≤200ppm chlorine, ensuring superior electrical insulation and device longevity.
Q: How does the phase transfer catalyst improve the synthesis compared to traditional methods?
A: Traditional methods use strong alkali for both etherification and epoxidation, causing hydrolysis of epichlorohydrin and polymerization side reactions. The phase transfer catalyst allows mild, selective reaction conditions, significantly increasing epoxy value and reducing by-products.
Q: What are the key physical properties of the synthesized resin?
A: The resulting resin is a white crystalline powder with an epoxy equivalent of 185-210g/eq, a softening point of 80-85°C, and a molecular weight distribution of 528-591, making it ideal for high-performance molding compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraphenol Ethane Epoxy Resin Supplier
As the demand for high-performance electronic materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex synthesis is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-quality results. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical hydrolyzable chlorine and epoxy value metrics defined in the patent. We understand that in the semiconductor sector, batch-to-batch consistency is not just a preference but a requirement, and our quality management systems are designed to ensure that every shipment meets the exacting standards necessary for IC packaging and advanced composite applications.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant operational efficiencies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of superior material science and reliable supply chain partnership.
