Breakthrough Synthesis of Pseudo-Ginsenoside Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access rare bioactive compounds, particularly those derived from natural products like ginseng. Patent CN106866769B introduces a transformative preparation method for pseudo-ginsenoside derivatives, specifically targeting the E-type and Z-type configurations of pseudo-ginsenoside Rg2, Rh1, and PPT. This technology addresses the critical bottleneck of low natural abundance by utilizing a sophisticated chemical conversion strategy rather than relying solely on extraction. The core innovation lies in the strategic protection of hydroxyl groups followed by a novel acid-catalyzed side chain transformation under controlled low-temperature conditions. This approach not only enhances the conversion rate significantly but also ensures the structural integrity of the complex saponin backbone. For R&D directors and procurement specialists, this patent represents a viable route to secure high-purity intermediates that were previously difficult to source in commercial quantities. The ability to directionally prepare these rare derivatives from abundant raw materials like ginsenoside Re or Rg1 marks a substantial leap forward in synthetic biology and organic chemistry applications within the nutraceutical and pharmaceutical sectors.
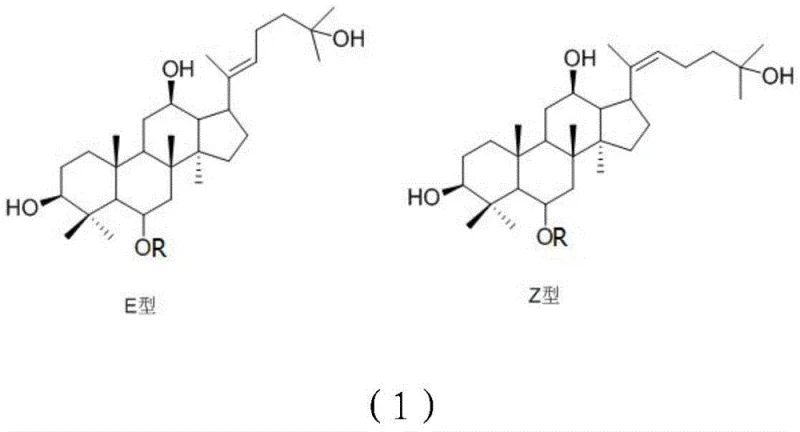
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of rare ginsenosides and their pseudo-derivatives has been plagued by inefficient extraction and degradation techniques that fail to meet modern industrial standards. Traditional methods often rely on harsh alkali degradation or non-specific acid hydrolysis, which frequently result in the destruction of the sensitive saponin structure and extremely low yields. For instance, prior art indicates that acid hydrolysis methods for obtaining specific side-chain modified ginsenosides like Rh2 often yield less than 1%, rendering them economically unfeasible for large-scale manufacturing. Furthermore, conventional processes struggle with stereoselectivity, often producing complex mixtures of isomers that are difficult and costly to separate. The reliance on high-temperature and high-pressure conditions in older protocols also introduces significant safety hazards and increases energy consumption, which negatively impacts the overall cost of goods sold. These limitations create a supply chain vulnerability for pharmaceutical companies requiring consistent, high-quality batches of these bioactive intermediates for drug development and clinical trials.
The Novel Approach
The methodology disclosed in the patent data offers a robust solution by implementing a multi-step synthesis that prioritizes selectivity and yield through chemical protection strategies. By first protecting the hydroxyl groups of the starting panaxatriol saponins, the process prevents unwanted side reactions during the critical conversion phase. The subsequent acid-catalyzed reaction is conducted at low temperatures, typically ranging from -50°C to 25°C depending on the specific substrate, which preserves the stereochemistry of the molecule. This novel approach allows for the simultaneous occurrence of elimination and alcoholization processes at different positions of the ginsenoside side chain in a single reaction step. The result is a streamlined workflow that achieves conversion rates exceeding 60% for target compounds like E-type pseudo-ginsenoside Rg2. This drastic improvement in efficiency transforms the economic viability of producing these rare compounds, making them accessible for broader commercial applications in health and wellness products.
