Advanced Synthesis of Coumarin-Containing Polymethine Cyanine Dyes for High-Performance Electronic Applications
Advanced Synthesis of Coumarin-Containing Polymethine Cyanine Dyes for High-Performance Electronic Applications
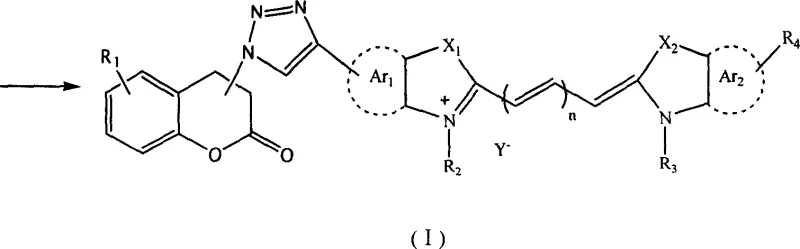
The rapid evolution of optoelectronic materials and biological imaging technologies has necessitated the development of fluorophores that combine high quantum efficiency with exceptional chemical stability. In this context, patent CN1869129A introduces a groundbreaking class of coumarin-containing polymethine cyanine dye derivatives that address critical limitations in current dye sensitization and fluorescence probing. This technology leverages the precision of copper-catalyzed click chemistry to covalently link coumarin moieties to polymethine chains, resulting in compounds with tunable absorption spectra ranging across the visible region. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a significant opportunity to access high-performance intermediates that offer superior solvability and thermal conductivity compared to traditional cyanine architectures. The strategic integration of the coumarin scaffold not only enhances the fluorescence quantum yield but also provides versatile sites for further chemical modification, making these derivatives ideal candidates for next-generation solar cells and protein research tools.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polymethine cyanine dyes has relied on condensation reactions that often suffer from poor regioselectivity and limited structural diversity. Traditional methods typically involve the reaction of quaternary nitrogen heterocycles with activated methylene groups, a process that can be sensitive to moisture and requires rigorous control of stoichiometry to prevent the formation of unwanted oligomers. Furthermore, extending the conjugated methine chain to achieve red-shifted absorption often compromises the thermal and photochemical stability of the resulting dye, leading to rapid degradation under operational conditions. Conventional linkers between functional groups, such as simple ester or ether bonds, may also exhibit hydrolytic instability in physiological or electrolyte environments, restricting their utility in long-term applications like dye-sensitized solar cells (DSSCs). These inherent drawbacks create significant bottlenecks in the supply chain, as manufacturers struggle to produce batches with consistent purity and performance characteristics, ultimately driving up the cost of goods for downstream electronic and biomedical applications.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing a copper-catalyzed azide-alkyne cycloaddition (CuAAC) strategy, widely recognized as a premier "click chemistry" reaction. This approach allows for the modular assembly of complex dye structures under mild conditions, typically between 10°C and 150°C, using readily available solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). By reacting an azide-functionalized coumarin precursor with an alkyne-substituted cyanine intermediate, the process forms a robust 1,2,3-triazole ring that serves as a stable, conjugated bridge. This structural motif not only preserves the electronic communication between the coumarin and the polymethine chain but also significantly enhances the overall rigidity and stability of the molecule. The versatility of this method is evidenced by its compatibility with various catalytic systems, including CuI, CuBr, and CuSO4/sodium ascorbate, allowing manufacturers to optimize the process for cost or environmental compliance without sacrificing yield.
