Advanced Rhodamine Fluorescent Dyes for Multi-Color Detection and Commercial Scale-Up
Advanced Rhodamine Fluorescent Dyes for Multi-Color Detection and Commercial Scale-Up
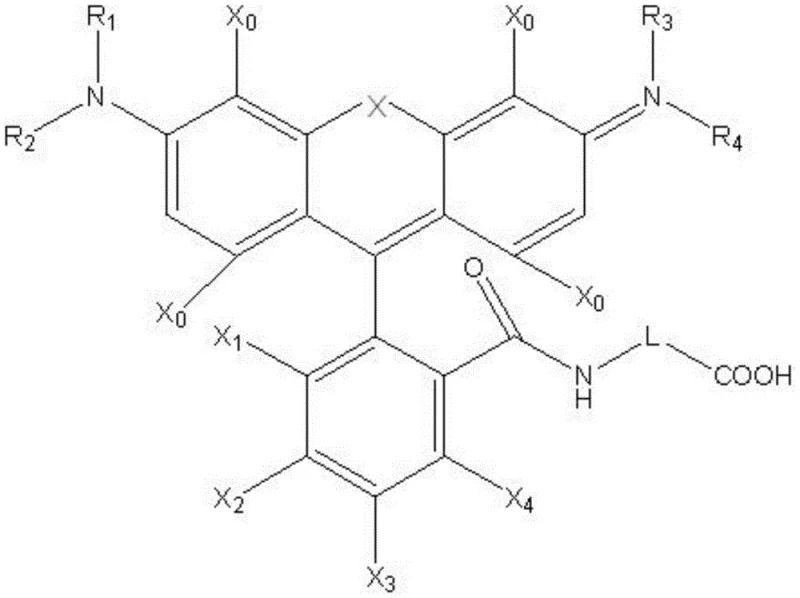
The rapid evolution of multiplex diagnostic technologies has created an urgent demand for fluorescent probes that offer superior spectral resolution and photostability. Patent CN113321638A addresses this critical gap by disclosing a novel class of high-performance fluorescent dyes specifically engineered for complex fluorescence detection scenarios, including advanced 8-color, 9-color, and 10-color test kits. Unlike traditional rhodamine derivatives which often suffer from limited thermal stability or insufficient quantum yields, this invention introduces a versatile structural framework where the central bridge atom X can be oxygen, carbon, or silicon. This structural flexibility allows for precise tuning of emission wavelengths across the visible to near-infrared spectrum, specifically targeting the 500nm to 800nm range. For R&D directors and procurement specialists in the life sciences sector, this patent represents a significant leap forward in reagent reliability, offering a pathway to synthesize dyes with quantum yields exceeding 50% while maintaining rigorous control over impurity profiles through a well-defined synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of multi-color flow cytometry and PCR kits has been bottlenecked by the scarcity of fluorophores that possess both high photostability and narrow emission bandwidths. Conventional rhodamine dyes often exhibit broad emission spectra, leading to significant spectral overlap that complicates data analysis in high-parameter assays. Furthermore, many existing silicon-rhodamine or carbon-rhodamine analogues are synthetically challenging to produce on a commercial scale, often requiring expensive transition metal catalysts or harsh conditions that degrade product purity. The lack of water-soluble variants with consistent batch-to-batch performance has forced manufacturers to rely on imported reagents with long lead times and inflated costs. Additionally, the thermal instability of many legacy dyes limits their shelf-life and utility in point-of-care devices that may be exposed to varying environmental conditions, thereby restricting the design of robust, all-in-one diagnostic platforms.
The Novel Approach
The methodology outlined in the patent data circumvents these historical hurdles by employing a streamlined activation strategy using phosphorus oxychloride (POCl3) to generate highly reactive intermediates under controlled inert atmospheres. This approach eliminates the need for precious metal catalysts, significantly reducing the risk of heavy metal contamination which is a critical quality attribute for biological reagents. By utilizing a modular synthesis where substituents R1 through R4 and X0 through X4 can be systematically varied, manufacturers can generate a library of dyes with emission wavelength differences of less than 5nm. This precision enables the design of tightly packed spectral channels for next-generation sequencing and multiplex immunoassays. The process also incorporates a final coupling step with amino acid derivatives like 3-methylaminobutyric acid, which not only enhances the solubility profile but also provides a convenient handle for bioconjugation to antibodies or nucleic acids without compromising the fluorescence quantum yield.
Mechanistic Insights into POCl3-Mediated Activation and Cyclization
The core of this synthetic innovation lies in the efficient transformation of the rhodamine lactone scaffold into a reactive electrophile capable of undergoing nucleophilic substitution. The mechanism initiates with the condensation of 3-N,N'-dimethylaminophenol and phthalic anhydride in concentrated sulfuric acid at elevated temperatures, typically around 180°C, to form the foundational xanthene structure. Subsequent treatment with phosphorus oxychloride serves a dual purpose: it acts as a dehydrating agent to drive the equilibrium forward and as a chlorinating agent to convert the hydroxyl or carbonyl functionalities into a highly reactive chloro-species. This activation step is conducted in 1,2-dichloroethane at 70°C under argon protection, ensuring that moisture-sensitive intermediates are preserved. The resulting intermediate is then subjected to nucleophilic attack by the amine group of the amino acid derivative in the presence of a base like triethylamine. This step is critical for installing the solubilizing and conjugating tail, and it is performed under ice bath conditions to suppress side reactions and ensure regioselectivity.