Mechanistic Insights into Acid-Catalyzed Side Chain Conversion
The chemical mechanism underpinning this synthesis is a testament to precise organic manipulation, beginning with the acetylation of the starting material to mask reactive hydroxyl sites. This protection step is crucial as it directs the subsequent acid catalysis specifically towards the side chain modification without compromising the glycosidic bonds or the triterpenoid core. During the acid-catalyzed conversion, the protonation of specific oxygen atoms facilitates a rearrangement that alters the side chain structure, effectively converting the native ginsenoside into its pseudo-form. The reaction conditions are meticulously controlled to favor the formation of the thermodynamically stable E-type configuration while still allowing for the isolation of the Z-type isomer. This level of control is achieved through the careful selection of acid catalysts, such as sulfuric acid or hydrochloric acid, and the regulation of reaction time and temperature. The mechanistic pathway ensures that the resulting derivatives possess the desired pharmacological activity profiles associated with the modified side chain configurations.
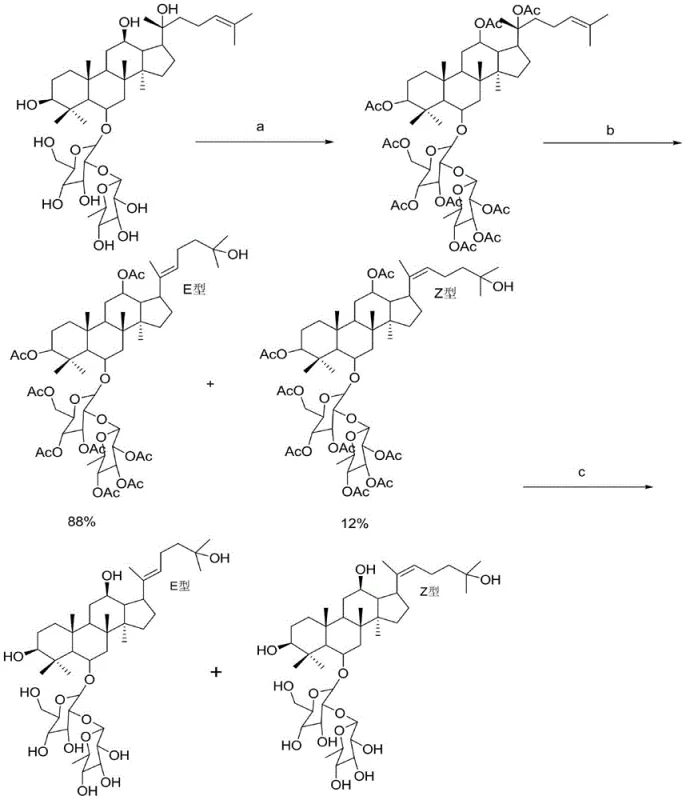
Impurity control is inherently built into this synthetic route through the use of specific purification protocols following the reaction. The process employs silica gel column chromatography with a tailored eluent system comprising chloroform, methanol, ethyl acetate, and water to separate the target pseudo-ginsenosides from byproducts and unreacted starting materials. This chromatographic separation is further refined by preparative liquid phase separation, utilizing a methanol-water mixture to achieve high-resolution isolation of the E-type and Z-type isomers. The final recrystallization step using methanol containing a specific percentage of water ensures the removal of trace solvents and residual impurities, resulting in a product that meets stringent purity specifications. For quality assurance teams, this multi-stage purification strategy provides a reliable framework for consistent batch-to-batch reproducibility, which is essential for regulatory compliance in pharmaceutical manufacturing. The ability to consistently produce high-purity materials reduces the risk of downstream processing failures and ensures the safety and efficacy of the final therapeutic or nutraceutical products.
How to Synthesize Pseudo-Ginsenoside Rg2 Efficiently
The synthesis of pseudo-ginsenoside Rg2 via this patented route involves a sequence of well-defined chemical transformations that can be adapted for industrial scale-up. The process begins with the dissolution of the starting ginsenoside in an organic solvent, followed by the addition of acetic anhydride and pyridine to effect hydroxyl protection. Once the acetylated intermediate is obtained, it undergoes the critical acid-catalyzed conversion step where the side chain structure is modified under low-temperature reflux conditions. Following the conversion, the protecting groups are removed using an alkali solution, and the crude product is subjected to rigorous purification. Detailed standardized synthesis steps see the guide below.
