Advanced Indoloδ-sultone Synthesis: Technical Breakthroughs and Commercial Scalability for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking novel scaffolds that offer enhanced biological activity alongside manufacturability. Patent CN116178393A introduces a significant advancement in this domain by disclosing a new class of indoloδ-sultone compounds and a highly efficient preparation method. This technology addresses the critical need for diverse heterocyclic structures in drug discovery, particularly for antitumor applications. The core innovation lies in a tandem cyclization strategy that constructs the complex indoloδ-sultone skeleton in a single operational step from readily available starting materials. This approach not only simplifies the synthetic route but also aligns with modern green chemistry principles by reducing waste and energy consumption. For R&D directors and procurement specialists, this patent represents a valuable opportunity to access high-purity pharmaceutical intermediates with a robust and scalable supply chain foundation. The structural novelty of these compounds opens new avenues for medicinal chemistry optimization, potentially leading to next-generation therapeutics with improved efficacy profiles.
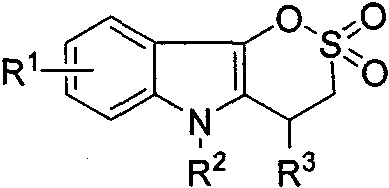
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex sultone-fused heterocycles has been fraught with challenges that hinder commercial viability and research progress. Conventional routes often require multi-step sequences involving harsh reaction conditions, such as high temperatures or strong acidic environments, which can degrade sensitive functional groups and lower overall yields. Furthermore, many existing methods rely on expensive transition metal catalysts that necessitate rigorous purification steps to remove trace metal residues, a critical requirement for pharmaceutical grade intermediates. These legacy processes frequently suffer from poor atom economy and generate substantial chemical waste, increasing both the environmental footprint and the cost of goods sold. The reliance on specialized reagents that are not commercially available in bulk also creates supply chain bottlenecks, leading to extended lead times and vulnerability to raw material shortages. Consequently, scaling these traditional methods to industrial levels often results in unpredictable outcomes and significant process safety risks.
The Novel Approach
In stark contrast, the methodology disclosed in CN116178393A offers a transformative solution by utilizing a base-mediated tandem reaction between indolin-3-one and substituted vinyl sulfonyl fluoride. This novel approach enables the simultaneous formation of two new chemical bonds in a single pot, drastically reducing the number of unit operations required. The reaction proceeds under remarkably mild conditions, typically at temperatures ranging from 20°C to 25°C, which minimizes energy consumption and thermal stress on the reactants. By avoiding the use of transition metals, this process eliminates the need for costly and time-consuming metal scavenging steps, thereby streamlining the downstream processing workflow. The use of common organic solvents and commercially available bases further enhances the practicality of this method for large-scale manufacturing. This streamlined protocol not only accelerates the timeline from laboratory discovery to pilot production but also ensures a more consistent and reliable supply of high-quality intermediates for drug development programs.
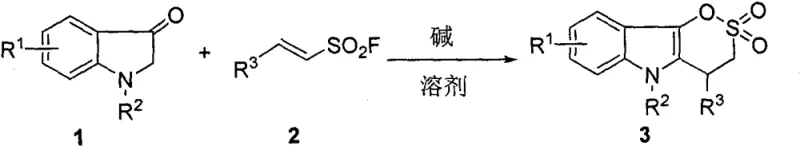
Mechanistic Insights into Base-Mediated Tandem Cyclization
The chemical elegance of this synthesis lies in its mechanistic pathway, which leverages the nucleophilic properties of indolin-3-one under basic conditions. The reaction initiates with a Michael addition, where the activated methylene group of the indolin-3-one attacks the electron-deficient double bond of the vinyl sulfonyl fluoride. This step is facilitated by the presence of an organic base, such as DBU, which deprotonates the indolin-3-one to generate a reactive nucleophile. Following the initial addition, an intramolecular cyclization occurs spontaneously, driven by the displacement of the fluoride leaving group and the formation of the stable sultone ring. This tandem sequence is highly efficient because it avoids the isolation of unstable intermediates, which can often decompose or undergo side reactions in stepwise protocols. The regioselectivity of the reaction is excellent, ensuring that the desired indoloδ-sultone scaffold is formed predominantly without significant isomeric byproducts. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for specific derivatives within this chemical class.
