Advanced Synthesis of 5-Nitro Caffeic Acid Adamantanol Ester for Antitumor Drug Development
Advanced Synthesis of 5-Nitro Caffeic Acid Adamantanol Ester for Antitumor Drug Development
The pharmaceutical industry is constantly seeking novel intermediates that offer superior therapeutic profiles combined with robust manufacturability. Patent CN102993018A introduces a significant breakthrough in this domain with the disclosure of 5-nitro caffeic acid adamantanol ester, a compound designated as Formula (I) in the specification. This molecule represents a strategic evolution of caffeic acid phenethyl ester (CAPE) derivatives, engineered to enhance antitumor efficacy while maintaining a favorable safety profile. The patent details a sophisticated synthetic route that transforms simple starting materials into a high-value active pharmaceutical ingredient (API) intermediate capable of resisting gastric cancer, liver cancer, and leukemia. For R&D directors and procurement specialists alike, understanding the structural nuances and process efficiency of this compound is critical for integrating it into next-generation oncology pipelines.
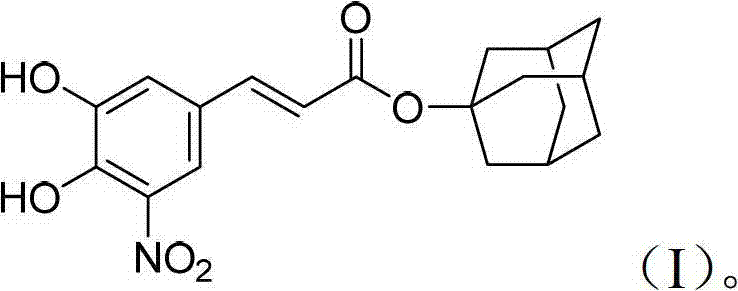
The core innovation lies not just in the biological potential of the molecule but in the elegance of its construction. By incorporating an adamantyl group, the inventors have modified the lipophilicity and metabolic stability of the parent caffeic acid structure. This structural modification is pivotal for improving bioavailability and targeting specific tumor microenvironments. As a reliable pharmaceutical intermediates supplier, recognizing the value of such structurally complex esters allows us to better serve clients who are pushing the boundaries of medicinal chemistry. The following analysis dissects the technical merits of this patent, offering a clear roadmap for those looking to source or manufacture this critical building block for antitumor drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing caffeic acid esters often rely on direct Fischer esterification or coupling reactions that can be plagued by low yields and difficult purification steps. In many legacy processes, the presence of multiple hydroxyl groups on the aromatic ring necessitates the use of protecting groups, which adds unnecessary synthetic steps, increases waste generation, and drives up the overall cost of goods sold (COGS). Furthermore, conventional methods frequently employ harsh acidic conditions or expensive coupling reagents that are not ideal for large-scale commercial production. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inconsistent batch-to-batch quality, which are unacceptable for GMP-compliant manufacturing environments. Additionally, the separation of unreacted starting materials and side products from the desired ester often requires extensive chromatography, a technique that is notoriously difficult to scale economically.
The Novel Approach
The methodology outlined in CN102993018A circumvents these traditional pitfalls through a cleverly designed tandem sequence involving alcoholysis and condensation. Instead of struggling with direct esterification of the acid, the process utilizes isopropylidene malonate as a reactive synthon. This strategy allows for the in situ generation of the ester linkage with 1-adamantanol under reflux conditions, followed immediately by a condensation reaction with 3,4-dihydroxy-5-nitrobenzaldehyde. This one-pot or streamlined approach significantly reduces the number of isolation steps required, thereby minimizing material loss and solvent consumption. The use of toluene as a solvent and piperidine as a catalyst creates a homogeneous reaction environment that promotes high conversion rates. By eliminating the need for complex protecting group strategies and harsh reagents, this novel approach offers a cleaner, more atom-economical pathway that is inherently more suitable for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Piperidine-Catalyzed Condensation
The heart of this synthetic transformation is a base-catalyzed condensation reaction, likely proceeding through a Knoevenagel-type mechanism facilitated by piperidine. In this mechanistic pathway, piperidine acts as a nucleophilic catalyst, activating the methylene group of the malonate derivative to form a reactive enamine or carbanion intermediate. This activated species then attacks the carbonyl carbon of the 3,4-dihydroxy-5-nitrobenzaldehyde, forming a new carbon-carbon bond. The subsequent elimination of water or alcohol moieties leads to the formation of the conjugated alkene system observed in the final product. The presence of the electron-withdrawing nitro group on the aromatic ring further activates the aldehyde towards nucleophilic attack, enhancing the reaction kinetics. Understanding this mechanism is vital for process chemists, as it highlights the importance of precise stoichiometry and temperature control to prevent polymerization or side reactions that could compromise the purity of the final intermediate.
