Advanced Sulindac Synthesis: Technical Breakthroughs for Commercial Scale API Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for non-steroidal anti-inflammatory drugs (NSAIDs), and the preparation method disclosed in patent CN100412057C represents a significant evolution in Sulindac synthesis. This technical documentation provides a comprehensive analysis of the novel route, which addresses critical bottlenecks found in legacy processes such as WO9747295 and US5902827. By fundamentally restructuring the synthetic sequence, this methodology achieves superior control over impurity profiles while drastically simplifying the operational complexity required for commercial production. The strategic elimination of noble metal catalysts and hazardous phosphoric reagents positions this route as a preferred choice for supply chain resilience and environmental compliance. Our analysis confirms that the integration of solvent-free decarboxylation and mild oxidation steps creates a distinct competitive advantage in the global API intermediate market. This report details the mechanistic underpinnings and commercial implications for R&D and procurement leaders evaluating high-purity Sulindac sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Sulindac have long been plagued by inefficiencies that escalate production costs and environmental liabilities. Conventional methods typically rely on a palladium-catalyzed hydrogenation step, which introduces the risk of heavy metal contamination and necessitates expensive purification protocols to meet stringent pharmaceutical standards. Furthermore, the cyclization step in legacy processes often employs large quantities of polyphosphoric acid, generating substantial volumes of phosphorus-containing wastewater that require complex treatment before disposal. The reliance on sodium periodate as a final oxidant not only inflates raw material costs but also introduces safety hazards associated with handling strong oxidizing agents on a large scale. Additionally, the use of excessive sodium methoxide in methanol solutions creates significant solvent recovery challenges, leading to high chemical oxygen demand (COD) in waste streams. These cumulative factors result in a fragmented process flow that is difficult to scale reliably without compromising yield or purity.
The Novel Approach
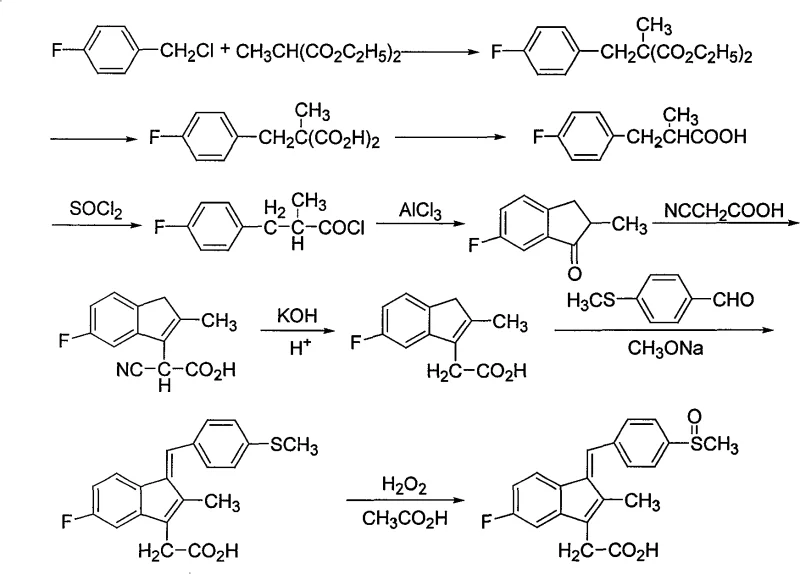
The innovative pathway outlined in the patent data overcomes these historical constraints through a series of strategic chemical modifications that enhance both efficiency and sustainability. By initiating the synthesis with a condensation reaction between p-fluorobenzyl chloride and diethyl methylmalonate, the process establishes a robust carbon framework without requiring precious metal catalysis. A pivotal improvement is the direct thermal decarboxylation step, which proceeds without solvents to quantitatively yield the propionic acid derivative, thereby eliminating solvent waste and recovery costs. The subsequent Friedel-Crafts acylation utilizes standard Lewis acids like aluminum trichloride or zinc chloride, which are more cost-effective and easier to handle than polyphosphoric acid. Finally, the replacement of sodium periodate with hydrogen peroxide or organic peracids in an organic acid medium significantly lowers reagent costs and improves reaction safety. This holistic redesign ensures a cleaner reaction profile and facilitates easier downstream processing for commercial manufacturing.
Mechanistic Insights into Friedel-Crafts Acylation and Oxidation
The core of this synthetic strategy lies in the precise execution of the Friedel-Crafts acylation and the final oxidation steps, which dictate the overall purity and yield of the Sulindac intermediate. In the acylation phase, the conversion of 3-(4-fluorophenyl)-2-methylpropionic acid to its acid chloride using thionyl chloride activates the molecule for intramolecular ring closure. The use of anhydrous aluminum trichloride or zinc chloride as a catalyst in halogenated solvents ensures high regioselectivity, producing 6-fluoro-2-methylindanone as a high-purity off-white solid rather than the viscous liquids often reported in literature. This solid state facilitates easier filtration and washing, directly contributing to the removal of inorganic salts and catalyst residues. The subsequent condensation with cyanoacetic acid and hydrolysis is carefully managed with ammonium acetate catalysis to minimize side reactions, ensuring the indene acetic acid backbone is formed with minimal structural impurities that could complicate final crystallization.