Mechanistic Insights into Copper-Catalyzed Azide-Alkyne Cycloaddition
The core of this synthetic innovation lies in the mechanistic efficiency of the Cu(I)-catalyzed cycloaddition, which proceeds through a metallacycle intermediate to ensure exclusive formation of the 1,4-disubstituted 1,2,3-triazole product. Unlike thermal Huisgen cycloadditions which require high temperatures and yield mixtures of regioisomers, the copper-catalyzed variant operates efficiently at temperatures as low as 50°C to 60°C, minimizing thermal stress on the sensitive polymethine backbone. The catalytic cycle involves the coordination of the terminal alkyne to the copper center, followed by deprotonation to form a copper-acetylide species. This nucleophilic species then attacks the terminal nitrogen of the organic azide, initiating ring closure. The resulting triazole ring is aromatic and chemically inert, providing a thermodynamically stable linkage that resists hydrolysis and enzymatic degradation, which is crucial for in vivo fluorescence imaging applications. This mechanistic pathway ensures that the extended pi-conjugation system remains intact, preserving the high molar extinction coefficient characteristic of cyanine dyes while imparting the favorable photophysical properties of the coumarin unit.
From an impurity control perspective, this reaction pathway offers distinct advantages over traditional condensation routes. The high specificity of the click reaction minimizes the formation of side products, simplifying the downstream purification process. In the provided embodiments, crude products are often purified simply by precipitation in ethanol followed by column chromatography, achieving isolated yields ranging from 76% to 90%. The absence of complex byproduct profiles reduces the burden on analytical quality control teams, who can rely on standard HPLC and NMR techniques to verify purity without needing to separate difficult-to-remove isomeric impurities. Furthermore, the choice of counterions (Y-) such as iodide, perchlorate, or hexafluorophosphate allows for fine-tuning of the dye's solubility and crystallization behavior, facilitating the production of high-purity solids suitable for vacuum deposition in electronic device fabrication.
How to Synthesize Coumarin Cyanine Dyes Efficiently
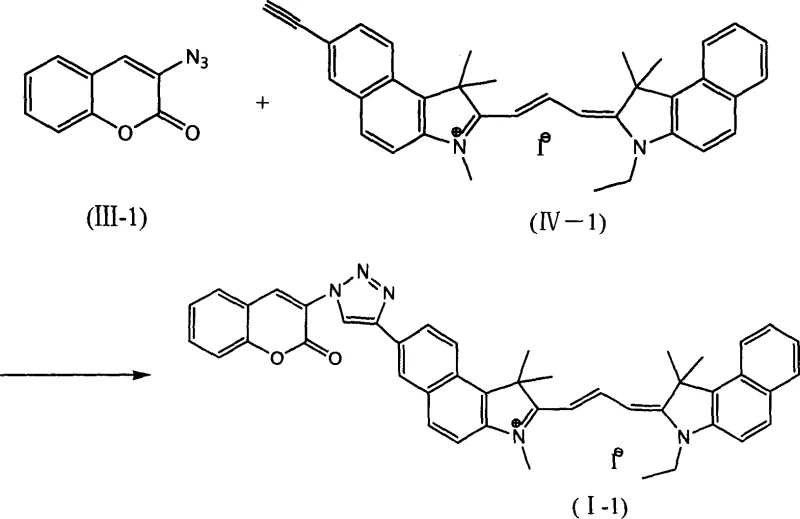
The practical implementation of this technology involves a straightforward protocol that balances reaction kinetics with product stability. As demonstrated in the patent embodiments, the process begins with the dissolution of the azide-coumarin and alkyne-cyanine precursors in a polar aprotic solvent, followed by the addition of the copper catalyst and a base such as diisopropylethylamine. The reaction is typically conducted under an inert argon atmosphere to prevent the oxidation of the Cu(I) species to inactive Cu(II), although some embodiments successfully utilize Cu(II) salts reduced in situ by sodium ascorbate. Detailed standardized synthesis steps for scaling this reaction are provided in the guide below, ensuring reproducibility across different manufacturing sites.
- Prepare the reaction mixture by combining the azide-functionalized coumarin precursor and the alkyne-functionalized cyanine dye intermediate in a polar aprotic solvent such as DMF or DMSO.
- Introduce the copper catalyst system, such as CuI with diisopropylethylamine or CuSO4 with sodium ascorbate, under an inert argon atmosphere to prevent oxidation.
