Industrial Scale-Up of Octafluorocyclopentene: A Green Route for Electronic Chemical Manufacturing
Introduction to Advanced Octafluorocyclopentene Synthesis
The global demand for high-performance electronic chemicals continues to surge, driven by the relentless miniaturization of semiconductor devices and the expanding market for organic photochromic materials. At the forefront of this technological evolution is octafluorocyclopentene (perfluorocyclopentene), a critical compound valued for its exceptional etching properties and utility as a precursor in advanced material synthesis. The industrial production method disclosed in patent CN108276243B represents a significant paradigm shift in how this valuable intermediate is manufactured, moving away from hazardous, multi-step legacy processes toward a streamlined, environmentally benign protocol. This patent details a robust two-stage synthesis starting from readily available cyclopentene, utilizing temperature-programmed chlorination followed by a high-temperature substitution reaction with anhydrous potassium fluoride. By addressing the critical pain points of raw material availability, reaction safety, and waste management, this technology offers a compelling value proposition for manufacturers seeking to secure a reliable supply chain for next-generation etching gases and fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octafluorocyclopentene has been plagued by significant operational hazards and inefficiencies that hinder scalable commercial production. Traditional literature methods often rely on the depolymerization of dicyclopentadiene at extreme temperatures ranging from 300°C to 350°C to generate cyclopentadiene monomers, a process that is not only energy-intensive but also poses severe safety risks due to the thermal instability of the monomers. Furthermore, subsequent chlorination of these monomers frequently results in uncontrolled polymerization, leading to viscous reaction systems with high tar content and abysmal target product yields, often reported as low as 5%. Alternative routes involving gas-phase fluorination with anhydrous hydrogen fluoride (HF) require specialized, corrosion-resistant equipment and present acute toxicity risks, while generating substantial amounts of acidic waste that complicate environmental compliance. These legacy pathways, characterized by their complex separation requirements and reliance on unavailable or dangerous intermediates like nonafluorocyclopentane chloride, create bottlenecks that limit supply continuity and inflate manufacturing costs.
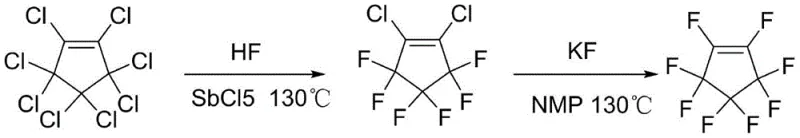
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel industrial method introduced in CN108276243B leverages a direct, solvent-free chlorination strategy followed by a liquid-phase halogen exchange reaction. The process initiates with the controlled addition of chlorine to cyclopentene, employing a sophisticated temperature programming regimen that ramps from ambient conditions to 210°C. This precise thermal control ensures complete chlorination to octachlorocyclopentene while suppressing the formation of polymeric byproducts, achieving conversion rates where intermediate content is less than 2%. The subsequent fluorination step utilizes solid anhydrous potassium fluoride in sulfolane, a high-boiling polar aprotic solvent that facilitates efficient nucleophilic substitution without the need for gaseous HF. This approach not only simplifies the reactor engineering requirements by eliminating the need for exotic alloys resistant to anhydrous HF but also enables a closed-loop solvent recovery system. The result is a streamlined workflow that transforms commodity chemicals into high-value electronic materials with a separation yield reaching 45-55% and product purity exceeding 99%, fundamentally redefining the economic and safety landscape of octafluorocyclopentene manufacturing.
