Advanced Palladium-Catalyzed Synthesis of Ethyl 2-Alkyl-4,4-Diaryl Trans-2-Butenoates for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex molecular scaffolds that serve as critical building blocks for bioactive compounds. Patent CN101735042B introduces a significant advancement in this domain by detailing a robust synthetic method for ethyl 2-alkyl-4,4-diaryl trans-2-butenoates. These compounds are not merely academic curiosities; they represent vital intermediates found in various natural products and possess substantial potential in medicinal chemistry applications. The core innovation lies in the utilization of palladium-catalyzed carbon-hydrogen bond functionalization, a technique that allows for the direct construction of carbon-carbon bonds without the need for pre-functionalized halide partners. This approach fundamentally shifts the paradigm from traditional cross-coupling methods, offering a more atom-economical pathway that aligns with modern green chemistry principles. By leveraging electron-rich aromatic compounds and 2,3-allenoic acid esters as primary feedstocks, this technology enables the high regio- and stereoselective generation of the target trans-alkene structures. For R&D directors and process chemists, this represents a tangible opportunity to streamline synthesis workflows while maintaining rigorous control over product purity and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of highly substituted alkene derivatives like ethyl 2-alkyl-4,4-diaryl trans-2-butenoates has been fraught with challenges related to step count and reagent availability. Traditional routes often rely on multi-step sequences involving the preparation of specific organometallic reagents or halogenated precursors, which inherently increases the overall cost of goods and extends production timelines. Furthermore, achieving high stereoselectivity for the trans-isomer typically requires stringent reaction conditions or specialized chiral auxiliaries that complicate the purification process. In many legacy methods, the formation of cis-isomers or other regioisomeric byproducts is a persistent issue, necessitating resource-intensive chromatographic separations that reduce overall throughput. The reliance on harsh bases or elevated temperatures in conventional protocols also poses safety risks and limits the compatibility with sensitive functional groups often present in advanced pharmaceutical intermediates. Consequently, manufacturers face significant bottlenecks when attempting to scale these processes, as the cumulative yield losses across multiple steps can render the final product commercially unviable for large-scale applications.
The Novel Approach
In stark contrast, the methodology outlined in CN101735042B offers a streamlined, direct functionalization strategy that bypasses many of these historical hurdles. By employing a palladium catalyst system in conjunction with trifluoroacetic acid, the reaction activates the inert C-H bonds of electron-rich aromatics directly, coupling them with the allenoate moiety in a single operational step. This direct approach eliminates the need for pre-halogenation, thereby reducing waste generation and simplifying the supply chain for raw materials. The reaction proceeds with remarkable stereocontrol, predominantly yielding the thermodynamically stable trans-isomer, which significantly eases the burden on downstream purification teams. Moreover, the mild reaction temperature of 40°C ensures that thermally labile substituents remain intact, expanding the scope of accessible chemical space for drug discovery programs. This novel pathway not only enhances the efficiency of the synthesis but also provides a versatile platform capable of accommodating a wide variety of substituents on both the aromatic ring and the alkene chain, making it an invaluable tool for the rapid generation of diverse compound libraries.
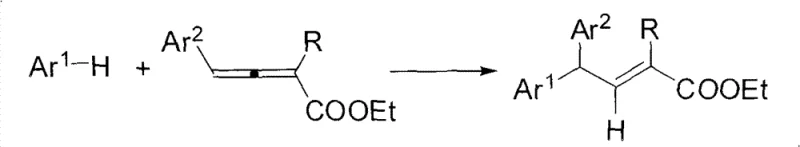
Mechanistic Insights into Palladium-Catalyzed C-H Functionalization
The mechanistic underpinning of this transformation involves a sophisticated catalytic cycle driven by the palladium center, which orchestrates the activation and coupling events with high precision. Initially, the palladium species coordinates with the electron-rich aromatic substrate, facilitating the cleavage of the carbon-hydrogen bond through an electrophilic palladation mechanism. This step is crucial as it generates an aryl-palladium intermediate that serves as the nucleophilic partner in the subsequent coupling. The presence of trifluoroacetic acid plays a dual role, acting both as a proton source to assist in the C-H activation and as a co-solvent to stabilize the cationic palladium species. Following the formation of the aryl-palladium complex, the 2,3-allenoic acid ester enters the coordination sphere, where it undergoes migratory insertion into the palladium-carbon bond. This insertion step is highly regioselective, favoring the formation of the specific carbon-carbon connectivity observed in the final product. The stereochemical outcome is dictated by the steric and electronic environment of the transition state, which preferentially stabilizes the trans-configuration of the resulting double bond. Finally, protonolysis of the vinyl-palladium intermediate releases the desired ethyl 2-alkyl-4,4-diaryl trans-2-butenoate and regenerates the active palladium catalyst, completing the cycle and allowing for turnover.
From an impurity control perspective, the specificity of this catalytic system is paramount for ensuring high-purity outputs suitable for pharmaceutical applications. The high regioselectivity minimizes the formation of ortho-substituted byproducts or alternative coupling isomers that could complicate the impurity profile. Additionally, the mild conditions prevent the degradation of sensitive functional groups such as methoxy or hydroxyl moieties, which might otherwise decompose under harsher acidic or basic environments. The use of a homogeneous catalyst system allows for uniform reaction kinetics throughout the batch, reducing the likelihood of localized hot spots that could lead to polymerization or side reactions of the allenoate starting material. For quality assurance teams, this means that the crude reaction mixture typically contains fewer unknown impurities, simplifying the analytical characterization and validation processes. The ability to tune the electronic properties of the aromatic substrate further allows chemists to optimize the reaction rate and selectivity, providing a robust handle for process optimization during scale-up activities.
