Advanced Synthesis of Novel Solanesol Derivatives for Oncology Applications and Commercial Scale-Up
Advanced Synthesis of Novel Solanesol Derivatives for Oncology Applications and Commercial Scale-Up
The pharmaceutical landscape is constantly evolving, driven by the need for more effective antitumor agents with reduced toxicity profiles. A significant breakthrough in this domain is documented in patent CN101104608A, which discloses a series of novel amide-type solanesol derivatives. These compounds represent a strategic evolution in the modification of natural polyterpene alcohols, specifically targeting the preservation of the critical all-trans stereoconfiguration inherent to solanesol. Unlike previous iterations that may have compromised structural integrity during synthesis, this technology leverages a mild amide coupling strategy to link N-solanesyl-N'-aminoalkylpiperazine with various functionalized aromatic acids. For R&D directors and procurement specialists seeking a reliable solanesol derivative supplier, this patent offers a robust pathway to high-purity intermediates that demonstrate superior antitumor synergism compared to earlier generations of solanesol-based compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical modification of solanesol and its derivatives has been fraught with challenges related to the stability of the polyisoprene chain. Conventional synthetic routes often necessitate harsh reaction conditions, including elevated temperatures or strong acidic environments, which pose a severe risk of inducing cis-trans isomerization. Such stereochemical degradation not only diminishes the biological efficacy of the final product but also complicates the purification process, leading to lower overall yields and increased production costs. Furthermore, traditional methods for linking bioactive moieties to the solanesol backbone frequently suffer from poor regioselectivity and the formation of difficult-to-remove byproducts. These inefficiencies create significant bottlenecks in the supply chain for high-purity pharmaceutical intermediates, forcing manufacturers to contend with inconsistent batch quality and extended lead times for complex terpenoid derivatives.
The Novel Approach
The methodology outlined in the patent data introduces a transformative approach by utilizing carbodiimide-mediated amide coupling under strictly controlled mild conditions. By maintaining the reaction temperature between 0°C and 25°C, the process effectively eliminates the thermal energy required for double-bond isomerization, thereby locking the solanesol unit in its biologically active all-trans form. This novel route employs efficient coupling agents such as dicyclohexylcarbodiimide (DCC) or diisopropylcarbodiimide (DIC) in combination with additives like N-hydroxysuccinimide (NHS) or 1-hydroxybenzotriazole (HOBt). This catalytic system activates the carboxylic acid component gently, facilitating a rapid and high-yielding nucleophilic attack by the piperazine nitrogen. The result is a streamlined synthesis that not only simplifies post-reaction workup through standard filtration and washing but also delivers products with exceptional purity, addressing the critical demands for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Carbodiimide-Mediated Amide Coupling
The core of this synthetic innovation lies in the precise mechanistic execution of the amide bond formation, which serves as the linchpin for the molecule's stability and function. The reaction initiates with the activation of the raw material acid, such as 5-fluorouracil-1-acetic acid or biphenyl acetic acid, by the carbodiimide reagent to form an unstable O-acylisourea intermediate. This intermediate is highly susceptible to rearrangement into an unreactive N-acylurea; however, the presence of the nucleophilic additive (NHS or HOBt) intercepts this species to generate a stable active ester. This active ester is significantly more electrophilic towards the amine nucleophile, N-solanesyl-N'-aminoalkylpiperazine, ensuring that the coupling proceeds efficiently even at ambient temperatures. This mechanism is crucial because it bypasses the need for thermal activation, which is the primary driver of degradation in sensitive polyterpene systems.
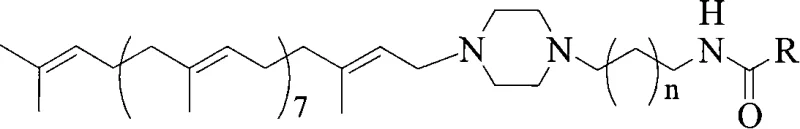
Furthermore, the structural diversity achievable through this mechanism is vast, allowing for the fine-tuning of pharmacological properties. As illustrated in the general formula, the 'R' group can be varied extensively, ranging from halogenated pyrimidinyl methyl groups to bulky aromatic systems like 6-alkoxynaphthalene substituted ethyl groups. This modularity enables medicinal chemists to optimize the lipophilicity and steric profile of the derivative to enhance cell membrane permeability and target binding affinity. The preservation of the solanesol tail, a long hydrophobic chain composed of nine isoprene units, ensures that the molecule retains its ability to integrate into lipid bilayers, a key factor in its observed synergistic activity with other antitumor agents. The rigorous control over impurity profiles, achieved by avoiding side reactions common in harsher conditions, means that the final API intermediates meet stringent regulatory standards with minimal downstream processing.