The final oxidation mechanism represents a critical control point for ensuring the correct sulfoxide functionality without over-oxidation to the sulfone. By employing hydrogen peroxide or peracids in an organic acid solvent such as acetic acid, the reaction conditions remain mild, typically between 30°C and 60°C. This temperature control is vital for preventing the degradation of the sensitive indene double bond while selectively oxidizing the thioether group. The stoichiometry of the oxidant is carefully balanced, with hydrogen peroxide usage ranging from 10% to 25% by weight relative to the substrate, ensuring complete conversion without excess oxidant that would require quenching. The use of organic acid as a solvent also aids in the solubility of the intermediate, promoting homogeneous reaction kinetics and consistent product quality. This mechanistic precision allows for a single recrystallization to achieve the required purity specifications, streamlining the final isolation process.
How to Synthesize Sulindac Efficiently
The implementation of this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and safety. The process begins with the alkylation of diethyl methylmalonate, followed by hydrolysis and a critical solvent-free decarboxylation step that demands precise temperature control between 130°C and 160°C. Subsequent acylation and condensation steps utilize standard industrial solvents like dichloromethane and toluene, which are easily recovered and recycled. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures, are outlined in the structured guide below to ensure reproducibility at scale.
- Condense p-fluorobenzyl chloride with diethyl methylmalonate using sodium alkoxide base.
- Hydrolyze the diester and perform solvent-free decarboxylation at high temperature.
- Execute Friedel-Crafts acylation followed by condensation and final oxidation with hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational benefits that extend beyond simple chemical yield. The elimination of palladium catalysts removes a significant cost driver and mitigates the supply risk associated with volatile precious metal markets. By avoiding polyphosphoric acid, the process significantly reduces the burden on wastewater treatment facilities, leading to lower environmental compliance costs and faster regulatory approvals for manufacturing sites. The use of hydrogen peroxide as a terminal oxidant offers a substantial cost advantage over sodium periodate, while also simplifying logistics due to the widespread availability and safer transport classification of peroxide solutions. These cumulative efficiencies create a more resilient supply chain capable of sustaining long-term production volumes without the bottlenecks typical of older synthetic routes.
- Cost Reduction in Manufacturing: The structural simplification of the synthesis route directly correlates with reduced operational expenditures through the removal of expensive reagents and complex purification stages. Eliminating the need for noble metal catalysts and expensive oxidants like sodium periodate drastically lowers the raw material cost per kilogram of the final API intermediate. Furthermore, the solvent-free decarboxylation step reduces energy consumption associated with solvent heating and recovery, contributing to overall utility savings. The ability to achieve high purity with a single recrystallization minimizes the loss of material during purification, effectively increasing the mass yield from starting materials. These factors combine to deliver a highly competitive cost structure for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrogen peroxide, thionyl chloride, and common organic solvents ensures a stable and diversified supply base. Unlike processes dependent on specialized catalysts or reagents with limited suppliers, this route utilizes materials that are readily available from multiple global vendors. The robustness of the reaction conditions, particularly the tolerance for standard industrial equipment in the acylation and oxidation steps, reduces the risk of production delays due to equipment failure or specialized maintenance requirements. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting strict delivery schedules for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with steps like the solvent-free decarboxylation and standard Friedel-Crafts acylation being easily transferable from pilot to commercial scale reactors. The reduction in hazardous waste generation, specifically the avoidance of phosphorus-containing wastewater and heavy metal residues, simplifies the environmental permitting process for new manufacturing lines. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing partner. The streamlined workflow supports rapid capacity expansion to meet market demand for Sulindac without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Sulindac synthesis method. These answers are derived directly from the patent specifications and practical manufacturing considerations to provide clarity for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production portfolios or for qualifying new suppliers for long-term contracts.
Q: How does this method improve upon traditional Sulindac synthesis?
A: This method eliminates the need for expensive palladium catalysts and polyphosphoric acid, significantly reducing heavy metal waste and phosphorus-containing wastewater while improving overall yield.
Q: What oxidants are used in the final step?
A: The process utilizes cost-effective hydrogen peroxide or organic peracids in an organic acid solvent, replacing expensive sodium periodate used in conventional routes.
Q: Is the decarboxylation step solvent-free?
A: Yes, the decarboxylation of 2-(4-fluorobenzyl)-2-methylmalonic acid is conducted directly at high temperatures without solvents, minimizing by-products and simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulindac Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging deep technical expertise to bring complex synthetic routes like the one described in CN100412057C to commercial reality. Our facility is equipped to handle the specific requirements of this process, from the high-temperature decarboxylation steps to the precise oxidation controls needed for sulfoxide formation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite procurement leaders to engage with our technical team to explore how this optimized synthesis can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how this route compares to your current sourcing strategy in terms of total landed cost and risk mitigation. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to secure a stable, high-quality supply of Sulindac intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