Controlling the impurity profile in such complex polycyclic aromatic systems is paramount for achieving high-purity fluorescent dyes suitable for clinical diagnostics. The patent specifies a rigorous workup procedure involving washing with slightly acidic water to remove unreacted amines and base residues, followed by extraction with dichloromethane. The use of silica gel column chromatography as the final purification step is particularly advantageous for removing trace isomers and oligomeric byproducts that could otherwise quench fluorescence or cause non-specific binding in assay formats. By strictly controlling the stoichiometry of the POCl3 activation step and the temperature during the coupling phase, the process minimizes the formation of decomposition products. This level of mechanistic control ensures that the final product meets the stringent purity specifications required for regulatory approval in medical device applications, providing a reliable supply of high-performance optical probes.
How to Synthesize High-Performance Rhodamine Dyes Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating these advanced fluorophores, starting from readily available commodity chemicals. The process is designed to be scalable, moving from gram-scale laboratory optimization to multi-kilogram production without significant loss in yield or quality. The key to success lies in the strict adherence to anhydrous conditions during the activation phase and the precise control of pH during the extraction steps. Detailed standard operating procedures for each reaction stage, including specific molar ratios and stirring times, are essential for maintaining consistency. For technical teams looking to implement this chemistry, the following guide outlines the critical operational parameters derived from the patent examples.
- Condense 3-N,N'-dimethylaminophenol with phthalic anhydride in concentrated sulfuric acid at 180°C to form the initial rhodamine lactone scaffold.
- Activate the scaffold by reacting with phosphorus oxychloride (POCl3) in 1,2-dichloroethane at 70°C under inert gas protection to generate the reactive chloro-intermediate.
- Couple the activated intermediate with 3-methylaminobutyric acid and triethylamine in acetonitrile under ice bath conditions, followed by extraction and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits by decoupling production from volatile markets for precious metal catalysts. Traditional methods for synthesizing silicon-rhodamines often rely on palladium or copper catalysis, which introduces supply chain risks associated with the sourcing and price fluctuation of these rare earth metals. By shifting to a phosphorus-based activation chemistry, manufacturers can secure a more stable and cost-effective raw material base. The starting materials, such as phthalic anhydride and dimethylaminophenol, are bulk industrial chemicals with established global supply chains, ensuring continuous availability even during periods of market disruption. This resilience is critical for maintaining the production schedules of high-volume diagnostic kits where reagent consistency is non-negotiable.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts directly translates to lower raw material costs and simplified downstream processing. Without the need for specialized scavengers to remove trace metals, the purification workflow is drastically simplified, reducing solvent consumption and waste disposal costs. The use of standard silica gel chromatography instead of preparative HPLC for final purification further drives down operational expenditures, making the commercial production of these high-value dyes economically viable. Additionally, the high quantum yield of the final product means that less dye is required per test unit, effectively lowering the cost-in-use for the end customer while maintaining margin integrity for the manufacturer.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route allows for flexible manufacturing strategies, including the potential for toll manufacturing or multi-site production to mitigate geographic risks. Since the reaction conditions do not require exotic equipment or extreme cryogenic temperatures beyond standard industrial capabilities, the technology can be easily transferred to contract manufacturing organizations (CMOs) with diverse capacity levels. This flexibility ensures that procurement managers can establish redundant supply sources, safeguarding against single-point failures. The stability of the intermediates also allows for the strategic stocking of key precursors, enabling rapid response to sudden spikes in demand for diagnostic reagents during public health emergencies.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction parameters that translate linearly from laboratory flasks to large-scale reactors, facilitating the commercial scale-up of complex fluorescent dyes. The solvent system, primarily utilizing dichloromethane and acetonitrile, is compatible with standard industrial recovery and recycling units, minimizing environmental impact and aligning with green chemistry initiatives. By avoiding the generation of heavy metal waste streams, the facility reduces its regulatory burden and liability associated with hazardous waste disposal. This environmental compliance not only lowers operational costs but also enhances the corporate sustainability profile, which is increasingly a deciding factor for multinational partnerships in the life sciences sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent dye technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the fit of these dyes for specific assay formats and for procurement teams assessing the long-term viability of the supply source.
Q: What is the quantum yield of the synthesized fluorescent dyes?
A: According to patent CN113321638A, the synthesized dyes exhibit a quantum yield of more than 50%, ensuring high emission efficiency suitable for sensitive detection applications.
Q: How can the emission wavelength be tuned in this dye series?
A: The emission wavelength is tunable by modifying the central bridge atom X (O, -C(CH3)2, or -Si(CH3)2) and varying the substituents R1-R4 and X0-X4, allowing for precise spectral separation less than 5nm between adjacent dyes.
Q: Are these dyes suitable for aqueous biological environments?
A: Yes, the molecular design allows for the introduction of water-soluble groups such as sulfonic acid (-SO3H) or phosphonic acid derivatives, enabling the synthesis of hydrophilic dyes adaptable for various biological application scenes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhodamine Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent dyes play in the advancement of precision medicine and diagnostic accuracy. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from research to market is seamless and efficient. We are committed to delivering products with stringent purity specifications and comprehensive analytical data packages, supported by our rigorous QC labs equipped with state-of-the-art HPLC and mass spectrometry instrumentation. Our capability to customize the substituents on the rhodamine core allows us to tailor the photophysical properties of the dyes to your specific instrument requirements, whether for flow cytometry, microscopy, or in vivo imaging applications.
We invite you to collaborate with us to leverage this patented technology for your next-generation diagnostic products. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that demonstrates how switching to our optimized synthetic route can improve your bottom line. We are prepared to provide specific COA data and route feasibility assessments to validate the performance of our high-purity rhodamine derivatives in your specific assay environment. Let us be your partner in driving innovation and efficiency in the competitive landscape of life science reagents.