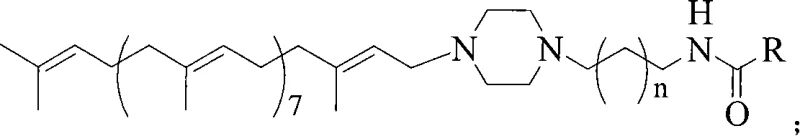
How to Synthesize Solanesol Derivatives Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to maximize the benefits of the mild coupling conditions. The process begins with the dissolution of the specific aromatic acid and the coupling catalysts in a halogenated solvent such as chloroform or dichloromethane, creating a homogeneous reaction medium. Following the activation period, the solanesyl-piperazine precursor is introduced, and the mixture is allowed to react for a duration of 2 to 48 hours, depending on the steric hindrance of the specific acid employed. The detailed standardized synthesis steps, including specific molar ratios and purification protocols validated by experimental data, are provided below to guide your technical team in replicating these high-yield results.
- Dissolve the corresponding raw material acid (e.g., 5-fluorouracil-1-acetic acid or biphenyl acetic acid) and coupling agents (DCC/DIC and HOBt/NHS) in an organic solvent like chloroform or dichloromethane at 0-25°C.
- Add N-solanesyl-N'-aminoalkylpiperazine to the activated acid mixture and stir for 2 to 48 hours to ensure complete amide bond formation while preserving stereochemistry.
- Filter the reaction mixture, wash the filtrate with alkali solution to remove unreacted acid, dry the organic layer, and purify the final product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic advantages beyond mere technical superiority. The elimination of extreme temperature requirements translates directly into reduced energy consumption and lower capital expenditure on specialized cryogenic reactors, facilitating a more agile and cost-effective production environment. Moreover, the high reaction yields reported in the patent data, reaching upwards of 81 percent in specific embodiments, signify a drastic reduction in raw material waste. This efficiency is paramount when dealing with complex natural product derivatives where the starting materials, such as high-purity solanesol extracted from tobacco leaves, represent a significant portion of the cost of goods sold. By minimizing loss during the transformation step, manufacturers can achieve significant cost savings and improve the overall margin profile of the final oncology intermediates.
- Cost Reduction in Manufacturing: The adoption of carbodiimide coupling agents, while seemingly expensive on a per-gram basis, actually drives down total manufacturing costs by simplifying the purification workflow. The formation of dicyclohexylurea (DCU) as a byproduct allows for easy removal via simple filtration, obviating the need for complex aqueous extractions or chromatographic separations at the intermediate stage. This streamlining of the workup process reduces solvent usage and labor hours, contributing to a leaner manufacturing operation. Additionally, the robustness of the reaction conditions means that batch-to-batch variability is minimized, reducing the financial risk associated with failed runs and reprocessing.
- Enhanced Supply Chain Reliability: Sourcing high-quality solanesol derivatives has historically been challenging due to the complexity of maintaining stereochemical integrity during transport and storage. However, the stability imparted by the amide linkage in these new derivatives enhances their shelf-life and resistance to degradation. This chemical stability allows for more flexible logistics planning and reduces the pressure on just-in-time delivery models. Furthermore, the scalability of the reaction, which does not rely on scarce transition metal catalysts or air-sensitive reagents, ensures that supply can be ramped up rapidly to meet surging demand in the oncology sector without encountering bottlenecks related to specialized catalyst availability.
- Scalability and Environmental Compliance: From an environmental perspective, the use of standard organic solvents like dichloromethane and chloroform, which are easily recovered and recycled in modern facilities, aligns well with green chemistry initiatives when managed correctly. The absence of heavy metal catalysts eliminates the need for expensive and wasteful metal scavenging steps, which are often a regulatory hurdle in pharmaceutical production. The process generates solid urea byproducts that are easier to handle and dispose of compared to the toxic sludge often associated with traditional coupling methods. This cleaner profile facilitates smoother regulatory approvals and supports the corporate sustainability goals of major pharmaceutical partners seeking eco-friendly supply chains.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries regarding the synthesis and application of these solanesol derivatives. These insights are derived directly from the experimental data and background analysis of the patent, providing a transparent view of the compound's potential. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating these intermediates into their drug development pipelines.
Q: How does this synthesis method preserve the biological activity of solanesol?
A: The method utilizes mild reaction conditions (0-25°C) and avoids harsh reagents that could cause isomerization. This ensures the original all-trans stereoconfiguration of the solanesol unit is maintained, which is critical for its antitumor and synergistic properties.
Q: What are the primary applications of these solanesol derivatives?
A: These compounds exhibit significant antitumor drug activity and act as potent synergists for clinical anticancer agents like vincristine (VCR), enhancing cytotoxicity against tumor cells such as L1210 and CHO.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features simple operation, mild temperature requirements, and high reaction yields (up to 81.1%), making it highly suitable for industrial scale-up without the need for complex cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solanesol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of these novel solanesol derivatives in the next generation of antitumor therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the most stringent purity specifications, utilizing our rigorous QC labs to verify the retention of the critical all-trans stereoconfiguration in every batch. Our capability to handle complex terpenoid chemistry positions us as a strategic partner for global pharmaceutical companies aiming to accelerate their oncology programs.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this advanced manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