From an impurity control perspective, this mechanism offers distinct advantages over traditional cyclization methods. The mild reaction conditions significantly reduce the formation of thermal degradation products and polymerization byproducts that are common in high-temperature processes. Furthermore, the absence of transition metals eliminates a major class of genotoxic impurities, simplifying the regulatory filing process for any resulting drug candidates. The reaction profile is clean, as evidenced by the high yields reported across a broad scope of substrates with varying electronic properties. This robustness suggests that the process is tolerant to different functional groups, allowing for the synthesis of a diverse library of analogs for structure-activity relationship studies. For quality control teams, the predictable nature of this reaction means that establishing specification limits and validation protocols is more straightforward, ensuring consistent batch-to-batch quality essential for commercial supply.
How to Synthesize Indoloδ-sultone Efficiently
Implementing this synthesis route in a production environment requires careful attention to reagent quality and process parameters to maximize efficiency. The protocol is designed to be user-friendly, utilizing standard laboratory and plant equipment without the need for specialized high-pressure or cryogenic setups. Operators simply combine the indolin-3-one derivative and the substituted vinyl sulfonyl fluoride in a suitable organic solvent, followed by the controlled addition of the base. Reaction progress can be easily monitored using standard analytical techniques such as TLC or HPLC, allowing for precise determination of the endpoint to prevent over-reaction. The detailed standardized synthesis steps see below guide.
- Combine indolin-3-one and substituted vinyl sulfonyl fluoride in a reaction vessel with an organic solvent.
- Add a catalytic amount of organic or inorganic base, such as DBU, to initiate the tandem cyclization reaction.
- Stir the mixture at mild temperatures until completion, then perform standard workup and purification to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant reduction of manufacturing costs driven by the simplification of the process workflow. By consolidating multiple synthetic steps into a single tandem reaction, the consumption of solvents, reagents, and labor hours is drastically reduced, leading to a lower cost of goods sold. The elimination of transition metal catalysts further contributes to cost savings by removing the expense of precious metals and the associated purification resins required for their removal. Additionally, the use of readily available starting materials ensures a stable supply chain that is less susceptible to market volatility or geopolitical disruptions. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined one-pot process significantly lowers operational expenses by reducing the number of isolation and purification stages required. This efficiency gain means less waste generation and lower utility costs associated with heating, cooling, and solvent recovery. The avoidance of expensive catalysts and specialized reagents further enhances the economic viability of producing these high-value intermediates at scale. Consequently, this route offers a competitive pricing structure that can improve margins for downstream drug manufacturers while maintaining high quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as indolin-3-one and common organic bases ensures that raw material sourcing is robust and diversified. Unlike processes dependent on custom-synthesized building blocks with long lead times, this method allows for rapid procurement and inventory management. The mild reaction conditions also reduce the risk of process safety incidents, ensuring uninterrupted plant operations and consistent output. This stability is essential for building long-term partnerships with key stakeholders who require guaranteed supply continuity for their critical drug development programs.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates easy scale-up from laboratory grams to multi-ton commercial production without significant re-engineering. The reduced solvent usage and absence of heavy metals align with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only minimizes the environmental footprint but also simplifies waste disposal procedures and regulatory compliance reporting. Companies adopting this technology can demonstrate a commitment to sustainable manufacturing practices, which is becoming a key differentiator in the global chemical marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of indoloδ-sultone compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing portfolios. The responses cover aspects ranging from reaction scalability to biological potential, providing a comprehensive overview for decision-makers.
Q: What are the primary advantages of this new indoloδ-sultone synthesis route?
A: The primary advantages include mild reaction conditions, high yields, and a one-step tandem process that eliminates the need for complex multi-step sequences or expensive transition metal catalysts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of readily available raw materials, common organic solvents, and mild temperature requirements makes this methodology highly scalable for industrial manufacturing environments.
Q: What is the biological potential of the synthesized indoloδ-sultone compounds?
A: Preliminary data indicates significant antitumor activity against specific cell lines such as HepG2 and Caco-2, suggesting strong potential for development as novel therapeutic agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoloδ-sultone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the indoloδ-sultone scaffold in modern medicinal chemistry and are committed to supporting its development from bench to bedside. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We understand the critical nature of time-to-market in the pharmaceutical industry and have optimized our workflows to deliver high-quality intermediates with consistent reliability. Our team of expert chemists is ready to assist in process optimization to further enhance yield and efficiency for your specific needs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug discovery program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your supply chain objectives. By partnering with us, you gain access to a reliable source of high-purity indoloδ-sultone derivatives that can accelerate your research and development timelines. Let us help you navigate the complexities of chemical manufacturing so you can focus on delivering life-saving therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