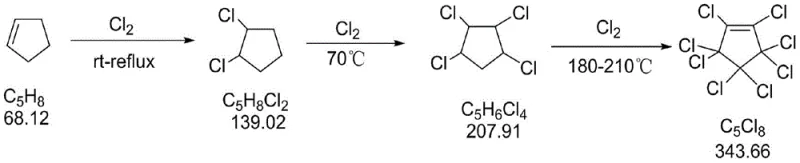
Mechanistic Insights into Temperature-Programmed Chlorination and Halex Reaction
The core innovation of this synthesis lies in the meticulous control of reaction kinetics during the chlorination phase, which serves as the foundation for high-yield fluorination. The mechanism begins with the electrophilic addition of chlorine across the double bond of cyclopentene at room temperature, forming 1,2-dichlorocyclopentane. As the temperature is progressively increased to 70°C and subsequently to the 180-210°C range, the reaction transitions into a radical substitution regime. This temperature programming is critical; it provides the necessary activation energy to overcome the steric hindrance of adding multiple chlorine atoms to the cyclopentane ring while simultaneously managing the exothermic nature of the chlorination to prevent thermal runaway. By maintaining a chlorine-to-cyclopentene molar ratio of 1:18-20 and monitoring the reaction until over-chlorination states are minimized, the process ensures the formation of octachlorocyclopentene with minimal structural isomers or degradation products. This high-fidelity intermediate is essential for the subsequent step, as impurities here would propagate through the fluorination stage, complicating purification.
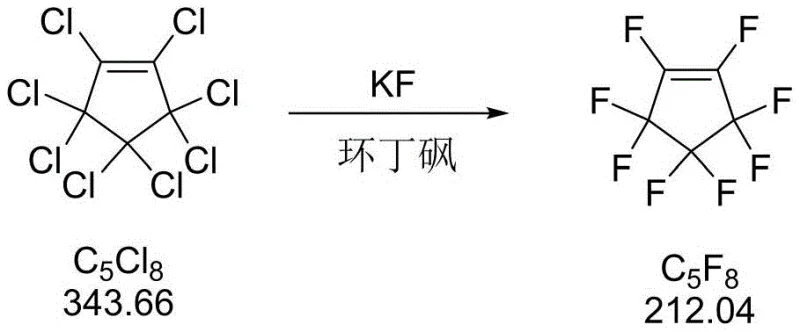
The second stage involves a classic Halex (halogen exchange) mechanism facilitated by the unique solvation properties of sulfolane. In this polar environment, the fluoride ion from potassium fluoride is sufficiently activated to act as a potent nucleophile, attacking the electron-deficient carbon centers of the perchlorinated ring. The reaction proceeds via an SNAr-like pathway where the chloride leaving groups are sequentially displaced by fluoride ions. The use of sulfolane is particularly advantageous as it stabilizes the transition state and remains stable at the required reaction temperatures of 140-170°C. Crucially, the process incorporates a post-reaction workup where the kettle residue is neutralized with alkali, converting residual HF and unreacted KF into soluble salts that can be removed via centrifugation. This step is vital for controlling the ionic impurity profile, ensuring the final product meets the stringent chloride ion detection limits of less than 0.1% required for semiconductor applications. The ability to recover and reuse the sulfolane solvent via reduced pressure distillation further underscores the mechanistic efficiency of this cycle, minimizing waste and maximizing atom economy.
How to Synthesize Octafluorocyclopentene Efficiently
The synthesis of octafluorocyclopentene via this patented route requires precise adherence to thermal profiles and stoichiometric ratios to ensure safety and maximize yield. The process is divided into two distinct operational phases: the initial solvent-free chlorination to generate the perchlorinated precursor, and the subsequent liquid-phase fluorination in a polar solvent system. Operators must carefully manage the introduction of chlorine gas to match the consumption rate, preventing pressure buildup while ensuring complete conversion. The detailed standardized synthesis steps, including specific equipment setups, safety protocols for handling chlorine and hot sulfolane, and precise distillation parameters for product isolation, are outlined below to guide technical teams in replicating this high-efficiency pathway.
- Perform temperature-programmed chlorination of cyclopentene with chlorine gas, ramping from room temperature to 210°C to obtain octachlorocyclopentene.
- React the purified octachlorocyclopentene with anhydrous potassium fluoride in sulfolane solvent at 140-170°C.
