Advanced Synthesis of 7,7'-Bis(4-Methoxyphenyl)-6,6'-Dimethyl-2,3'-Bi(1,8-Naphthyridine) for Commercial Sensor Applications
Advanced Synthesis of 7,7'-Bis(4-Methoxyphenyl)-6,6'-Dimethyl-2,3'-Bi(1,8-Naphthyridine) for Commercial Sensor Applications
The escalating global concern regarding heavy metal contamination, particularly mercury, has necessitated the development of highly sensitive and selective detection tools for environmental and biological monitoring. Patent CN109180705B introduces a breakthrough fluorescent compound, chemically defined as 7,7'-bis(4-methoxyphenyl)-6,6'-dimethyl-2,3'-bi(1,8-naphthyridine), which exhibits superior performance in mercury ion (Hg2+) sensing. This novel organic luminescent material addresses critical limitations found in prior art, such as insufficient specificity and poor water solubility, by leveraging a robust bi-naphthyridine scaffold. The compound's unique electronic structure allows for significant fluorescence decay upon binding with Hg2+, providing a clear and quantifiable signal for trace detection. As a reliable fluorescent probe supplier, understanding the structural integrity and synthesis of such advanced materials is paramount for developing next-generation sensors.
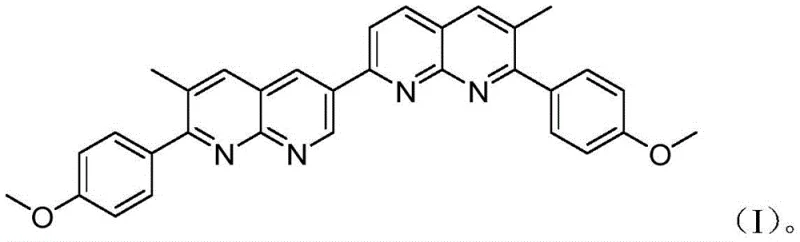
Beyond its analytical capabilities, the commercial viability of this compound is underpinned by its synthetic accessibility. The patent outlines a concise two-step preparation method starting from inexpensive and commercially abundant precursors: 2-amino-3-pyridinecarboxaldehyde and p-methoxypropiophenone. This strategic design minimizes the number of unit operations required, directly translating to potential cost reduction in electronic chemical manufacturing. For R&D teams focused on sensor fabrication, the compound's stability in aqueous environments is a decisive factor, enabling direct application in water quality testing without extensive sample pretreatment. The integration of methoxy groups enhances solubility profiles while maintaining the rigidity of the conjugated system necessary for efficient fluorescence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for complex heterocyclic fluorescent probes often suffer from excessive step counts, requiring intricate protection and deprotection strategies that drastically reduce overall yield and increase production costs. Many existing mercury probes rely on structures that are difficult to synthesize on a large scale, involving hazardous reagents or extreme reaction conditions that pose significant safety risks in a manufacturing setting. Furthermore, conventional probes frequently exhibit cross-reactivity with other transition metals like copper or iron, leading to false positives that compromise the reliability of environmental monitoring data. The reliance on organic solvents with poor water miscibility in older methodologies also limits their utility in biological systems or direct water analysis, necessitating additional formulation steps that add complexity and expense to the final sensor device assembly.
The Novel Approach
The methodology described in CN109180705B represents a paradigm shift by utilizing a direct condensation followed by an oxidative coupling strategy to construct the bi-naphthyridine core efficiently. This approach eliminates the need for toxic heavy metal catalysts in the initial cyclization step, relying instead on potassium tert-butoxide, a standard and manageable base. The second step employs a versatile range of transition metal catalysts, allowing manufacturers to optimize for cost or availability without sacrificing reaction efficiency. By streamlining the synthesis into just two distinct stages, the process significantly reduces solvent consumption and waste generation, aligning with modern green chemistry principles. This simplified workflow not only accelerates the time-to-market for new sensor technologies but also ensures a consistent supply of high-purity OLED material precursors and sensing agents.
Mechanistic Insights into Base-Mediated Cyclization and Oxidative Coupling
The first stage of the synthesis involves a base-mediated condensation between 2-amino-3-pyridinecarboxaldehyde and p-methoxypropiophenone, facilitated by potassium tert-butoxide in solvents such as methanol or dichloromethane. This reaction likely proceeds through an initial aldol-type condensation followed by cyclization and aromatization to form the 1,8-naphthyridine ring system of the intermediate 3a. The choice of base and solvent is critical here, as it influences the rate of enolate formation and the subsequent nucleophilic attack on the aldehyde functionality. Operating at moderate temperatures between 25°C and 80°C ensures that the reaction proceeds smoothly without degrading the sensitive functional groups, preserving the methoxy moiety which is essential for the final compound's solubility and electronic properties. This mild condition profile is indicative of a robust process capable of tolerating minor variations in feedstock quality.
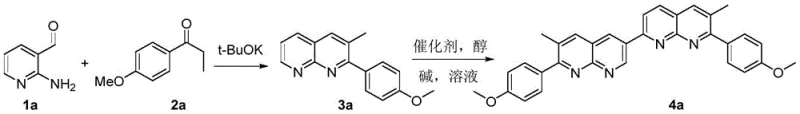
The second stage transforms the monomeric intermediate into the final dimeric target through a metal-catalyzed oxidative coupling reaction. This step typically involves heating the intermediate with a small molecular alcohol, a base, and a transition metal catalyst such as cobalt acetate, cuprous chloride, or palladium complexes under an inert atmosphere. The mechanism likely involves the activation of the C-H bond at the reactive position of the naphthyridine ring by the metal center, followed by radical coupling or reductive elimination to form the C-C bond linking the two units. The use of inert gases like nitrogen or argon prevents unwanted oxidation side reactions, ensuring high selectivity for the desired dimer. The flexibility in catalyst selection—ranging from earth-abundant cobalt and iron to noble palladium—provides a strategic lever for cost reduction in sensor manufacturing, allowing producers to balance catalytic activity against raw material expenses.
