Advanced Rhodium-Catalyzed Synthesis of Pyrazolone Benzodiazepine Intermediates for Commercial Scale-Up
Introduction to Next-Generation Heterocyclic Synthesis
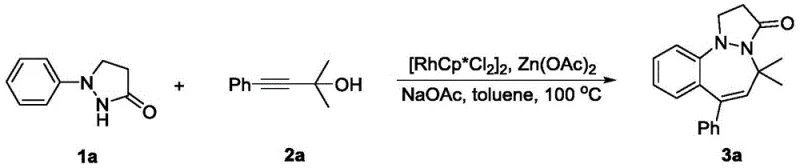
The pharmaceutical industry continuously demands efficient routes to complex nitrogen-containing fused heterocycles due to their prevalence in bioactive molecules exhibiting antibacterial, anticancer, and neuroprotective properties. Patent CN111675712B discloses a groundbreaking synthetic methodology for pyrazolone benzodiazepine compounds, utilizing a regioselective [4+3] cyclization strategy. This innovation addresses critical bottlenecks in traditional synthesis by employing readily available 1-arylpyrazolidinones and propargyl alcohol compounds as starting materials. The process leverages a robust rhodium catalytic system to achieve high atom economy and operational simplicity, marking a significant advancement over multi-step conventional pathways. For R&D teams seeking reliable pharmaceutical intermediate supplier partnerships, understanding the nuances of this catalytic transformation is essential for developing next-generation therapeutic candidates with improved purity profiles and reduced development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrazolone benzodiazepine scaffolds has been plagued by synthetic inefficiencies that hinder rapid drug discovery and commercial production. Traditional methods often rely on cumbersome multi-step sequences involving harsh reagents, extreme temperatures, and protection-deprotection strategies that significantly lower overall yields. These legacy processes frequently suffer from poor regioselectivity, leading to difficult-to-separate impurity profiles that complicate downstream purification and increase manufacturing costs. Furthermore, the reliance on exotic or unstable starting materials limits the structural diversity accessible to medicinal chemists, restricting the exploration of structure-activity relationships (SAR). The low atom economy associated with these older routes generates substantial chemical waste, conflicting with modern green chemistry principles and increasing the environmental burden on production facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct C-H activation strategy mediated by a rhodium catalyst to forge the seven-membered diazepine ring in a single pot. By reacting 1-arylpyrazolidinones with propargyl alcohols in the presence of [RhCp*Cl2]2 and specific acetate additives, the method achieves remarkable regioselectivity and efficiency. The reaction proceeds under relatively mild conditions, typically in toluene at 100 °C, avoiding the need for cryogenic temperatures or highly reactive organometallic reagents. This streamlined one-pot tandem reaction not only simplifies the operational workflow but also dramatically improves the overall yield, with representative examples achieving isolation yields as high as 88%. The broad substrate tolerance allows for the incorporation of diverse functional groups, enabling the rapid generation of chemical libraries for biological screening.
![General reaction scheme showing Rh-catalyzed [4+3] cyclization of 1-arylpyrazolidinones and propargyl alcohols](/insights/img/rhodium-catalyzed-pyrazolone-benzodiazepine-pharma-supplier-20260307080816-01.webp)
Mechanistic Insights into Rhodium-Catalyzed [4+3] Cyclization
The success of this transformation hinges on a sophisticated catalytic cycle initiated by the active Rh(III) species generated in situ from the dimeric precursor [RhCp*Cl2]2. The mechanism begins with the coordination of the rhodium center to the nitrogen atom of the pyrazolidinone, facilitating directed C-H activation at the ortho-position of the aryl ring to form a five-membered rhodacycle intermediate. Subsequent coordination and migratory insertion of the propargyl alcohol alkyne moiety into the Rh-C bond creates a new carbon-carbon bond while establishing the stereochemical framework of the product. A critical feature of this mechanism is the involvement of the hydroxyl group on the alkyne, which assists in the subsequent steps through hydrogen bonding or coordination, guiding the regioselectivity of the insertion event.
