Advanced Synthesis of Solanesyl Polyamine Derivatives for Oncology Drug Discovery
The pharmaceutical industry is constantly seeking novel scaffolds that combine natural product stability with synthetic versatility, and Patent CN101104609B presents a significant advancement in this domain by disclosing a series of Solanesyl polyamine derivatives with potent antitumor potential. This technology leverages the unique all-trans steric configuration of solanesol, a natural polyterpene found in tobacco and potato leaves, and conjugates it with polyamine structures known to enhance cellular uptake and biological activity. The core innovation lies in a robust synthetic methodology that preserves the delicate stereochemistry of the solanesol unit while introducing diverse aromatic functionalities through a piperazine linker. By optimizing the connection between the lipophilic terpene tail and the pharmacophoric aromatic head group, this patent provides a library of compounds that exhibit significantly improved cytotoxicity compared to previous analogs. For R&D teams focused on oncology, this represents a critical opportunity to access high-purity intermediates that serve as powerful lead compounds for next-generation cancer therapies. 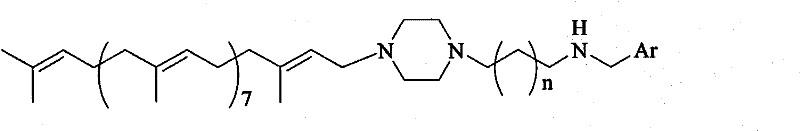
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the structural modification of solanesol has been fraught with challenges, primarily due to the sensitivity of its nine isoprene units to acidic or thermal degradation which often leads to cis-trans isomerization and loss of biological efficacy. Traditional methods for attaching nitrogenous bases to terpene backbones frequently require aggressive reaction conditions, such as high temperatures or strong Lewis acids, which can compromise the integrity of the polyterpene chain. Furthermore, earlier attempts to synthesize solanesol derivatives often resulted in complex mixtures that were difficult to purify, leading to low overall yields and inconsistent batch-to-batch quality. The lack of a standardized, mild protocol for introducing polyamine segments meant that many promising candidates were abandoned during the early discovery phase due to supply chain bottlenecks and prohibitive manufacturing costs. Additionally, conventional routes often failed to maintain the strict all-trans configuration required for optimal interaction with biological targets, rendering the resulting compounds less effective as antitumor agents.
The Novel Approach
The methodology described in CN101104609B overcomes these historical barriers by employing a mild reductive amination strategy that operates effectively at temperatures between 0°C and 20°C, thereby safeguarding the stereochemical purity of the solanesol backbone. This novel approach utilizes N-solanesyl-N'-aminoalkylpiperazine as a versatile building block, allowing for the facile introduction of various aromatic aldehydes to create a diverse array of derivatives without the need for complex protection-deprotection sequences. The process is characterized by its operational simplicity, involving straightforward solvent evaporation and standard reduction steps that are easily adaptable to large-scale reactors. By avoiding harsh conditions, this method ensures that the final products retain the native all-trans steric configuration, which is crucial for their observed high cytotoxicity against leukemia and cervical cancer cell lines. Moreover, the use of common reducing agents and accessible solvents makes this route economically viable and environmentally friendlier than previous heavy-metal catalyzed alternatives.
Mechanistic Insights into Reductive Amination of Solanesyl Amines
The core chemical transformation in this patent involves a two-step one-pot sequence beginning with the condensation of a secondary amine, specifically the N-solanesyl-N'-aminoalkylpiperazine, with an aromatic aldehyde to form an imine or iminium intermediate. This condensation step is kinetically controlled and proceeds efficiently in solvents like dichloromethane or methanol, where the nucleophilic nitrogen attacks the carbonyl carbon of the aldehyde. The reaction mixture is typically stirred for 2 to 24 hours at low temperatures to ensure complete conversion while minimizing side reactions such as polymerization of the aldehyde. Following the formation of the imine intermediate, the solvent is removed, and the residue is subjected to reduction using agents such as sodium borohydride, lithium aluminum hydride, or sodium cyanoborohydride. This reduction step converts the unstable imine bond into a stable secondary amine linkage, permanently locking the aromatic group onto the polyamine scaffold.
From an impurity control perspective, the mechanism is advantageous because the mild conditions prevent the isomerization of the solanesol double bonds, which is a common degradation pathway in terpene chemistry. The use of column chromatography for final purification allows for the precise separation of the desired all-trans product from any minor cis-isomers or unreacted starting materials that may form. The stoichiometry is carefully balanced, typically using a molar ratio of amine to aldehyde to reducing agent of 1:1~3:3~8, ensuring that the aldehyde is fully consumed and excess reducing agent does not interfere with downstream processing. This mechanistic precision results in products with high chemical purity, as evidenced by the sharp spectral data and consistent elemental analysis reported in the patent examples. The retention of the all-trans configuration is critical, as it mimics the natural biosynthetic precursor, facilitating better recognition by cellular transport mechanisms and enhancing the drug-like properties of the final molecule.