Impurity control is another critical aspect addressed by the specific reaction conditions and workup procedures described in the patent. The use of pyridine as an acid binding agent helps to neutralize any acidic byproducts generated during the reaction, preventing the degradation of the sensitive catechol moiety. Following the reaction, the purification protocol involves a series of aqueous washes with dilute hydrochloric acid and sodium bicarbonate solution. This step is essential for removing residual catalysts, unreacted aldehydes, and acidic impurities that could affect the stability of the final API. The final recrystallization from a 1:1 ether and benzene mixed solvent system ensures the removal of trace organic impurities, yielding a product with the high purity specifications required for clinical applications. This rigorous attention to detail in the purification stage underscores the feasibility of producing high-purity pharmaceutical intermediates on a commercial scale.
How to Synthesize 5-Nitro Caffeic Acid Adamantanol Ester Efficiently
The synthesis of this valuable antitumor intermediate is achieved through a optimized sequence that balances reaction efficiency with operational simplicity. The process begins with the preparation of the key aldehyde precursor, followed by the crucial esterification-condensation cascade. Operators must pay close attention to the molar ratios of the reactants, specifically maintaining a ratio of approximately 1:1.25:1.25 for the aldehyde, malonate, and adamantanol respectively, to maximize yield. The reaction temperature is carefully controlled between 100°C and 120°C to ensure complete conversion without thermal degradation. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency.
- Perform alcoholysis reaction by refluxing isopropylidene malonate and 1-adamantanol in toluene for 3 to 5 hours.
- Add 3,4-dihydroxy-5-nitrobenzaldehyde, pyridine, and piperidine to the cooled reaction mixture.
- Conduct condensation reaction at 100-120°C for 10 to 24 hours, followed by purification via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical curiosity. The streamlined nature of the process directly translates to operational efficiencies that can significantly impact the bottom line. By reducing the number of unit operations and simplifying the purification workflow, manufacturers can achieve faster turnaround times and lower production costs. This efficiency is particularly valuable in the competitive landscape of oncology drug development, where speed to market is a critical success factor. Furthermore, the use of readily available and relatively inexpensive starting materials ensures a stable supply chain that is less susceptible to raw material volatility.
- Cost Reduction in Manufacturing: The elimination of protecting group chemistry and the use of a tandem reaction sequence drastically simplify the manufacturing process. This reduction in synthetic complexity means fewer reactors are tied up for shorter periods, leading to substantial cost savings in terms of labor, energy, and equipment usage. Additionally, the avoidance of expensive coupling reagents and chromatographic purification steps further drives down the variable costs associated with production. These factors combine to create a highly cost-effective manufacturing route that enhances the commercial viability of the final drug product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, pyridine, and piperidine ensures that the supply chain remains robust and resilient. Unlike processes that depend on exotic catalysts or specialized reagents with long lead times, this method utilizes materials that are widely available from multiple global suppliers. This diversity in sourcing options mitigates the risk of supply disruptions and allows for greater flexibility in procurement planning. Consequently, partners can secure a consistent flow of high-quality intermediates, ensuring uninterrupted production schedules for downstream drug formulation.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale batch processing, utilizing standard glass-lined or stainless steel reactors found in most fine chemical facilities. The solvent system, primarily based on toluene, allows for efficient recovery and recycling, aligning with modern green chemistry principles and environmental regulations. The simplified workup procedure generates less hazardous waste compared to traditional methods, reducing the burden on waste treatment facilities. This scalability and environmental compatibility make the process an attractive option for companies looking to expand their production capacity while adhering to strict sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-nitro caffeic acid adamantanol ester. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of incorporating this intermediate into their specific development programs.
Q: What is the biological activity of 5-Nitro Caffeic Acid Adamantanol Ester?
A: The compound exhibits broad-spectrum antitumor activity, specifically showing significant inhibition against human gastric cancer (SGC-7901), liver cancer (HepG2), and leukemia (HL-60) cell lines with low acute toxicity.
Q: What are the key reaction conditions for this synthesis?
A: The synthesis utilizes toluene as a polar solvent with piperidine as a condensation catalyst and pyridine as an acid binding agent, operating effectively at temperatures between 100°C and 120°C.
Q: How is the purity of the final product ensured?
A: High purity is achieved through a rigorous workup process involving solvent removal, washing with dilute HCl and NaHCO3 solution, and final recrystallization using a 1:1 ether and benzene mixed solvent system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro Caffeic Acid Adamantanol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced intermediates like 5-nitro caffeic acid adamantanol ester in the fight against cancer. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory to the marketplace. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch we produce adheres to the highest industry standards, providing you with the confidence needed to advance your clinical trials.
We invite you to collaborate with us to unlock the full commercial potential of this innovative compound. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to your success, offering both the technical expertise and the manufacturing capacity required to bring life-saving antitumor drugs to patients worldwide.