- Distill the reaction mixture under normal pressure to collect octafluorocyclopentene, cooling the product to -10 to 0°C, and recover solvents from the residue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis methodology translates into tangible strategic advantages regarding cost stability and operational resilience. By shifting the raw material base from specialized, hard-to-source intermediates to bulk commodity chemicals like cyclopentene and chlorine, the supply chain becomes significantly more robust against market volatility. The elimination of gas-phase HF handling removes the need for costly, specialized infrastructure maintenance and reduces the regulatory burden associated with storing large quantities of highly toxic gases. Furthermore, the integrated solvent recovery system, which allows for the recycling of sulfolane and ethanol with high efficiency, drastically reduces the volume of chemical waste requiring disposal. This reduction in waste treatment overhead, combined with the higher overall yield compared to traditional depolymerization routes, results in a substantially lower cost of goods sold (COGS), enabling more competitive pricing structures for downstream electronic chemical applications.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the replacement of expensive and hazardous reagents with cost-effective alternatives. Utilizing solid potassium fluoride instead of anhydrous hydrogen fluoride eliminates the capital expenditure associated with Hastelloy reactors and complex scrubbing systems required for HF containment. Additionally, the solvent-free nature of the initial chlorination step reduces the energy load associated with heating and cooling large solvent volumes, while the high recovery rate of sulfolane in the second step minimizes recurring raw material expenses. These factors collectively drive down the variable costs per kilogram of produced octafluorocyclopentene, enhancing margin potential for manufacturers operating in the competitive electronic materials sector.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the primary inputs—cyclopentene, chlorine, and potassium fluoride—are widely produced commodity chemicals with established global supply networks. Unlike legacy methods that depend on the thermal cracking of dicyclopentadiene, a process prone to yield fluctuations and safety shutdowns, this continuous-flow compatible chlorination method offers consistent throughput. The simplified purification train, which relies on standard distillation and centrifugation rather than complex chromatographic separations, further reduces the risk of production bottlenecks. This operational stability ensures that procurement teams can secure long-term supply agreements with greater confidence, mitigating the risk of shortages that could disrupt semiconductor fabrication lines.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns with modern green chemistry principles by minimizing waste generation and maximizing resource efficiency. The closed-loop solvent recovery system significantly lowers the facility's environmental footprint, reducing the volume of hazardous wastewater and solid waste sent for treatment. The absence of heavy metal catalysts and the use of less corrosive reagents simplify the end-of-life processing for reactor components and reduce the liability associated with environmental remediation. These attributes facilitate easier permitting for capacity expansion and ensure compliance with increasingly stringent international environmental regulations, making the technology future-proof for large-scale industrial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this octafluorocyclopentene production technology. These insights are derived directly from the experimental data and process descriptions within the patent, providing clarity on yield expectations, purity specifications, and operational safety. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing incoming inspection criteria for the final electronic grade product.
Q: What are the primary advantages of this octafluorocyclopentene synthesis route over traditional methods?
A: This route eliminates the need for hazardous gas-phase HF fluorination and avoids the dangerous high-temperature depolymerization of dicyclopentadiene required in older methods. It utilizes readily available cyclopentene and solid KF, offering a safer, more environmentally friendly process with simplified solvent recovery.
Q: How is product purity controlled in this manufacturing process?
A: High purity (>99%) is achieved through precise temperature programming during chlorination to minimize tar formation, followed by efficient fractional distillation during the fluorination step. The process also includes a specific post-reaction neutralization and filtration step to remove inorganic salts and residual ions.
Q: Is the solvent system sustainable for large-scale production?
A: Yes, the process uses sulfolane as a high-boiling polar solvent which can be recovered via reduced pressure distillation from the kettle residue after neutralization. The patent indicates a solvent recovery rate of over 90%, significantly reducing waste generation and raw material costs for continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octafluorocyclopentene Supplier
As the semiconductor and fine chemical industries continue to evolve, the demand for high-purity etching gases and functional intermediates like octafluorocyclopentene is accelerating. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver solutions that meet the rigorous demands of modern manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in the pilot phase or full-scale manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of octafluorocyclopentene meets the exacting standards required for semiconductor etching and advanced material synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced synthesis capabilities can drive value and reliability for your organization.