- Hydroxyl Protection: React panaxatriol saponins with acetic anhydride and pyridine to form acetylated intermediates.
- Acid Catalytic Conversion: Treat acetylated products with dilute acid solution at low temperatures to induce side chain transformation.
- Deprotection and Purification: Remove protecting groups using alkali solution and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis technology offers substantial benefits by mitigating the risks associated with natural extraction variability. The reliance on abundant starting materials like ginsenoside Re or Rg1, which are readily available from ginseng extracts, ensures a stable and continuous supply of raw materials. This stability is crucial for maintaining production schedules and avoiding delays caused by seasonal fluctuations in crop yields or extraction efficiencies. Furthermore, the use of common industrial solvents and reagents simplifies the sourcing process and reduces the dependency on specialized or hazardous chemicals that might face regulatory restrictions. The simplified operational steps also translate to lower labor costs and reduced equipment complexity, making the process highly attractive for contract development and manufacturing organizations looking to optimize their production lines.
- Cost Reduction in Manufacturing: The elimination of complex extraction procedures and the significant increase in reaction yield directly contribute to a lower cost of goods. By avoiding the need for extensive purification of low-yield natural extracts, manufacturers can allocate resources more efficiently. The use of universal reagents means that procurement teams can leverage existing supplier relationships and bulk purchasing power to further drive down material costs. Additionally, the reduced reaction time and milder conditions lower energy consumption, adding another layer of cost efficiency to the overall manufacturing process. These factors combined create a compelling economic case for adopting this synthetic route over traditional methods.
- Enhanced Supply Chain Reliability: The robustness of this chemical synthesis pathway ensures that supply chains are less vulnerable to external disruptions. Since the process does not rely on the direct isolation of rare compounds from limited natural sources, production can be scaled up or down based on market demand without compromising quality. The ability to produce both E-type and Z-type isomers from the same general process flow adds flexibility to the product portfolio, allowing suppliers to meet diverse customer requirements. This reliability is a key value proposition for pharmaceutical companies that require guaranteed delivery timelines for their clinical and commercial programs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction vessels and separation techniques that are easily transferable from laboratory to pilot and commercial scales. The waste streams generated are primarily composed of common organic solvents and aqueous solutions, which can be managed using standard industrial waste treatment protocols. This aligns with increasing global demands for environmentally sustainable manufacturing practices. The high atom economy of the conversion step further minimizes waste generation, supporting corporate sustainability goals and reducing the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of pseudo-ginsenoside derivatives. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating these materials into their supply chains.
Q: What is the primary advantage of the acid-catalyzed method for pseudo-ginsenoside synthesis?
A: The primary advantage is the significantly improved yield, often exceeding 60%, compared to traditional alkali degradation methods which often yield less than 1%. This method also allows for the specific formation of both E-type and Z-type configurations.
Q: Are the reagents used in this synthesis process commercially available?
A: Yes, the process utilizes common organic solvents and reagents such as acetic anhydride, pyridine, dichloromethane, and dilute sulfuric or hydrochloric acid, ensuring high universal applicability and ease of sourcing for industrial production.
Q: How does this method impact the purity of the final ginsenoside derivatives?
A: By employing a specific hydroxyl protection strategy followed by controlled low-temperature acid catalysis, the method minimizes side reactions and degradation of the saponin core, resulting in high-purity E-type and Z-type pseudo-ginsenosides suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pseudo-Ginsenoside Rg2 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our technical team has extensively analyzed the synthetic pathways described in patent CN106866769B and is fully equipped to implement these processes at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of pseudo-ginsenoside Rg2, Rh1, or PPT meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to complex synthesis challenges.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By leveraging our expertise in fine chemical synthesis and our commitment to quality, we can help you accelerate your product development timelines and bring innovative solutions to market faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