- Maintain the reaction temperature between 50°C and 60°C for 6 to 12 hours, monitoring progress via TLC, followed by precipitation in ethanol and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this click chemistry-based synthesis route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the manufacturing workflow, which translates directly into reduced operational expenditures. By eliminating the need for harsh reaction conditions and multi-step protection/deprotection sequences often required in traditional dye synthesis, manufacturers can significantly lower energy consumption and reduce the consumption of expensive reagents. The high atom economy of the cycloaddition reaction ensures that a large proportion of the starting material mass is incorporated into the final product, minimizing waste generation and lowering the cost of raw material inputs per kilogram of active dye produced. This efficiency is critical for maintaining competitive pricing in the high-volume markets of solar cell manufacturing and diagnostic reagents.
- Cost Reduction in Manufacturing: The streamlined nature of the CuAAC reaction eliminates the need for complex purification infrastructure. Since the reaction yields are consistently high (often exceeding 85% as seen in Embodiments 1, 3, and 6), the throughput of the production facility is maximized without the need for extensive recycling of unreacted starting materials. The ability to use common, commodity-grade solvents like ethanol for precipitation and purification further drives down the variable costs associated with solvent recovery and disposal. Additionally, the robustness of the triazole linkage reduces the risk of product degradation during storage and transport, minimizing losses due to shelf-life expiration and ensuring that the delivered material meets stringent specifications upon arrival.
- Enhanced Supply Chain Reliability: The modularity of this synthetic approach allows for flexible sourcing of raw materials. The azide and alkyne precursors can be synthesized independently and stockpiled, creating a buffer against supply disruptions for any single component. This decoupling of the supply chain enhances resilience, as manufacturers can qualify multiple vendors for the intermediate building blocks without compromising the quality of the final dye. Furthermore, the mild reaction conditions (50-60°C) reduce the dependency on specialized high-pressure or cryogenic reactor equipment, enabling production to be shifted more easily between different contract manufacturing organizations (CMOs) if capacity constraints arise, thereby securing continuity of supply for critical downstream applications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the exothermic nature of the click reaction, which is easily manageable with standard cooling jackets, removing the need for complex thermal runaway prevention systems. The use of copper catalysts, while requiring removal to meet heavy metal specifications for certain applications, is well-established in the industry with efficient scavenging resins available to ensure compliance with regulatory limits. The overall reduction in solvent volume and reaction time compared to traditional methods contributes to a smaller environmental footprint, aligning with the increasing corporate mandates for sustainable manufacturing practices and reducing the regulatory burden associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced dye materials. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners.
Q: What are the primary applications of these coumarin-containing cyanine dyes?
A: According to patent CN1869129A, these derivatives are specifically designed for use as fluorescent probes in protein research, agents for fluorescence ion electrophoresis, and sensitizing dyes for solid-state solar cells due to their high molar extinction coefficients.
Q: How does the click chemistry approach improve dye stability compared to traditional methods?
A: The formation of the 1,2,3-triazole ring via CuAAC creates a chemically robust and rigid linkage between the coumarin and the polymethine chain, significantly enhancing thermal stability and resistance to photobleaching compared to labile ester or ether linkages.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes mild reaction temperatures (50-60°C) and common organic solvents like DMF and ethanol, avoiding extreme pressure or cryogenic conditions, which facilitates straightforward scale-up from gram to kilogram quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Cyanine Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the coumarin-polymethine architecture described in CN1869129A for the fields of optoelectronics and biotechnology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of dye meets the exacting standards required for high-performance solar cells and sensitive fluorescent probes. Our commitment to quality assurance ensures that the optical properties of our materials remain consistent, providing our clients with the reliability they need for their own product development cycles.
We invite you to collaborate with us to optimize your supply chain for these critical functional materials. Whether you require custom modifications to the R-groups to tune absorption maxima or need assistance with regulatory filings for new dye formulations, our technical team is ready to support your goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