How to Synthesize Solanesyl Polyamine Derivatives Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must strictly adhere to the temperature controls and stoichiometric ratios defined in the patent to maximize yield and purity. The process begins with the dissolution of the amine precursor in a dry organic solvent, followed by the slow addition of the aromatic aldehyde to manage the exotherm and ensure homogeneous mixing. Detailed standard operating procedures regarding the specific workup protocols, including washing steps with sodium hydroxide solution and drying over anhydrous salts, are essential for removing inorganic byproducts before the final chromatographic purification.
- Dissolve N-solanesyl-N'-aminoalkylpiperazine in an organic solvent such as dichloromethane or methanol.
- Add the selected aromatic aldehyde (e.g., benzaldehyde, naphthaldehyde) and react at 0-20°C for 2-24 hours to form the imine intermediate.
- Remove the solvent, redissolve the residue, and add a reducing agent like sodium borohydride or lithium aluminum hydride at 0°C to complete the reduction to the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits by simplifying the sourcing of raw materials and reducing the complexity of the manufacturing process. The reliance on commercially available aromatic aldehydes and standard reducing agents eliminates the need for exotic or tightly regulated reagents, thereby stabilizing the supply chain against market volatility. Furthermore, the mild reaction conditions translate directly into lower energy consumption and reduced safety risks associated with high-pressure or high-temperature operations, contributing to a safer and more sustainable production environment. The high yields reported in the patent examples suggest that material throughput can be optimized, reducing the cost of goods sold and improving the overall margin profile for these high-value intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, bulk-grade solvents significantly lowers the direct material costs associated with production. By avoiding complex multi-step protection strategies, the process reduces labor hours and reactor occupancy time, leading to a more efficient utilization of manufacturing assets. The straightforward workup procedure minimizes solvent usage and waste generation, which further drives down disposal costs and environmental compliance expenses. Consequently, the overall cost structure for producing these solanesyl derivatives is markedly lower than that of traditional terpene modification routes.
- Enhanced Supply Chain Reliability: Since the key starting materials, such as solanesol and common aromatic aldehydes, are sourced from abundant natural or petrochemical feedstocks, the risk of supply disruption is minimal. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in utility supplies or ambient conditions, ensuring consistent output even in varying operational environments. This reliability allows for more accurate forecasting and inventory planning, enabling pharmaceutical companies to maintain steady production schedules for their downstream drug development programs without fear of intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to enlarge, such as cryogenic reactions or specialized filtration steps, making the transition from gram-scale to ton-scale production seamless. The use of standard organic solvents that can be readily recovered and recycled aligns with modern green chemistry principles and helps facilities meet stringent environmental regulations. Additionally, the absence of heavy metal residues in the final product simplifies the regulatory filing process for new drug applications, as extensive testing for metal impurities is not required, accelerating the time to market for potential therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these solanesyl polyamine derivatives, based on the detailed specifications provided in the patent literature. Understanding these aspects is crucial for partners looking to integrate this technology into their existing R&D pipelines or manufacturing portfolios.
Q: What is the primary advantage of this Solanesyl polyamine synthesis route?
A: The primary advantage is the use of mild reaction conditions (0-20°C) and standard reducing agents, which avoids harsh thermal stress on the sensitive all-trans solanesol chain while achieving high yields.
Q: Can this method be scaled for industrial production of antitumor intermediates?
A: Yes, the process utilizes common organic solvents and straightforward workup procedures like solvent evaporation and column chromatography, making it highly suitable for commercial scale-up.
Q: What represents the key structural feature of these derivatives?
A: The key feature is the conjugation of a lipophilic solanesyl tail with a hydrophilic polyamine-piperazine head and an aromatic moiety, which enhances cytotoxicity against tumor cells like L1210 and Hela.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solanesyl Polyamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of Solanesyl polyamine derivatives as pivotal intermediates in the development of novel antitumor therapies, and we are uniquely positioned to support your research and commercial needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early discovery phase or preparing for clinical trials. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Solanesyl polyamine derivative meets the highest standards of chemical integrity and stereochemical consistency required for pharmaceutical applications.
We invite you to contact our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall development costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and reliability as your long-term partner in fine chemical synthesis.