Following the insertion, the catalytic cycle proceeds through a unique C-O bond cleavage step, likely assisted by acetic acid generated in the system, which releases the oxygen functionality and forms a new rhodium-alkyl species. This intermediate undergoes reductive elimination to close the seven-membered diazepine ring, releasing the final pyrazolone benzodiazepine product and regenerating the active Rh(III) catalyst for the next turnover. Understanding this mechanistic pathway is vital for process optimization, as it highlights the importance of the acetate additives in proton shuttling and the specific role of the hydroxyl group in directing the reaction trajectory. This deep mechanistic understanding ensures that impurities arising from alternative insertion modes or incomplete cyclization are minimized, resulting in a high-purity pharmaceutical intermediate suitable for stringent regulatory requirements.
How to Synthesize Pyrazolone Benzodiazepine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and reproducibility. The standard protocol involves charging a reaction vessel with the 1-arylpyrazolidinone substrate and the propargyl alcohol coupling partner in a molar ratio ranging from 1:1 to 1:1.5. The choice of solvent is critical, with toluene identified as the optimal medium due to its ability to dissolve reactants effectively while supporting the catalytic activity at elevated temperatures. The addition of zinc acetate and sodium acetate serves a dual purpose: acting as bases to facilitate C-H activation and stabilizing the cationic rhodium species. Detailed standardized synthesis steps see the guide below.
- Combine 1-arylpyrazolidinone compound 1 and propargyl alcohol compound 2 in a reaction vessel with toluene solvent.
- Add the rhodium catalyst [RhCp*Cl2]2 along with zinc acetate and sodium acetate additives under inert atmosphere.
- Heat the mixture to 100 °C for 4 hours, then cool, filter, and purify via silica gel column chromatography to isolate product 3.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The reliance on simple, commercially available starting materials such as substituted pyrazolidinones and propargyl alcohols eliminates the need for custom-synthesized precursors, thereby reducing raw material costs and lead times. The robustness of the catalytic system, which tolerates a wide array of functional groups including halogens and trifluoromethyl groups, ensures that supply chain disruptions for specific niche reagents do not halt production. This flexibility allows manufacturers to pivot quickly between different analogues without retooling the entire process, enhancing the agility of the supply chain in response to market demands.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and protection groups significantly reduces the consumption of reagents, solvents, and labor hours. By consolidating the ring formation into a single catalytic event, the process minimizes waste generation and lowers the cost of goods sold (COGS). The use of earth-abundant rhodium catalysts in low loading amounts (0.02-0.03 equivalents) further optimizes the cost structure, making the production of these complex heterocycles economically viable for large-scale applications without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The mild reaction conditions (100 °C) and the use of common industrial solvents like toluene reduce the safety risks associated with high-pressure or cryogenic operations. This translates to fewer regulatory hurdles and easier integration into existing manufacturing infrastructure. The high regioselectivity of the reaction ensures consistent product quality batch after batch, reducing the risk of failed quality control tests and shipment delays. Suppliers can confidently guarantee delivery schedules knowing that the process is robust against minor variations in raw material quality.
- Scalability and Environmental Compliance: The high atom economy of this [4+3] cyclization aligns perfectly with green chemistry initiatives, minimizing the environmental footprint of the manufacturing process. The simplified workup procedure, often requiring only filtration and column chromatography, reduces the volume of aqueous waste and organic solvents needed for purification. This efficiency facilitates seamless scale-up from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production, ensuring that the technology can meet the growing global demand for these bioactive intermediates while maintaining strict environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these specifics helps in assessing the feasibility of integrating this chemistry into existing pipelines.
Q: What are the optimal reaction conditions for this cyclization?
A: The patent specifies using [RhCp*Cl2]2 as the catalyst with Zn(OAc)2 and NaOAc additives in toluene at 100 °C for optimal yields up to 88%.
Q: Can this method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent substrate scope, tolerating halogens (F, Cl, Br), electron-withdrawing groups (CF3, CN, NO2), and electron-donating groups (OMe, Me).
Q: Is the process suitable for large-scale manufacturing?
A: The use of common solvents like toluene, mild temperatures (100 °C), and commercially available catalysts suggests high feasibility for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolone Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic methodologies in accelerating drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in rhodium-catalyzed transformations allows us to optimize this specific [4+3] cyclization for maximum efficiency and cost-effectiveness, providing our partners with a competitive edge in the marketplace.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive innovation in your therapeutic programs.