How to Synthesize Ethyl 2-Alkyl-4,4-Diaryl Trans-2-Butenoates Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and solvent composition to maximize yield and reproducibility. The protocol dictates dissolving the electron-rich aromatic compound and the palladium dichlorodiacetonitrile catalyst in N,N-dimethylacetamide, followed by the addition of the 2,3-allenoic acid ester which has been rinsed with trifluoroacetic acid. The entire system is then heated to a moderate temperature of 40°C and stirred for approximately 15 hours to ensure complete conversion of the starting materials. Upon completion, the reaction is quenched using a saturated aqueous sodium bicarbonate solution to neutralize the acidic components, followed by extraction with ether and drying over anhydrous sodium sulfate. The final product is isolated through concentration and purification via flash column chromatography, yielding the target compound as either a solid or liquid depending on the specific substituents employed.
- Dissolve electron-rich aromatic compound and palladium dichlorodiacetonitrile catalyst in N,N-dimethylacetamide, then add 2,3-allenoate rinsed with trifluoroacetic acid.
- Heat the reaction mixture to 40°C and maintain stirring for approximately 15 hours to ensure complete conversion.
- Quench the reaction with saturated aqueous sodium bicarbonate, extract with ether, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers distinct strategic advantages that translate directly into operational efficiency and cost competitiveness. The primary benefit stems from the simplification of the synthetic route, which reduces the number of unit operations required to produce the final intermediate. By eliminating the need for pre-functionalized halide precursors, the supply chain becomes less dependent on specialized reagents that may have long lead times or volatile pricing structures. Instead, the process relies on readily available electron-rich aromatics and allenoates, which are commodity chemicals with stable market availability. This shift not only mitigates supply risk but also facilitates better inventory management and forecasting accuracy. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility costs and a smaller environmental footprint. The high selectivity of the reaction minimizes waste generation, thereby reducing the costs associated with waste disposal and environmental compliance. Collectively, these factors create a more resilient and cost-effective manufacturing process that enhances the overall value proposition for downstream customers.
- Cost Reduction in Manufacturing: The elimination of multi-step precursor synthesis significantly lowers the raw material costs and labor hours associated with production. By utilizing a direct C-H functionalization approach, the process avoids the expensive reagents and complex purification steps often required in traditional cross-coupling reactions. This streamlining results in a substantial reduction in the cost of goods sold, allowing for more competitive pricing in the global market. Additionally, the high atom economy of the reaction ensures that a greater proportion of the input mass is converted into the desired product, further optimizing resource utilization. The reduced need for extensive chromatographic purification due to high selectivity also lowers solvent consumption and processing time, driving down operational expenditures.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of common, commercially available starting materials rather than custom-synthesized intermediates. This reduces the dependency on single-source suppliers for niche reagents and shortens the overall lead time for raw material procurement. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification results. Consequently, manufacturers can maintain higher service levels and respond more敏捷 ly to fluctuations in market demand. The scalability of the process from gram to kilogram scales ensures that supply continuity can be maintained as customer requirements grow, providing a stable foundation for long-term partnerships.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with mild temperatures and standard solvent systems that are easily managed in large-scale reactors. The absence of hazardous reagents or extreme pressure conditions simplifies the engineering controls required for safe operation, facilitating faster technology transfer from R&D to production. From an environmental standpoint, the reduced waste generation and lower energy usage align with increasingly stringent regulatory standards for sustainable manufacturing. This compliance not only avoids potential fines but also enhances the corporate reputation among environmentally conscious clients. The ease of waste treatment due to the simpler impurity profile further streamlines the environmental management process, ensuring that production activities remain within permissible emission limits.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, providing clarity for potential partners evaluating its feasibility for their specific projects. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for assessing the fit of this methodology within existing manufacturing frameworks and for identifying opportunities for process optimization.
Q: What is the typical yield range for this palladium-catalyzed synthesis?
A: According to patent CN101735042B, the isolated yields for various substituted ethyl 2-alkyl-4,4-diaryl trans-2-butenoates range from 37% to 70%, depending on the specific electronic nature of the aryl substituents.
Q: Which substrates are compatible with this C-H functionalization method?
A: The method demonstrates broad substrate scope, accommodating electron-rich aromatics such as 1,3,5-trimethoxybenzene, anisole, and phenol, alongside 2,3-allenoic acid esters with methyl, ethyl, allyl, or benzyl groups at the R position.
Q: Does this process require harsh reaction conditions?
A: No, the process operates under mild conditions, specifically at a temperature of 40°C in a DMA and trifluoroacetic acid solvent system, which simplifies thermal management and enhances operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-Alkyl-4,4-Diaryl Trans-2-Butenoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in palladium-catalyzed transformations allows us to navigate the complexities of this specific synthesis, optimizing yields and minimizing impurities to provide you with a superior product. Whether you require custom synthesis services or bulk supply of established intermediates, our infrastructure is designed to support your growth from early-stage development to full-scale commercialization.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages this technology offers compared to your current supply sources. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to become your strategic partner in chemical innovation, providing not just products but comprehensive solutions that drive efficiency and value across your entire supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