How to Synthesize 7,7'-Bis(4-Methoxyphenyl)-6,6'-Dimethyl-2,3'-Bi(1,8-Naphthyridine) Efficiently
The synthesis of this high-performance fluorescent probe is designed for operational simplicity and scalability, making it an ideal candidate for industrial adoption. The process begins with the precise stoichiometric mixing of the pyridine aldehyde and the ketone precursor in the presence of a strong base, followed by a controlled heating phase to drive the cyclization to completion. Once the intermediate is isolated and purified, it undergoes the critical coupling step where reaction parameters such as temperature (100-160°C) and time (5-48 h) are tuned to maximize conversion while minimizing byproduct formation. Detailed standard operating procedures for this synthesis, including specific workup and purification protocols, are essential for maintaining batch-to-batch consistency.
- Condense 2-amino-3-pyridinecarboxaldehyde with p-methoxypropiophenone using potassium tert-butoxide in a suitable solvent at 25-80°C to form the intermediate 2-(4-methoxyphenyl)-3-methyl-1,8-naphthyridine.
- Perform oxidative coupling of the intermediate using a transition metal catalyst (such as cobalt, copper, or palladium salts) and a base in a high-boiling solvent under inert atmosphere at 100-160°C.
- Purify the crude reaction mixture via filtration and column chromatography using a dichloromethane and ammonia-methanol eluent system to isolate the final fluorescent compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in this patent offers substantial advantages due to its reliance on commodity chemicals that are widely available in the global market. The primary starting materials, 2-amino-3-pyridinecarboxaldehyde and p-methoxypropiophenone, are established intermediates with stable supply chains, mitigating the risk of raw material shortages that often plague specialty chemical production. The elimination of exotic or highly regulated reagents simplifies the logistics of sourcing and storage, reducing the administrative burden on supply chain managers. Furthermore, the ability to use a variety of solvents and catalysts provides flexibility; if a specific metal salt becomes scarce or expensive, the process can be adapted to use an alternative without redesigning the entire workflow, ensuring supply chain reliability even in volatile market conditions.
- Cost Reduction in Manufacturing: The two-step nature of the synthesis inherently lowers processing costs by reducing the number of isolation and purification stages required compared to multi-step alternatives. The use of non-toxic, low-cost raw materials directly decreases the Bill of Materials (BOM) cost, while the moderate reaction conditions reduce energy consumption associated with heating and cooling. Additionally, the avoidance of complex protection group chemistry means fewer reagents are consumed per kilogram of product, leading to significant waste reduction and lower disposal costs. This economic efficiency allows for competitive pricing strategies when supplying commercial scale-up of complex optoelectronic materials.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures high process reliability and minimizes the risk of batch failures. Since the catalysts employed include common transition metals like copper and iron, the supply of these critical inputs is not subject to the same geopolitical constraints as rare earth elements or precious metals alone. The straightforward purification via column chromatography using standard solvent systems further ensures that production bottlenecks are minimized, facilitating reducing lead time for high-purity fluorescent probes and ensuring timely delivery to sensor manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram production without requiring specialized high-pressure equipment. The use of relatively benign solvents and the potential for solvent recovery systems aligns with stringent environmental regulations, reducing the carbon footprint of the manufacturing process. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying wastewater treatment and ensuring compliance with increasingly strict discharge standards. This environmental compatibility is a key asset for companies aiming to maintain a sustainable and responsible supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorescent compound. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing product lines or research pipelines.
Q: Does this fluorescent probe exhibit interference from other metal ions?
A: No, the compound demonstrates exceptional selectivity for Hg2+ ions. Competitive experiments confirm that the presence of other common metal ions such as Mg2+, Fe2+, Cu2+, and Zn2+ does not significantly alter the fluorescence response, ensuring accurate detection in complex matrices.
Q: Is the synthesized compound suitable for aqueous environmental monitoring?
A: Yes, the compound possesses good water solubility and chemical stability, making it highly effective for detecting mercury ions in water environment systems without requiring complex organic co-solvents that might interfere with biological or ecological samples.
Q: What are the primary advantages of this two-step synthesis route?
A: The route utilizes readily available, non-toxic raw materials and avoids complex multi-step protections or deprotections. The use of common transition metal catalysts and standard purification techniques like column chromatography facilitates easier scale-up and cost-effective manufacturing compared to traditional probe syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7,7'-Bis(4-Methoxyphenyl)-6,6'-Dimethyl-2,3'-Bi(1,8-Naphthyridine) Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the metal-catalyzed coupling steps described in this patent to ensure stringent purity specifications are met for every batch. With rigorous QC labs equipped for advanced structural characterization, we guarantee that our fluorescent probes meet the high sensitivity and selectivity standards required for critical mercury detection applications. We understand the nuances of handling air-sensitive intermediates and managing the purification of complex heterocyclic systems, ensuring a seamless transition from lab scale to full commercial supply.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall expenditure. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your project moves forward with the highest quality materials and the most efficient logistical support available in the industry.
